Maxxcare Company Limited
Pharmaceutical Importer · Vietnam · Antifungals Focus · $1.9M Total Trade · DGFT Verified
Maxxcare Company Limited is a pharmaceutical importer based in Vietnam with a total trade value of $1.9M across 2 products in 2 therapeutic categories. Based on 41 verified import shipments from Indian Customs (DGFT) records, the company actively imports across multiple product segments. Maxxcare Company Limited sources from 5 verified Indian suppliers, with Inventia Healthcare Limited accounting for 63.3% of imports.
Maxxcare Company Limited — Import Portfolio & Supplier Network
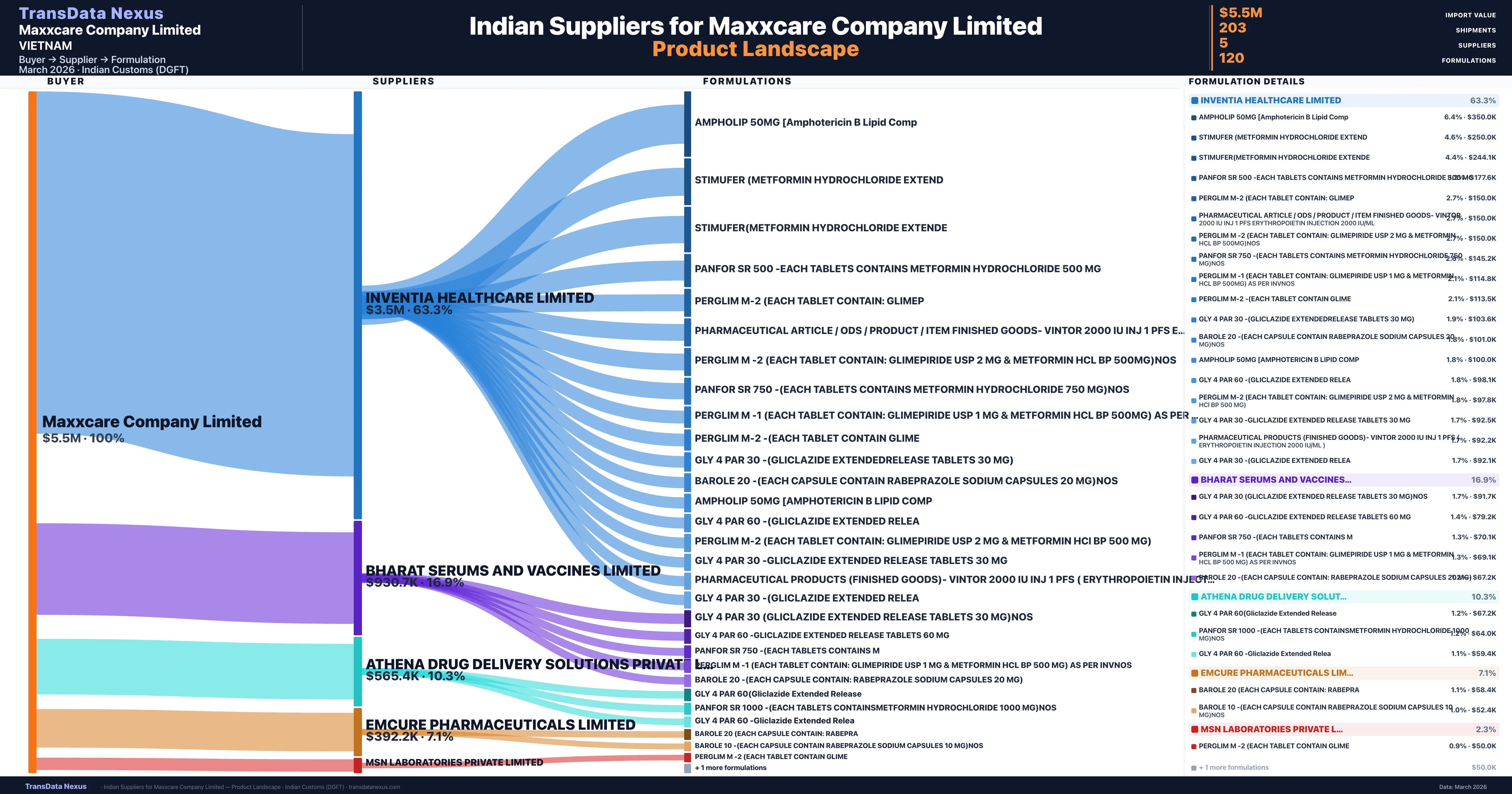
Who Are the Verified Indian Suppliers to Maxxcare Company Limited?
Customs-verified supplier relationships from Indian DGFT records
| Supplier | Value | Shipments | Share |
|---|---|---|---|
| Inventia Healthcare Limited | $3.5M | 132 | 63.3% |
| Bharat Serums And Vaccines Limited | $930.7K | 28 | 16.9% |
| Athena Drug Delivery Solutions Private Limited | $565.4K | 13 | 10.3% |
| Emcure Pharmaceuticals Limited | $392.2K | 18 | 7.1% |
| Msn Laboratories Private Limited | $125.1K | 12 | 2.3% |
Maxxcare Company Limited sources from 5 verified Indian suppliers across 120 distinct formulations. The supply base is diversified across 5 suppliers, reducing single-source dependency risk.
What Formulations Does Maxxcare Company Limited Import?
| Formulation | Value | Ships |
|---|---|---|
| Ampholip 50MG [amphotericin b lipid | $350.0K | 7 |
| Stimufer (metformin hydrochloride | $250.0K | 5 |
| Stimufer(metformin hydrochloride | $244.1K | 5 |
| Panfor sr 500 -Each tablets contains metformin hydrochloride 500 MG | $177.6K | 4 |
| Perglim m-2 (Each tablet contain | $150.0K | 3 |
| Pharmaceutical article / ods / product / item finished goods- vintor 2000 IU INJ 1 pfs erythropoietin injection 2000 IU/ML | $150.0K | 3 |
| Perglim m -2 (Each tablet contain: glimepiride USP 2 MG & metformin hcl BP 500MG | $150.0K | 3 |
| Panfor sr 750 -(Each tablets contains metformin hydrochloride 750 MG | $145.2K | 3 |
| Perglim m -1 (Each tablet contain: glimepiride USP 1 MG & metformin hcl BP 500MG) as per | $114.8K | 3 |
| Perglim m-2 -(Each tablet contain | $113.5K | 3 |
| Gly 4 par 30 -(gliclazide extendedrelease tablets 30 MG) | $103.6K | 6 |
| Barole 20 -(Each capsule contain rabeprazole sodium capsules 20 MG | $101.0K | 6 |
| Ampholip 50MG [amphotericin b lipid | $100.0K | 2 |
| Gly 4 par 60 -(gliclazide extended | $98.1K | 3 |
| Perglim m-2 (Each tablet contain: glimepiride USP 2 MG & metformin hcl BP 500 MG) | $97.8K | 2 |
Maxxcare Company Limited imports 120 distinct pharmaceutical formulations. Showing top 15 by value. For full formulation-level data, contact TransData Nexus.
What Products Does Maxxcare Company Limited Import?
Top Products by Import Value
Maxxcare Company Limited Therapeutic Categories — 2 Specializations
Maxxcare Company Limited imports across 2 therapeutic categories, with Antifungals (53.3%), Diabetes & Endocrine (46.7%), representing the largest segments. The portfolio is concentrated — top 5 products = 100% of total imports.
Antifungals
1 products · 53.3% · $1.0M
Diabetes & Endocrine
1 products · 46.7% · $876.0K
Import Portfolio — Top 2 by Import Value
| # | Product | Category | Value | Ships | Share | Rk |
|---|---|---|---|---|---|---|
| 1 | Amphotericin | Antifungals | $1.0M | 20 | 1.1% | 6 |
| 2 | Glimepiride | Diabetes & Endocrine | $876.0K | 21 | 0.6% | 11 |
Maxxcare Company Limited imports 2 pharmaceutical products across 2 categories into Vietnam totaling $1.9M.
Key Metrics
Top Categories
Indian Suppliers
Related Trade Data
Need Detailed Data?
Shipment-level records, supplier connections & pricing for Maxxcare Company Limited.
Request DemoMaxxcare Company Limited — Corporate Profile & Information
Company type, headquarters, distribution network, and industry role
1Company Overview
Maxxcare Company Limited, established on June 27, 2018, is a Vietnamese pharmaceutical importer headquartered in Tan Binh District, Ho Chi Minh City. The company specializes in the wholesale distribution of pharmaceutical products, focusing on finished formulations such as tablets, capsules, syrups, and injections. With a charter capital of VND 9.2 billion, Maxxcare demonstrates financial stability to support its operations.
Maxxcare's role in Vietnam's pharmaceutical distribution network is that of a primary wholesaler, sourcing finished pharmaceutical formulations from international suppliers and distributing them within the domestic market. This positioning enables the company to meet the growing demand for diverse pharmaceutical products across Vietnam.
2Distribution Network
Maxxcare Company Limited operates from its headquarters in Tan Binh District, Ho Chi Minh City, Vietnam. While specific details regarding additional warehouse locations and logistics capabilities are not publicly disclosed, the company's strategic location in Ho Chi Minh City suggests access to major transportation networks, facilitating efficient distribution throughout Vietnam.
3Industry Role
In Vietnam's pharmaceutical supply chain, Maxxcare Company Limited functions primarily as a wholesaler. By importing finished pharmaceutical formulations from international suppliers, the company plays a crucial role in ensuring the availability of a wide range of pharmaceutical products to meet domestic demand. This role is vital for maintaining the supply of essential medications across the country.
Supplier Relationship Intelligence — Maxxcare Company Limited
Sourcing concentration, supply chain resilience, and strategic implications
1Sourcing Concentration Analysis
Maxxcare Company Limited's sourcing strategy exhibits a high degree of concentration, with 100% of its imports originating from India. This focused approach indicates a strategic choice to leverage India's robust pharmaceutical manufacturing capabilities. The company's top five imported products—Amphotericin, Glimepiride, and others—account for the entirety of its import portfolio, highlighting a specialized product range. The consistent importation of these products over 41 shipments suggests a stable and ongoing relationship with Indian suppliers.
2Supply Chain Resilience
Maxxcare's supply chain resilience is closely tied to its reliance on Indian suppliers. The company's imports are sourced from five unique Indian suppliers, including Inventia Healthcare Limited, Bharat Serums and Vaccines Limited, and others. This diversified supplier base mitigates risks associated with single-source dependency. However, the absence of publicly available information regarding backup suppliers and specific shipping routes indicates potential areas for improvement in enhancing supply chain robustness.
3Strategic Implications
Maxxcare Company's concentrated sourcing from India positions it to benefit from India's competitive pricing and established pharmaceutical manufacturing standards. For Indian exporters, this presents an opportunity to strengthen partnerships with Maxxcare by offering a diverse range of high-quality pharmaceutical formulations that align with the company's import portfolio. Expanding product offerings could further solidify these partnerships and enhance market presence in Vietnam.
Importing Pharmaceuticals into Vietnam — Regulatory Framework
Regulatory authority, GMP requirements, import licensing for Vietnam
1Regulatory Authority & Framework
In Vietnam, the Ministry of Health (MOH) serves as the primary regulatory authority overseeing pharmaceutical imports. The Law on Pharmacy 2016, along with Decree No. 54/2017/ND-CP, outlines the legal framework governing the importation of pharmaceutical products. These regulations stipulate that only entities with an import license issued by the MOH are authorized to import pharmaceuticals into Vietnam.
2Import Licensing & GMP
To import pharmaceuticals into Vietnam, companies must obtain an import license from the MOH, demonstrating compliance with local business and operational standards. Additionally, importers are required to adhere to Good Storage Practices (GSP) to ensure proper handling and storage conditions of pharmaceutical products. (smartlinklogistics.com.vn)
3Quality & Labeling
Imported pharmaceutical products must meet Vietnam's quality control and safety standards, which are progressively aligning with international norms. The MOH mandates that all drugs be registered and approved by the Drug Administration of Vietnam (DAV) and granted Market Authorization (MA) before distribution. This process involves submitting comprehensive documentation, including product composition, manufacturing processes, and stability data.
4Recent Regulatory Changes
On December 11, 2023, the Vietnamese government issued Decree No. 88/2023/ND-CP, amending and supplementing provisions of Decree No. 54/2017/ND-CP and Decree No. 155/2018/ND-CP. This decree introduces new regulations concerning the export and import of pharmaceuticals, raw materials for pharmaceutical production, and drug pricing management. Notably, it clarifies import cases of pharmaceutical raw materials and outlines conditions for obtaining import licenses.
Maxxcare Company Limited — Procurement Pattern Analysis
Product strategy, sourcing profile, and market positioning
1Product Strategy
Maxxcare Company Limited's import portfolio is concentrated in two therapeutic categories: Antifungals (53.3%) and Diabetes & Endocrine (46.7%). The top two imported products, Amphotericin and Glimepiride, align with these categories, indicating a strategic focus on addressing prevalent health concerns in Vietnam. This targeted approach suggests that Maxxcare is responding to significant market demands for treatments in these therapeutic areas.
2Sourcing Profile
Maxxcare's sourcing strategy is centered on importing generic pharmaceutical formulations from India, a country renowned for its extensive pharmaceutical manufacturing capabilities. The company's preference for finished formulations, such as tablets and injections, indicates a focus on ready-to-market products that can be swiftly distributed within Vietnam. This approach allows Maxxcare to efficiently meet domestic demand without the complexities associated with raw material imports.
3Market Positioning
Based on its product mix, Maxxcare Company Limited primarily serves the wholesale distribution segment of the Vietnamese pharmaceutical market. By importing and distributing finished pharmaceutical formulations, the company ensures that a diverse range of medications is available to various stakeholders, including retail pharmacies, hospitals, and clinics across Vietnam.
Seller's Guide — How to Become a Supplier to Maxxcare Company Limited
Opportunity assessment, qualifications, and practical approach strategy
1Opportunity Assessment
There is a realistic opportunity for new Indian suppliers to collaborate with Maxxcare Company Limited. The company's concentrated sourcing from India and its focus on specific therapeutic categories suggest that introducing additional high-quality generic formulations in these areas could meet existing market demands. By aligning with Maxxcare's import portfolio, new suppliers can establish a foothold in the Vietnamese market.
2Requirements & Qualifications
Indian exporters seeking to supply Maxxcare Company Limited must ensure that their pharmaceutical products are registered with the Drug Administration of Vietnam (DAV) and have obtained Market Authorization (MA). Compliance with Good Manufacturing Practice (GMP) standards, recognized by the World Health Organization (WHO) or the Pharmaceutical Inspection Co-operation Scheme (PIC/S), is essential. Additionally, adherence to Vietnam's labeling requirements, including Vietnamese language labeling and compliance with Circular 01, is mandatory.
3How to Approach
To establish a partnership with Maxxcare Company Limited, Indian exporters should initiate contact by providing detailed product information, including compliance with Vietnamese regulatory standards. Participating in relevant pharmaceutical trade fairs and industry events in Vietnam can facilitate networking opportunities. Understanding and adhering to Vietnam's import licensing requirements and GMP standards will be crucial in building a successful business relationship.
Frequently Asked Questions — Maxxcare Company Limited
What products does Maxxcare Company Limited import from India?
Maxxcare Company Limited imports 2 pharmaceutical products across 2 categories. Top imports: Amphotericin ($1.0M), Glimepiride ($876.0K).
Who supplies pharmaceuticals to Maxxcare Company Limited from India?
Maxxcare Company Limited sources from 5 verified Indian suppliers. The primary supplier is Inventia Healthcare Limited (63.3% of imports, $3.5M).
What is Maxxcare Company Limited's total pharmaceutical import value?
Maxxcare Company Limited's total pharmaceutical import value from India is $1.9M, based on 41 verified shipments in Indian Customs (DGFT) data.
What therapeutic categories does Maxxcare Company Limited focus on?
Maxxcare Company Limited imports across 2 categories. The largest: Antifungals (53.3%), Diabetes & Endocrine (46.7%).
Get Full Maxxcare Company Limited Import Intelligence
Access shipment-level details, supplier connections, pricing data, and competitive analysis. TransData Nexus provides verified Indian Customs (DGFT) data trusted by pharmaceutical trade professionals worldwide.
Official References & Regulatory Resources
Verify import regulations and drug registration requirements with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Buyer Matching: Maxxcare Company Limited identified across shipments using consignee name normalization, aggregating 2 name variants.
- 2.Statistical Normalization: Shipment values are statistically normalized to ensure accurate market share representation.
- 3.Market Share: Calculated per product as Maxxcare Company Limited's capped value divided by total Indian exports for that product.
- 4.Shipment Count: Based on 41 individual customs records matching Maxxcare Company Limited.
- 5.Supplier Verification: Maxxcare Company Limited sources from 5 verified Indian suppliers across 120 formulations, confirmed from customs records.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
2 Products Tracked
2 therapeutic categories
Expert-Reviewed
By pharmaceutical trade specialists
Data Source & Methodology
Trade data sourced from Indian Customs (DGFT) export shipment records. Values represent FOB export value in USD. Profile aggregates 2 company name variants from customs records. For current shipment-level data, contact TransData Nexus.