M/skottakal Ayurvedic Medicals Llc
Pharmaceutical Importer · United Arab Emirates · Ayurvedic & Herbal Products Focus · $189.6K Total Trade · DGFT Verified
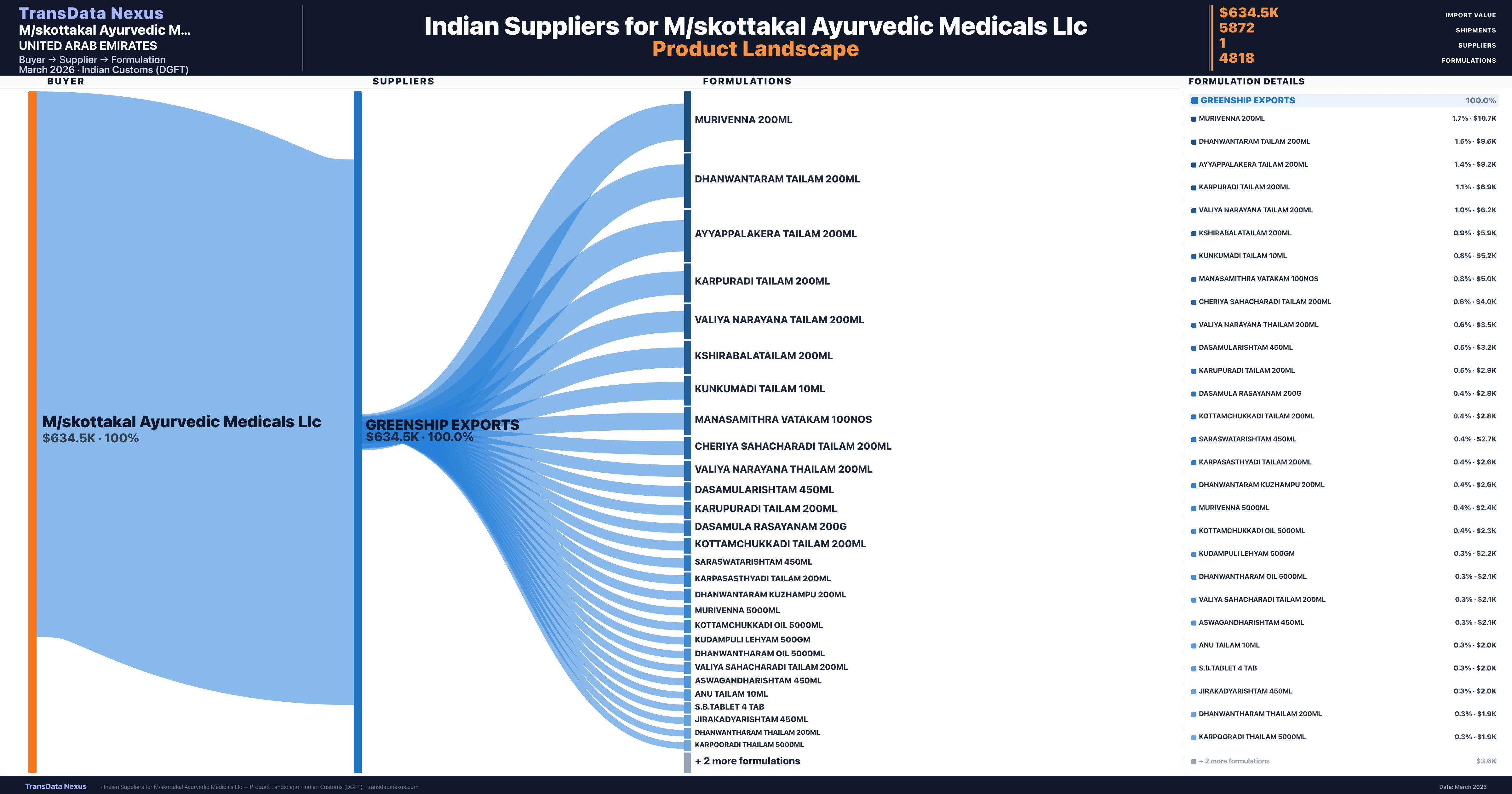
M/skottakal Ayurvedic Medicals Llc is a pharmaceutical importer based in United Arab Emirates with a total trade value of $189.6K across 5 products in 1 therapeutic categories. Based on 1,005 verified import shipments from Indian Customs (DGFT) records, the company actively imports across multiple product segments. M/skottakal Ayurvedic Medicals Llc sources from 1 verified Indian supplier, with Greenship Exports accounting for 100.0% of imports.
M/skottakal Ayurvedic Medicals Llc — Import Portfolio & Supplier Network
Who Are the Verified Indian Suppliers to M/skottakal Ayurvedic Medicals Llc?
Customs-verified supplier relationships from Indian DGFT records
| Supplier | Value | Shipments | Share |
|---|---|---|---|
| Greenship Exports | $634.5K | 5,872 | 100.0% |
M/skottakal Ayurvedic Medicals Llc sources from 1 verified Indian supplier across 4,818 distinct formulations. The sourcing is highly concentrated — Greenship Exports accounts for 100.0% of total imports, indicating a strategic single-source relationship.
What Formulations Does M/skottakal Ayurvedic Medicals Llc Import?
| Formulation | Value | Ships |
|---|---|---|
| Murivenna 200ML | $10.7K | 6 |
| Dhanwantaram tailam 200ML | $9.6K | 3 |
| Ayyappalakera tailam 200ML | $9.2K | 3 |
| Karpuradi tailam 200ML | $6.9K | 3 |
| Valiya narayana tailam 200ML | $6.2K | 2 |
| Kshirabalatailam 200ML | $5.9K | 3 |
| Kunkumadi tailam 10ML | $5.2K | 4 |
| Manasamithra vatakam | $5.0K | 2 |
| Cheriya sahacharadi tailam 200ML | $4.0K | 4 |
| Valiya narayana thailam 200ML | $3.5K | 2 |
| Dasamularishtam 450ML | $3.2K | 4 |
| Karupuradi tailam 200ML | $2.9K | 1 |
| Dasamula rasayanam | $2.8K | 3 |
| Kottamchukkadi tailam 200ML | $2.8K | 2 |
| Saraswatarishtam 450ML | $2.7K | 3 |
M/skottakal Ayurvedic Medicals Llc imports 4,818 distinct pharmaceutical formulations. Showing top 15 by value. For full formulation-level data, contact TransData Nexus.
What Products Does M/skottakal Ayurvedic Medicals Llc Import?
Top Products by Import Value
M/skottakal Ayurvedic Medicals Llc Therapeutic Categories — 1 Specializations
M/skottakal Ayurvedic Medicals Llc imports across 1 therapeutic categories, with Ayurvedic & Herbal Products (100.0%), representing the largest segments. The portfolio is concentrated — top 5 products = 100% of total imports.
Ayurvedic & Herbal Products
5 products · 100.0% · $189.6K
Import Portfolio — Top 5 by Import Value
| # | Product | Category | Value | Ships | Share | Rk |
|---|---|---|---|---|---|---|
| 1 | Taila | Ayurvedic & Herbal Products | $136.6K | 583 | 1.8% | 8 |
| 2 | Arishta | Ayurvedic & Herbal Products | $24.5K | 134 | 1.7% | 9 |
| 3 | Kwatha | Ayurvedic & Herbal Products | $15.9K | 112 | 3.0% | 10 |
| 4 | Ghrita | Ayurvedic & Herbal Products | $8.4K | 117 | 1.4% | 7 |
| 5 | Asava | Ayurvedic & Herbal Products | $4.2K | 59 | 0.9% | 11 |
M/skottakal Ayurvedic Medicals Llc imports 5 pharmaceutical products across 1 categories into United Arab Emirates totaling $189.6K.
Key Metrics
Top Categories
Indian Suppliers
Related Trade Data
Need Detailed Data?
Shipment-level records, supplier connections & pricing for M/skottakal Ayurvedic Medicals Llc.
Request DemoM/skottakal Ayurvedic Medicals Llc — Corporate Profile & Information
Company type, headquarters, distribution network, and industry role
1Company Overview
M/skottakal Ayurvedic Medicals LLC is a pharmaceutical importer and buyer based in the United Arab Emirates (UAE), specializing in the importation of Ayurvedic and herbal medical products. The company is headquartered in Deira, Dubai, with a registered address at P.O. Box 116859, Dubai, UAE. Contact details include a telephone number: +971 4 2729628.
As a key player in the UAE's pharmaceutical distribution network, M/skottakal Ayurvedic Medicals LLC focuses on sourcing finished pharmaceutical formulations from India. Their product portfolio is entirely concentrated on Ayurvedic and herbal products, reflecting a strategic emphasis on traditional medicine. The company's operations are characterized by a high degree of supplier concentration, with a single verified Indian supplier, GREENSHIP EXPORTS, accounting for all their imports.
2Distribution Network
Specific details regarding M/skottakal Ayurvedic Medicals LLC's warehouse locations and logistics capabilities are not publicly disclosed. However, given their substantial import volume and focus on Ayurvedic products, it is likely that the company maintains distribution centers within the UAE to facilitate efficient delivery to local markets. The company's geographic coverage appears to be primarily within the UAE, serving the domestic demand for Ayurvedic and herbal medical products.
3Industry Role
M/skottakal Ayurvedic Medicals LLC operates as a pharmaceutical importer and distributor within the UAE's supply chain. By sourcing finished pharmaceutical formulations from India, they play a crucial role in introducing Ayurvedic and herbal products to the UAE market. Their focus on finished products indicates a strategic position in the distribution network, catering to the growing demand for traditional medicine in the region.
Supplier Relationship Intelligence — M/skottakal Ayurvedic Medicals Llc
Sourcing concentration, supply chain resilience, and strategic implications
1Sourcing Concentration Analysis
The company's sourcing strategy exhibits a high degree of concentration, relying exclusively on a single supplier, GREENSHIP EXPORTS, for all their imports. This single-source dependency could pose risks related to supply chain disruptions, such as potential delays or quality control issues. However, the consistent volume of shipments—1,005 in total—suggests a stable and reliable relationship between M/skottakal Ayurvedic Medicals LLC and GREENSHIP EXPORTS. The absence of alternative suppliers may limit the company's flexibility in responding to market changes or negotiating better terms.
2Supply Chain Resilience
The resilience of M/skottakal Ayurvedic Medicals LLC's supply chain is closely tied to the stability of their relationship with GREENSHIP EXPORTS. The company's exclusive reliance on this single supplier for all imports indicates a deep integration and trust. However, the lack of backup suppliers introduces vulnerability to potential disruptions. The diversity of formulations imported—4,818 unique products—demonstrates a broad product range, which may help mitigate risks associated with supply chain disruptions. Ensuring that GREENSHIP EXPORTS adheres to international quality standards and maintains consistent delivery schedules is crucial for the company's operational stability.
3Strategic Implications
The concentrated sourcing pattern of M/skottakal Ayurvedic Medicals LLC positions them as a significant partner for GREENSHIP EXPORTS, potentially leading to favorable terms and prioritized service. For Indian exporters seeking to become alternative suppliers, this presents an opportunity to diversify the company's supply base. Offering competitive pricing, reliable delivery schedules, and compliance with international quality standards could attract M/skottakal Ayurvedic Medicals LLC as a new client. Additionally, understanding the company's specific product preferences and regulatory requirements will be essential for successful engagement.
Importing Pharmaceuticals into United Arab Emirates — Regulatory Framework
Regulatory authority, GMP requirements, import licensing for United Arab Emirates
1Regulatory Authority & Framework
In the UAE, the Ministry of Health and Prevention (MoH) is the primary regulatory authority overseeing the pharmaceutical sector. The MoH is responsible for the registration of pharmaceutical products, issuance of import licenses, and ensuring compliance with health and safety standards. Federal Law No. 4/1983 on the Pharmaceutical Professions and Establishments and the Import, Manufacture, and Distribution of Pharmaceutical Products mandates that all medicines imported into the UAE must be registered with the MoH.
The registration process involves submitting a new drug application to the MoH, which includes providing comprehensive product information, clinical data, and evidence of compliance with international quality standards. This process ensures that only safe and effective pharmaceutical products are available in the UAE market. Additionally, individual Emirates have established their local health regulators to oversee the healthcare and pharmaceutical sector of their specific jurisdiction, such as the Dubai Health Authority in Dubai and the Health Authority - Abu Dhabi.
2Import Licensing & GMP
To import pharmaceutical products into the UAE, companies must obtain a Pharmaceutical Import License from the MoH. This license is granted upon fulfilling prescribed conditions, including compliance with Good Manufacturing Practice (GMP) standards. The MoH recognizes GMP certificates from reputable international bodies, such as the World Health Organization (WHO) and the Pharmaceutical Inspection Co-operation Scheme (PIC/S). Importers must ensure that their suppliers hold valid GMP certifications to meet regulatory requirements.
In addition to the import license, companies must also obtain wholesale distribution authorization from the MoH. This authorization permits the distribution of pharmaceutical products within the UAE and ensures that distributors adhere to the regulatory standards set forth by the MoH. Maintaining valid licenses and certifications is essential for legal compliance and uninterrupted operations in the UAE pharmaceutical market.
3Quality & Labeling
Imported pharmaceutical products must undergo batch testing to ensure they meet the safety, efficacy, and quality standards set by the MoH. Stability studies are also required to confirm that products maintain their quality throughout their shelf life. Labeling requirements include providing information in Arabic, the official language of the UAE, and ensuring that labels contain all necessary details, such as product name, dosage form, strength, manufacturing date, expiry date, storage conditions, and instructions for use. Serialization mandates may also apply to facilitate traceability and prevent counterfeit products from entering the market.
4Recent Regulatory Changes
As of March 2026, there have been no publicly disclosed significant policy changes affecting the import of Indian pharmaceutical products into the UAE between 2024 and 2026. The regulatory framework remains consistent, with the MoH continuing to enforce existing laws and regulations governing the importation and distribution of pharmaceutical products. Importers and distributors should stay informed about any potential updates to ensure ongoing compliance with UAE regulations.
M/skottakal Ayurvedic Medicals Llc — Procurement Pattern Analysis
Product strategy, sourcing profile, and market positioning
1Product Strategy
M/skottakal Ayurvedic Medicals LLC's exclusive focus on Ayurvedic and herbal products aligns with the growing consumer interest in traditional and natural medicine within the UAE. This strategic emphasis caters to a niche market segment seeking alternative therapeutic options. The demand for such products is driven by cultural preferences, increasing awareness of holistic health approaches, and a desire for natural remedies. By concentrating on this therapeutic area, the company positions itself as a specialist in Ayurvedic and herbal medicine, differentiating itself from competitors in the broader pharmaceutical market.
2Sourcing Profile
M/skottakal Ayurvedic Medicals LLC's sourcing strategy is centered on importing finished pharmaceutical formulations from India, specifically Ayurvedic and herbal products. This approach allows the company to offer a diverse range of products without the complexities associated with manufacturing. India's well-established reputation in Ayurvedic medicine and its compliance with international quality standards make it an ideal sourcing partner. The company's reliance on a single supplier, GREENSHIP EXPORTS, indicates a preference for streamlined operations and consistent product quality.
3Market Positioning
By focusing exclusively on Ayurvedic and herbal products, M/skottakal Ayurvedic Medicals LLC serves a specialized segment of the UAE market. Their product mix suggests a positioning strategy aimed at consumers seeking traditional and natural medicinal options. This market segment includes retail pharmacies, health food stores, and consumers interested in alternative health solutions. The company's emphasis on quality and authenticity in Ayurvedic products further strengthens its position in this niche market.
Seller's Guide — How to Become a Supplier to M/skottakal Ayurvedic Medicals Llc
Opportunity assessment, qualifications, and practical approach strategy
1Opportunity Assessment
There is a realistic opportunity for new Indian suppliers to engage with M/skottakal Ayurvedic Medicals LLC, especially if they can offer unique Ayurvedic formulations or competitive pricing. The company's current sourcing pattern indicates a potential openness to diversifying its supplier base, provided that new suppliers can meet the required quality standards and regulatory compliance. Establishing a relationship with M/skottakal Ayurvedic Medicals LLC would require demonstrating product quality, reliability, and an understanding of the UAE's regulatory environment.
2Requirements & Qualifications
Indian exporters seeking to supply M/skottakal Ayurvedic Medicals LLC must ensure that their products are registered with the UAE's Ministry of Health and Prevention. This includes obtaining the necessary import licenses and adhering to GMP standards recognized by the MoH. Products must also comply with labeling requirements, including Arabic language labeling and adherence to stability and batch testing protocols. Understanding and complying with the UAE's regulatory framework is essential for successful market entry.
3How to Approach
To establish a relationship with M/skott
Frequently Asked Questions — M/skottakal Ayurvedic Medicals Llc
What products does M/skottakal Ayurvedic Medicals Llc import from India?
M/skottakal Ayurvedic Medicals Llc imports 5 pharmaceutical products across 1 categories. Top imports: Taila ($136.6K), Arishta ($24.5K), Kwatha ($15.9K), Ghrita ($8.4K), Asava ($4.2K).
Who supplies pharmaceuticals to M/skottakal Ayurvedic Medicals Llc from India?
M/skottakal Ayurvedic Medicals Llc sources from 1 verified Indian suppliers. The primary supplier is Greenship Exports (100.0% of imports, $634.5K).
What is M/skottakal Ayurvedic Medicals Llc's total pharmaceutical import value?
M/skottakal Ayurvedic Medicals Llc's total pharmaceutical import value from India is $189.6K, based on 1,005 verified shipments in Indian Customs (DGFT) data.
What therapeutic categories does M/skottakal Ayurvedic Medicals Llc focus on?
M/skottakal Ayurvedic Medicals Llc imports across 1 categories. The largest: Ayurvedic & Herbal Products (100.0%).
Get Full M/skottakal Ayurvedic Medicals Llc Import Intelligence
Access shipment-level details, supplier connections, pricing data, and competitive analysis. TransData Nexus provides verified Indian Customs (DGFT) data trusted by pharmaceutical trade professionals worldwide.
Official References & Regulatory Resources
Verify import regulations and drug registration requirements with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Buyer Matching: M/skottakal Ayurvedic Medicals Llc identified across shipments using consignee name normalization, aggregating 2 name variants.
- 2.Statistical Normalization: Shipment values are statistically normalized to ensure accurate market share representation.
- 3.Market Share: Calculated per product as M/skottakal Ayurvedic Medicals Llc's capped value divided by total Indian exports for that product.
- 4.Shipment Count: Based on 1,005 individual customs records matching M/skottakal Ayurvedic Medicals Llc.
- 5.Supplier Verification: M/skottakal Ayurvedic Medicals Llc sources from 1 verified Indian suppliers across 4,818 formulations, confirmed from customs records.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
5 Products Tracked
1 therapeutic categories
Expert-Reviewed
By pharmaceutical trade specialists
Data Source & Methodology
Trade data sourced from Indian Customs (DGFT) export shipment records. Values represent FOB export value in USD. Profile aggregates 2 company name variants from customs records. For current shipment-level data, contact TransData Nexus.