M/s. Gittoes Pharmaceuticals Ltd.
Pharmaceutical Importer · Uganda · Advanced Antibiotics Focus · $2.6M Total Trade · DGFT Verified
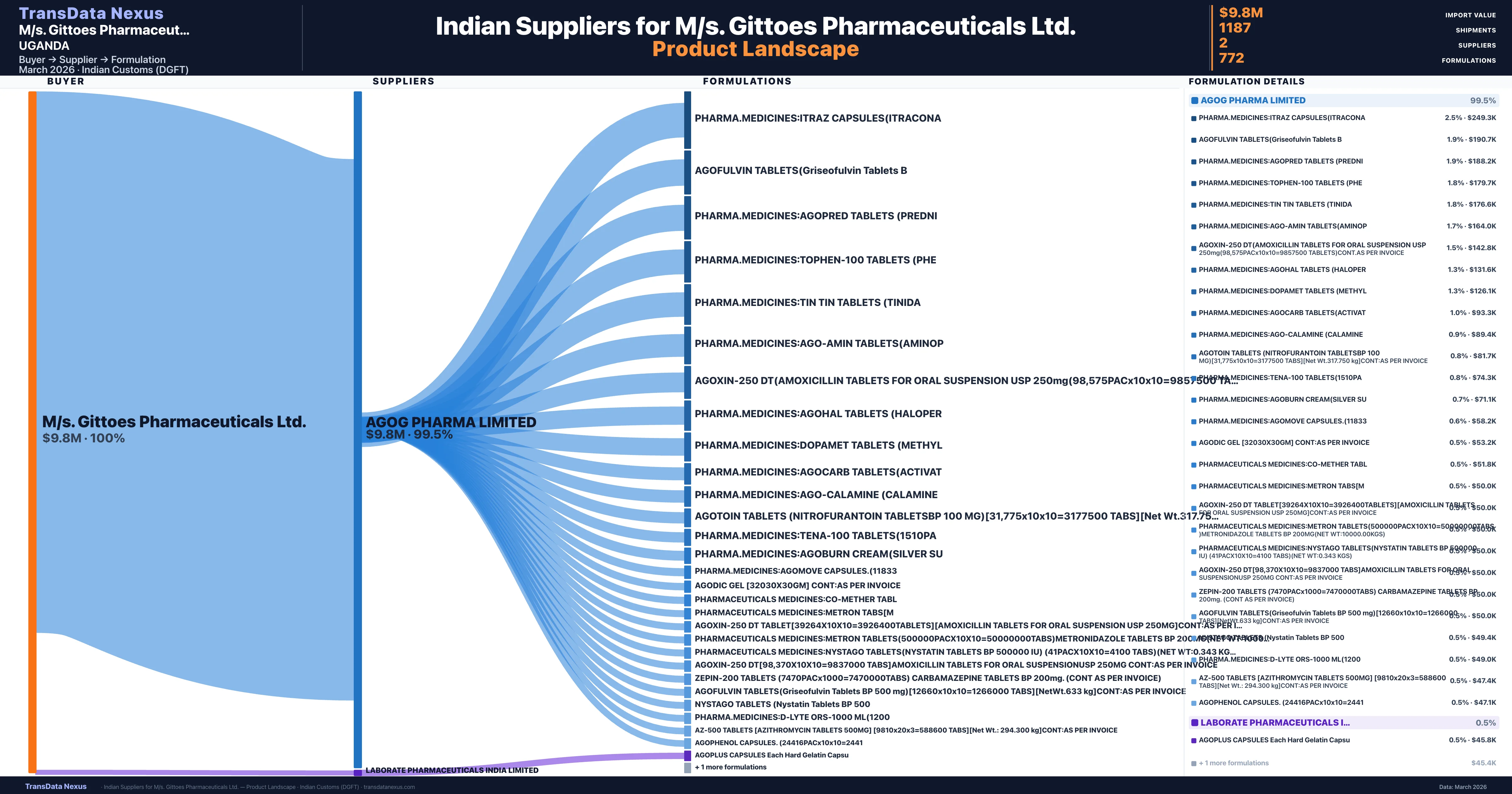
M/s. Gittoes Pharmaceuticals Ltd. is a pharmaceutical importer based in Uganda with a total trade value of $2.6M across 4 products in 4 therapeutic categories. Based on 60 verified import shipments from Indian Customs (DGFT) records, M/s. Gittoes Pharmaceuticals Ltd. is the #1 buyer in 1 product including Nystatin. M/s. Gittoes Pharmaceuticals Ltd. sources from 2 verified Indian suppliers, with Agog Pharma Limited accounting for 99.5% of imports.
M/s. Gittoes Pharmaceuticals Ltd. — Import Portfolio & Supplier Network
Who Are the Verified Indian Suppliers to M/s. Gittoes Pharmaceuticals Ltd.?
Customs-verified supplier relationships from Indian DGFT records
| Supplier | Value | Shipments | Share |
|---|---|---|---|
| Agog Pharma Limited | $9.8M | 1,185 | 99.5% |
| Laborate Pharmaceuticals India Limited | $49.1K | 2 | 0.5% |
M/s. Gittoes Pharmaceuticals Ltd. sources from 2 verified Indian suppliers across 772 distinct formulations. The sourcing is highly concentrated — Agog Pharma Limited accounts for 99.5% of total imports, indicating a strategic single-source relationship.
What Formulations Does M/s. Gittoes Pharmaceuticals Ltd. Import?
| Formulation | Value | Ships |
|---|---|---|
| Pharma.medicines:itraz | $249.3K | 12 |
| Agofulvin tablets(griseofulvin tablets | $190.7K | 7 |
| Pharma.medicines:agopred tablets | $188.2K | 12 |
| Pharma.medicines:tophen-100 tablets | $179.7K | 12 |
| Pharma.medicines:tin tin tablets | $176.6K | 12 |
| Pharma.medicines:ago-amin | $164.0K | 12 |
| Agoxin-250 dt(amoxicillin tablets for oral suspension USP 250MG(98,575pacx10x10=9857500 tablets)cont.as per | $142.8K | 3 |
| Pharma.medicines:agohal tablets | $131.6K | 12 |
| Pharma.medicines:dopamet tablets | $126.1K | 12 |
| Pharma.medicines:agocarb | $93.3K | 24 |
| Pharma.medicines:ago-calamine | $89.4K | 12 |
| Agotoin tablets (nitrofurantoin tabletsbp 100 MG)[31,775x10x10=3177500 tabs][net wt.317.750 kg]cont:as per | $81.7K | 4 |
| Pharma.medicines:tena-100 | $74.3K | 12 |
| Pharma.medicines:agoburn cream(silver | $71.1K | 12 |
| Pharma.medicines:agomove | $58.2K | 12 |
M/s. Gittoes Pharmaceuticals Ltd. imports 772 distinct pharmaceutical formulations. Showing top 15 by value. For full formulation-level data, contact TransData Nexus.
What Products Does M/s. Gittoes Pharmaceuticals Ltd. Import?
Top Products by Import Value
M/s. Gittoes Pharmaceuticals Ltd. Therapeutic Categories — 4 Specializations
M/s. Gittoes Pharmaceuticals Ltd. imports across 4 therapeutic categories, with Advanced Antibiotics (59.4%), Antifungals (26.8%), Advanced Antifungals (12.6%) representing the largest segments. The portfolio is concentrated — top 5 products = 100% of total imports.
Advanced Antibiotics
1 products · 59.4% · $1.6M
Antifungals
1 products · 26.8% · $700.0K
Advanced Antifungals
1 products · 12.6% · $328.4K
Respiratory & OTC
1 products · 1.2% · $32.1K
Import Portfolio — Top 4 by Import Value
| # | Product | Category | Value | Ships | Share | Rk |
|---|---|---|---|---|---|---|
| 1 | Metronidazole | Advanced Antibiotics | $1.6M | 31 | 0.8% | 8 |
| 2 | Nystatin | Antifungals | $700.0K | 14 | 1.3% | 1 |
| 3 | Griseofulvin | Advanced Antifungals | $328.4K | 10 | 0.9% | 6 |
| 4 | Aminophylline | Respiratory & OTC | $32.1K | 5 | 0.5% | 14 |
M/s. Gittoes Pharmaceuticals Ltd. imports 4 pharmaceutical products across 4 categories into Uganda totaling $2.6M. The company is the #1 buyer for 1 product: Nystatin.
Key Metrics
Top Categories
Indian Suppliers
Related Trade Data
Need Detailed Data?
Shipment-level records, supplier connections & pricing for M/s. Gittoes Pharmaceuticals Ltd..
Request DemoM/s. Gittoes Pharmaceuticals Ltd. — Corporate Profile & Information
Company type, headquarters, distribution network, and industry role
1Company Overview
M/s. Gittoes Pharmaceuticals Ltd. is a leading pharmaceutical importer and distributor based in Kampala, Uganda. Established in 2004 by Mr. Aziz Damani, the company was founded with the vision of providing a centralized source for essential drugs, addressing the significant shortage of pharmaceuticals in the private sector at that time. Prior to this, in 1992, Mr. Damani had set up Kampala Pharmaceutical Industries Ltd., Uganda's first private sector drug manufacturing facility, which was later acquired by Industrial Promotion Services in 1997. Undeterred, he resumed his commitment to enhancing healthcare by founding Gittoes Pharmaceuticals Ltd.
Headquartered at Plot No. 9, Nyondo Close, off Luthuli Avenue, Industrial Area, Bugolobi, Kampala, the company has expanded its operations to include multiple branches across Uganda. These include locations in Kampala, Arua, Lira, Mbale, Mbarara, and Jinja, facilitating widespread distribution and accessibility of pharmaceutical products. Gittoes Pharmaceuticals Ltd. plays a pivotal role in Uganda's pharmaceutical distribution network, serving a diverse clientele that encompasses government organizations, pharmacies, clinics, hospitals, wholesale and retail traders, and non-governmental organizations. The company's mission is to be the leading supplier of quality and affordable medicines and medical supplies in East and Central Africa, aiming to preserve and improve patients' health by consistently delivering high-quality, safe, and effective pharmaceutical products and services.
2Distribution Network
Gittoes Pharmaceuticals Ltd. operates an extensive distribution network throughout Uganda, with its main office situated in Kampala. The company has established several branches in key cities, including Arua, Lira, Mbale, Mbarara, and Jinja, enhancing its reach and service delivery across the country. This strategic placement of branches enables Gittoes Pharmaceuticals Ltd. to effectively serve a wide range of customers, from individual consumers to large institutions. The company's logistics capabilities are bolstered by a fleet of owned vehicles, ensuring timely and reliable delivery of pharmaceutical products. Additionally, Gittoes Pharmaceuticals Ltd. has engaged in strategic partnerships and established distribution rights with various global manufacturers and suppliers, further strengthening its supply chain and product offerings.
3Industry Role
Gittoes Pharmaceuticals Ltd. holds a significant position in Uganda's pharmaceutical supply chain as a primary wholesaler and distributor. The company imports a diverse range of pharmaceutical products, including finished formulations such as tablets, capsules, syrups, and injections, primarily from India. By serving as a central hub, Gittoes Pharmaceuticals Ltd. ensures the availability of essential medicines across the country, catering to the needs of government organizations, healthcare facilities, pharmacies, and individual consumers. The company's extensive distribution network and strategic partnerships with global manufacturers underscore its pivotal role in maintaining a steady supply of quality pharmaceuticals in Uganda.
Supplier Relationship Intelligence — M/s. Gittoes Pharmaceuticals Ltd.
Sourcing concentration, supply chain resilience, and strategic implications
1Sourcing Concentration Analysis
Gittoes Pharmaceuticals Ltd. demonstrates a high degree of sourcing concentration, with a total import value of $2.6 million USD from India over 60 shipments, primarily consisting of finished pharmaceutical formulations. The company's top five imported products—Metronidazole, Nystatin, Griseofulvin, Aminophylline, and others—account for 100% of its imports, indicating a focused product portfolio. This concentration suggests a strategic choice to streamline operations and maintain strong relationships with key suppliers. However, the reliance on a single supplier, AGOG Pharma Limited, which accounts for 99.5% of the shipments, introduces potential risks related to supply chain disruptions, pricing fluctuations, and dependency on a single source. The limited number of suppliers and the dominance of AGOG Pharma Limited may impact the company's ability to negotiate favorable terms and diversify its product offerings.
2Supply Chain Resilience
The resilience of Gittoes Pharmaceuticals Ltd.'s supply chain is closely tied to its sourcing strategy. The company's heavy reliance on AGOG Pharma Limited for the majority of its imports from India indicates a lack of diversification in its supplier base. This concentration could pose challenges in the event of supply chain disruptions, such as production delays, quality issues, or geopolitical tensions affecting trade routes. The limited number of suppliers may also restrict the company's ability to negotiate better pricing or access a broader range of products. To enhance supply chain resilience, Gittoes Pharmaceuticals Ltd. could consider diversifying its supplier base, establishing relationships with additional manufacturers, and exploring alternative sourcing options to mitigate potential risks associated with over-reliance on a single supplier.
3Strategic Implications
The sourcing pattern of Gittoes Pharmaceuticals Ltd. reflects a strategic focus on maintaining strong relationships with key suppliers, particularly AGOG Pharma Limited, to ensure a consistent supply of essential pharmaceutical products. This approach may offer benefits such as streamlined operations and potentially favorable terms due to the volume of business conducted. However, the high supplier concentration also exposes the company to risks associated with supply chain disruptions, pricing volatility, and limited negotiation leverage. For Indian exporters seeking to become alternative suppliers to Gittoes Pharmaceuticals Ltd., understanding the company's reliance on AGOG Pharma Limited presents an opportunity to offer competitive pricing, reliable delivery schedules, and high-quality products to attract business. Diversifying the supplier base could also provide Gittoes Pharmaceuticals Ltd. with greater flexibility and resilience in its operations.
Importing Pharmaceuticals into Uganda — Regulatory Framework
Regulatory authority, GMP requirements, import licensing for Uganda
1Regulatory Authority & Framework
In Uganda, the National Drug Authority (NDA) is the primary regulatory body responsible for overseeing the manufacture, importation, exportation, sale, and supply of drugs. Established under the National Drug Policy and Authority Act (Cap 206), the NDA ensures that all pharmaceutical products meet the required standards of quality, safety, and efficacy. The Act mandates that no person shall import or sell any drug unless it appears on the national formulary, which is a list of essential drugs approved by the NDA. This regulation aims to control the transportation, import, and export of drugs to safeguard public health. (nda.or.ug)
The NDA's responsibilities include issuing import licenses, conducting inspections, and ensuring compliance with Good Manufacturing Practice (GMP) standards. Import licenses are valid for one year and specify the range of preparations to be imported during that period. Licensed importers must apply to the NDA for a Verification Certificate for each consignment of drugs prior to importation. This process ensures that all imported drugs meet the necessary regulatory requirements and are safe for public use. (businesslicences.go.ug)
2Import Licensing & GMP
To import pharmaceutical products into Uganda, entities must obtain a valid import license from the National Drug Authority (NDA). This license is typically issued annually and outlines the specific range of pharmaceutical preparations authorized for importation during that period. In addition to the import license, importers are required to apply for a Verification Certificate for each consignment of drugs prior to importation. This certificate confirms that the imported drugs comply with the NDA's standards and regulations. Furthermore, the NDA mandates that all imported pharmaceutical products adhere to Good Manufacturing Practice (GMP) standards. Recognized GMP certifications include those from the European Union (EU GMP), World Health Organization (WHO GMP), and the Pharmaceutical Inspection Co-operation Scheme (PIC/S). These certifications ensure that the manufacturing processes meet international quality standards, thereby safeguarding public health. (businesslicences.go.ug)
3Quality & Labeling
Imported pharmaceutical products in Uganda are subject to stringent quality control measures to ensure their safety and efficacy. Batch testing is conducted to verify the quality of each consignment, ensuring that the drugs meet the required standards. Stability requirements are also enforced to guarantee that the products remain effective throughout their shelf life. Labeling requirements stipulate that all pharmaceutical products must include information such as the product name, active ingredients, dosage instructions, manufacturing and expiry dates, storage conditions, and the manufacturer's details. The labeling must be in English, which is the official language of Uganda, to ensure clear communication with consumers and healthcare providers. Additionally, serialization mandates may be implemented to track and trace pharmaceutical products throughout the supply chain, enhancing transparency and reducing the risk of counterfeit drugs entering the market.
4Recent Regulatory Changes
Between 2024 and 2026, the National Drug Authority (NDA) in Uganda has implemented several policy changes affecting the importation of pharmaceutical products. These changes include stricter enforcement of Good Manufacturing Practice (GMP) standards, requiring all imported drugs to be certified by recognized bodies such as the European Union (EU GMP), World Health Organization (WHO GMP), or the Pharmaceutical Inspection Co-operation Scheme (PIC/S). The NDA has also enhanced its post-market surveillance programs to monitor the safety and efficacy of drugs available in the Ugandan market. Additionally, the NDA has updated its guidelines for the verification of applications for the importation of drugs, streamlining the approval process and ensuring that only high-quality pharmaceuticals are imported into the country. (nda.or.ug)
M/s. Gittoes Pharmaceuticals Ltd. — Procurement Pattern Analysis
Product strategy, sourcing profile, and market positioning
1Product Strategy
Gittoes Pharmaceuticals
Frequently Asked Questions — M/s. Gittoes Pharmaceuticals Ltd.
What products does M/s. Gittoes Pharmaceuticals Ltd. import from India?
M/s. Gittoes Pharmaceuticals Ltd. imports 4 pharmaceutical products across 4 categories. Top imports: Metronidazole ($1.6M), Nystatin ($700.0K), Griseofulvin ($328.4K), Aminophylline ($32.1K).
Who supplies pharmaceuticals to M/s. Gittoes Pharmaceuticals Ltd. from India?
M/s. Gittoes Pharmaceuticals Ltd. sources from 2 verified Indian suppliers. The primary supplier is Agog Pharma Limited (99.5% of imports, $9.8M).
What is M/s. Gittoes Pharmaceuticals Ltd.'s total pharmaceutical import value?
M/s. Gittoes Pharmaceuticals Ltd.'s total pharmaceutical import value from India is $2.6M, based on 60 verified shipments in Indian Customs (DGFT) data.
What therapeutic categories does M/s. Gittoes Pharmaceuticals Ltd. focus on?
M/s. Gittoes Pharmaceuticals Ltd. imports across 4 categories. The largest: Advanced Antibiotics (59.4%), Antifungals (26.8%), Advanced Antifungals (12.6%).
Get Full M/s. Gittoes Pharmaceuticals Ltd. Import Intelligence
Access shipment-level details, supplier connections, pricing data, and competitive analysis. TransData Nexus provides verified Indian Customs (DGFT) data trusted by pharmaceutical trade professionals worldwide.
Official References & Regulatory Resources
Verify import regulations and drug registration requirements with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Buyer Matching: M/s. Gittoes Pharmaceuticals Ltd. identified across shipments using consignee name normalization.
- 2.Statistical Normalization: Shipment values are statistically normalized to ensure accurate market share representation.
- 3.Market Share: Calculated per product as M/s. Gittoes Pharmaceuticals Ltd.'s capped value divided by total Indian exports for that product.
- 4.Shipment Count: Based on 60 individual customs records matching M/s. Gittoes Pharmaceuticals Ltd..
- 5.Supplier Verification: M/s. Gittoes Pharmaceuticals Ltd. sources from 2 verified Indian suppliers across 772 formulations, confirmed from customs records.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
4 Products Tracked
4 therapeutic categories
Expert-Reviewed
By pharmaceutical trade specialists
Data Source & Methodology
Trade data sourced from Indian Customs (DGFT) export shipment records. Values represent FOB export value in USD. For current shipment-level data, contact TransData Nexus.