M/s. Delmaw Enterprises Ltd
Pharmaceutical Importer · Uganda · Antihistamines & Allergy Focus · $600.0K Total Trade · DGFT Verified
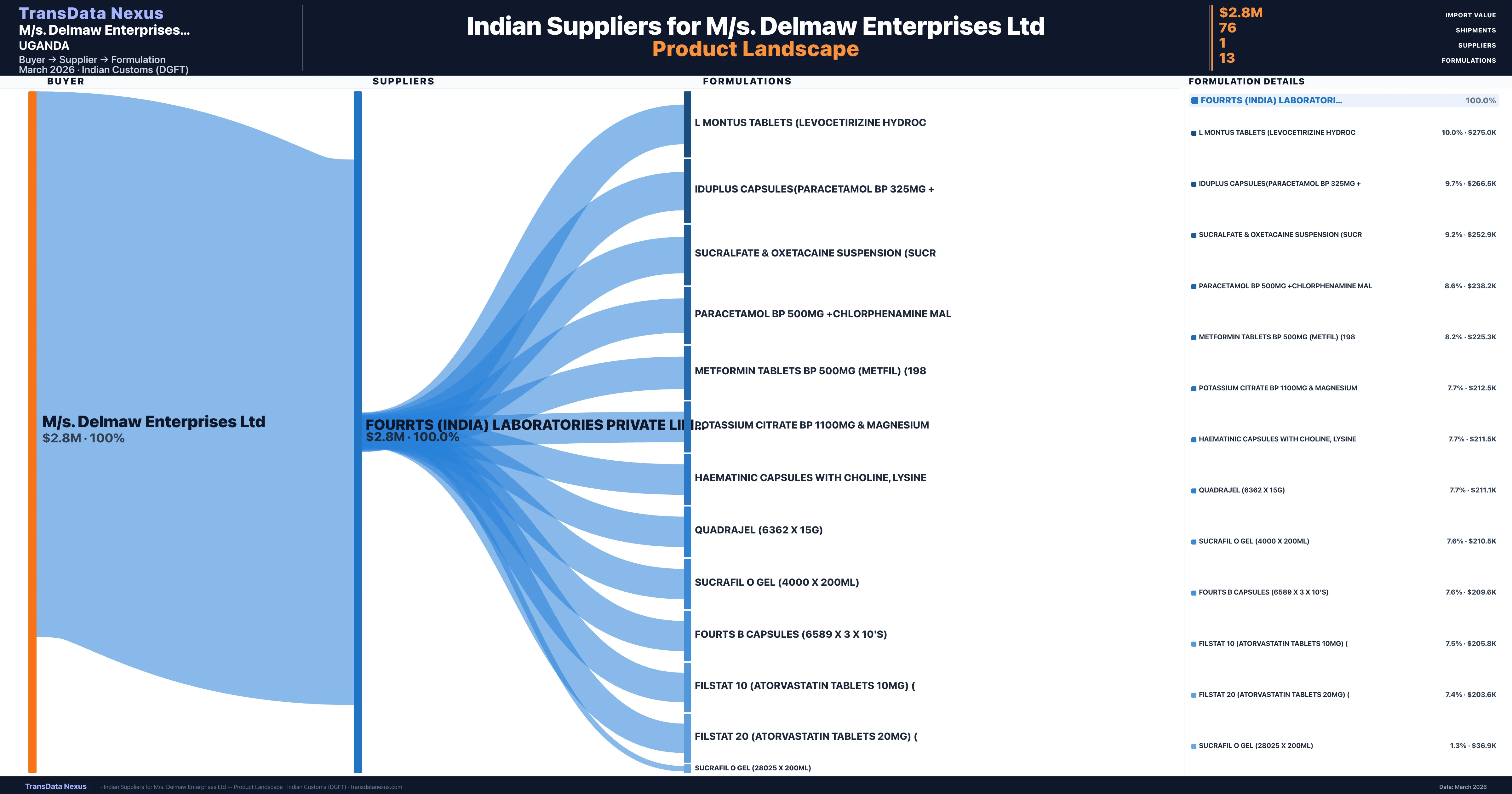
M/s. Delmaw Enterprises Ltd is a pharmaceutical importer based in Uganda with a total trade value of $600.0K across 2 products in 2 therapeutic categories. Based on 12 verified import shipments from Indian Customs (DGFT) records, M/s. Delmaw Enterprises Ltd is the #1 buyer in 1 product including Cetirizine. M/s. Delmaw Enterprises Ltd sources from 1 verified Indian supplier, with Fourrts (india) Laboratories Private Limited accounting for 100.0% of imports.
M/s. Delmaw Enterprises Ltd — Import Portfolio & Supplier Network
Who Are the Verified Indian Suppliers to M/s. Delmaw Enterprises Ltd?
Customs-verified supplier relationships from Indian DGFT records
| Supplier | Value | Shipments | Share |
|---|---|---|---|
| Fourrts (india) Laboratories Private Limited | $2.8M | 76 | 100.0% |
M/s. Delmaw Enterprises Ltd sources from 1 verified Indian supplier across 13 distinct formulations. The sourcing is highly concentrated — Fourrts (india) Laboratories Private Limited accounts for 100.0% of total imports, indicating a strategic single-source relationship.
What Formulations Does M/s. Delmaw Enterprises Ltd Import?
| Formulation | Value | Ships |
|---|---|---|
| L montus tablets (levocetirizine | $275.0K | 6 |
| Iduplus capsules(paracetamol BP 325MG | $266.5K | 7 |
| Sucralfate & oxetacaine suspension | $252.9K | 7 |
| Paracetamol BP 500MG +chlorphenamine | $238.2K | 7 |
| Metformin tablets BP 500MG (metfil) | $225.3K | 6 |
| Potassium citrate BP 1100MG & | $212.5K | 6 |
| Haematinic capsules with choline | $211.5K | 6 |
| Quadrajel (6362 x 15g) | $211.1K | 6 |
| Sucrafil o gel (4000 x 200ML) | $210.5K | 6 |
| Fourts b capsules (6589 x 3 x 10's) | $209.6K | 6 |
| Filstat 10 (atorvastatin tablets 10MG) | $205.8K | 6 |
| Filstat 20 (atorvastatin tablets 20MG) | $203.6K | 6 |
| Sucrafil o gel (28025 x 200ML) | $36.9K | 1 |
M/s. Delmaw Enterprises Ltd imports 13 distinct pharmaceutical formulations. Showing top 13 by value. For full formulation-level data, contact TransData Nexus.
What Products Does M/s. Delmaw Enterprises Ltd Import?
Top Products by Import Value
M/s. Delmaw Enterprises Ltd Therapeutic Categories — 2 Specializations
M/s. Delmaw Enterprises Ltd imports across 2 therapeutic categories, with Antihistamines & Allergy (50.0%), Diabetes & Endocrine (50.0%), representing the largest segments. The portfolio is concentrated — top 5 products = 100% of total imports.
Antihistamines & Allergy
1 products · 50.0% · $300.0K
Diabetes & Endocrine
1 products · 50.0% · $300.0K
Import Portfolio — Top 2 by Import Value
| # | Product | Category | Value | Ships | Share | Rk |
|---|---|---|---|---|---|---|
| 1 | Cetirizine | Antihistamines & Allergy | $300.0K | 6 | 0.2% | 1 |
| 2 | Metformin | Diabetes & Endocrine | $300.0K | 6 | 0.1% | 14 |
M/s. Delmaw Enterprises Ltd imports 2 pharmaceutical products across 2 categories into Uganda totaling $600.0K. The company is the #1 buyer for 1 product: Cetirizine.
Key Metrics
Top Categories
Indian Suppliers
Related Trade Data
Need Detailed Data?
Shipment-level records, supplier connections & pricing for M/s. Delmaw Enterprises Ltd.
Request DemoM/s. Delmaw Enterprises Ltd — Corporate Profile & Information
Company type, headquarters, distribution network, and industry role
1Company Overview
M/s. Delmaw Enterprises Ltd is a privately held pharmaceutical importer and wholesaler based in Kampala, Uganda. Established over 20 years ago, the company has built a reputation for distributing a wide range of pharmaceutical products, medical devices, and chemicals. As an ISO 9001:2005 certified entity, Delmaw Enterprises Ltd emphasizes quality management in its operations. The company is part of the Greater Artemis Life Sciences, under the Vista Group of Companies, and represents several international pharmaceutical manufacturers in Uganda, including Bliss GVS Healthcare Ltd., Fourrts Ltd., Strides Arcolab Ltd., Sance Laboratories, Universal Corporation Limited (UCL), and Xepa-Soul Pattinson (Malaysia).
The company's headquarters are located at Bombo Road, Gathani House, Kampala, Uganda. Delmaw Enterprises Ltd plays a pivotal role in Uganda's pharmaceutical distribution network, ensuring the availability of essential medicines and medical supplies across the country. By partnering with reputable international manufacturers, the company contributes significantly to the healthcare sector, addressing the diverse medical needs of the Ugandan population.
2Distribution Network
Delmaw Enterprises Ltd operates a centralized distribution model from its headquarters in Kampala. While specific details about warehouse locations and logistics capabilities are not publicly disclosed, the company's extensive import activities suggest a well-established infrastructure capable of handling significant volumes of pharmaceutical products. The company imports a diverse range of pharmaceutical formulations, including tablets, capsules, syrups, and injections, indicating a robust logistics network to manage these products efficiently. Given its strategic location in Kampala, Delmaw Enterprises Ltd is well-positioned to serve the entire Ugandan market, ensuring timely delivery of medical supplies to various regions.
3Industry Role
In Uganda's pharmaceutical supply chain, M/s. Delmaw Enterprises Ltd functions primarily as a wholesaler and importer. The company sources finished pharmaceutical formulations from international suppliers, particularly from India, and distributes these products to various stakeholders within the healthcare sector. By representing multiple international pharmaceutical manufacturers, Delmaw Enterprises Ltd acts as a key intermediary, facilitating the availability of a wide array of medical products in Uganda. This role is crucial in ensuring that healthcare providers have access to essential medicines and medical devices necessary for patient care.
Supplier Relationship Intelligence — M/s. Delmaw Enterprises Ltd
Sourcing concentration, supply chain resilience, and strategic implications
1Sourcing Concentration Analysis
Delmaw Enterprises Ltd demonstrates a high degree of sourcing concentration, with a total import value of $600,000 USD from India across 12 shipments. The company's portfolio is entirely concentrated on two products: Cetirizine and Metformin, each accounting for $300,000 USD in imports. This indicates a strategic focus on these two therapeutic categories—Antihistamines & Allergy and Diabetes & Endocrine—suggesting that Delmaw Enterprises Ltd is capitalizing on the significant demand for these medications in the Ugandan market.
The exclusive sourcing from a single supplier, Fourrts (India) Laboratories Private Limited, which accounts for all 12 shipments, reflects a strong and stable supplier relationship. However, this single-source dependency also poses potential risks, such as supply chain disruptions or changes in supplier pricing and policies. To mitigate these risks, it would be prudent for Delmaw Enterprises Ltd to consider diversifying its supplier base and exploring alternative sources for these key products.
2Supply Chain Resilience
The resilience of Delmaw Enterprises Ltd's supply chain is closely tied to its exclusive reliance on Fourrts (India) Laboratories Private Limited for the importation of Cetirizine and Metformin. While this single-source strategy may offer benefits such as streamlined logistics and potentially favorable pricing, it also exposes the company to risks associated with supply chain disruptions, regulatory changes, or operational challenges faced by the supplier. Currently, there is no publicly available information indicating the presence of backup suppliers or alternative sourcing strategies for these critical products.
To enhance supply chain resilience, Delmaw Enterprises Ltd should consider establishing relationships with additional suppliers and diversifying its product formulations. This approach would provide greater flexibility and security, ensuring a continuous and reliable supply of essential medications to the Ugandan market.
3Strategic Implications
Delmaw Enterprises Ltd's concentrated sourcing strategy positions the company as a dominant player in the Ugandan market for Cetirizine and Metformin. This focus allows the company to leverage economies of scale and establish strong relationships with its supplier, Fourrts (India) Laboratories Private Limited. However, the exclusive reliance on a single supplier and a narrow product portfolio may limit the company's ability to respond to market changes and customer demands for a broader range of pharmaceutical products.
For Indian exporters, the existing relationship between Delmaw Enterprises Ltd and Fourrts (India) Laboratories Private Limited may present challenges in entering the Ugandan market. To overcome this, Indian exporters should focus on offering differentiated products, competitive pricing, and value-added services that align with the specific needs of the Ugandan healthcare sector. Building strong relationships with local stakeholders and understanding the regulatory landscape will be crucial for establishing a foothold in this market.
Importing Pharmaceuticals into Uganda — Regulatory Framework
Regulatory authority, GMP requirements, import licensing for Uganda
1Regulatory Authority & Framework
The National Drug Authority (NDA) is the primary regulatory body responsible for overseeing the manufacture, importation, distribution, and licensing of pharmaceutical products in Uganda. Established in 1993, the NDA operates under the National Drug Policy and Authority Act, which provides the legal framework for drug regulation in the country.
The NDA's mandate includes ensuring the safety, efficacy, and quality of medicines available in Uganda. This involves regulating the importation and exportation of drugs, conducting inspections of manufacturing facilities, and enforcing compliance with Good Manufacturing Practices (GMP). The NDA also maintains a national list of essential drugs and formulary, which guides the importation and distribution of pharmaceutical products. (nda.or.ug)
2Import Licensing & GMP
Importers of pharmaceutical products into Uganda are required to obtain an import license from the National Drug Authority (NDA). This license is essential for ensuring that imported drugs meet the regulatory standards set by the NDA. The import license can be annual, issued to regular importers such as retail pharmacies, wholesale pharmacies, pharmaceutical manufacturers, or any organization that regularly imports drugs and related substances. Provisional import permits are issued to non-regular importers for specific consignments, typically valid for one month. (businesslicences.go.ug)
In addition to import licensing, the NDA enforces compliance with Good Manufacturing Practices (GMP) to ensure that pharmaceutical products are consistently produced and controlled according to quality standards. GMP inspections are conducted for both domestic and foreign manufacturing sites, and foreign manufacturing sites are required to pass an inspection prior to approval of the marketing authorization application. (nda.or.ug)
3Quality & Labeling
Imported pharmaceutical products must undergo batch testing to verify their quality, safety, and efficacy. The National Drug Authority (NDA) conducts inspections and sampling at ports of entry to ensure compliance with regulatory standards. Products are allowed entry into Uganda only through gazetted ports of entry, and importers must apply for import permits through the NDA Management Information System (NDAMIS). (nda.or.ug)
Labeling requirements for pharmaceutical products include the use of the English language and must comply with the NDA's guidelines. Specific requirements may include details such as the product name, active ingredients, dosage form, strength, batch number, manufacturing date, expiry date, storage conditions, and the name and address of the manufacturer or distributor. Serialization mandates may also apply to ensure traceability and prevent counterfeit products.
4Recent Regulatory Changes
Between 2024 and 2026, the National Drug Authority (NDA) implemented several policy changes affecting pharmaceutical imports into Uganda. These changes include the introduction of electronic applications for import permits through the NDA Management Information System (NDAMIS), streamlining the importation process and enhancing efficiency. Additionally, the NDA has updated its guidelines on the verification of applications for the importation and exportation of drugs, pharmaceutical raw materials, and packaging materials, emphasizing the need for compliance with international standards. (nda.or.ug)
The NDA has also revised the National Drug Policy and Authority (Importation and Exportation of Drugs) Regulations, 2014, to align with current international best practices and address emerging challenges in the pharmaceutical sector. These regulatory changes aim to improve the quality and safety of pharmaceutical products in Uganda and ensure that imported drugs meet the required standards. (ugandatrades.go.ug)
M/s. Delmaw Enterprises Ltd — Procurement Pattern Analysis
Product strategy, sourcing profile, and market positioning
1Product Strategy
M/s. Delmaw Enterprises Ltd's focus on importing Cetirizine and Metformin aligns with the significant demand for these medications in Uganda. Cetirizine, an antihistamine, is commonly used to treat allergy symptoms, while Metformin is a first-line medication for managing type 2 diabetes. The prevalence of allergic conditions and diabetes in Uganda underscores the importance of these drugs in the country's healthcare system.
By concentrating on these therapeutic areas, Delmaw Enterprises Ltd addresses critical health concerns and contributes to improving the quality of life
Frequently Asked Questions — M/s. Delmaw Enterprises Ltd
What products does M/s. Delmaw Enterprises Ltd import from India?
M/s. Delmaw Enterprises Ltd imports 2 pharmaceutical products across 2 categories. Top imports: Cetirizine ($300.0K), Metformin ($300.0K).
Who supplies pharmaceuticals to M/s. Delmaw Enterprises Ltd from India?
M/s. Delmaw Enterprises Ltd sources from 1 verified Indian suppliers. The primary supplier is Fourrts (india) Laboratories Private Limited (100.0% of imports, $2.8M).
What is M/s. Delmaw Enterprises Ltd's total pharmaceutical import value?
M/s. Delmaw Enterprises Ltd's total pharmaceutical import value from India is $600.0K, based on 12 verified shipments in Indian Customs (DGFT) data.
What therapeutic categories does M/s. Delmaw Enterprises Ltd focus on?
M/s. Delmaw Enterprises Ltd imports across 2 categories. The largest: Antihistamines & Allergy (50.0%), Diabetes & Endocrine (50.0%).
Get Full M/s. Delmaw Enterprises Ltd Import Intelligence
Access shipment-level details, supplier connections, pricing data, and competitive analysis. TransData Nexus provides verified Indian Customs (DGFT) data trusted by pharmaceutical trade professionals worldwide.
Official References & Regulatory Resources
Verify import regulations and drug registration requirements with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Buyer Matching: M/s. Delmaw Enterprises Ltd identified across shipments using consignee name normalization.
- 2.Statistical Normalization: Shipment values are statistically normalized to ensure accurate market share representation.
- 3.Market Share: Calculated per product as M/s. Delmaw Enterprises Ltd's capped value divided by total Indian exports for that product.
- 4.Shipment Count: Based on 12 individual customs records matching M/s. Delmaw Enterprises Ltd.
- 5.Supplier Verification: M/s. Delmaw Enterprises Ltd sources from 1 verified Indian suppliers across 13 formulations, confirmed from customs records.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
2 Products Tracked
2 therapeutic categories
Expert-Reviewed
By pharmaceutical trade specialists
Data Source & Methodology
Trade data sourced from Indian Customs (DGFT) export shipment records. Values represent FOB export value in USD. For current shipment-level data, contact TransData Nexus.