Lexon UK Limited
Pharmaceutical Importer · United Kingdom · Diuretics Focus · $1.4M Total Trade · DGFT Verified
Lexon UK Limited is a pharmaceutical importer based in United Kingdom with a total trade value of $1.4M across 2 products in 2 therapeutic categories. Based on 29 verified import shipments from Indian Customs (DGFT) records, the company actively imports across multiple product segments. Lexon UK Limited sources from 3 verified Indian suppliers, with Piramal Pharma Limited accounting for 58.3% of imports.
Lexon UK Limited — Import Portfolio & Supplier Network
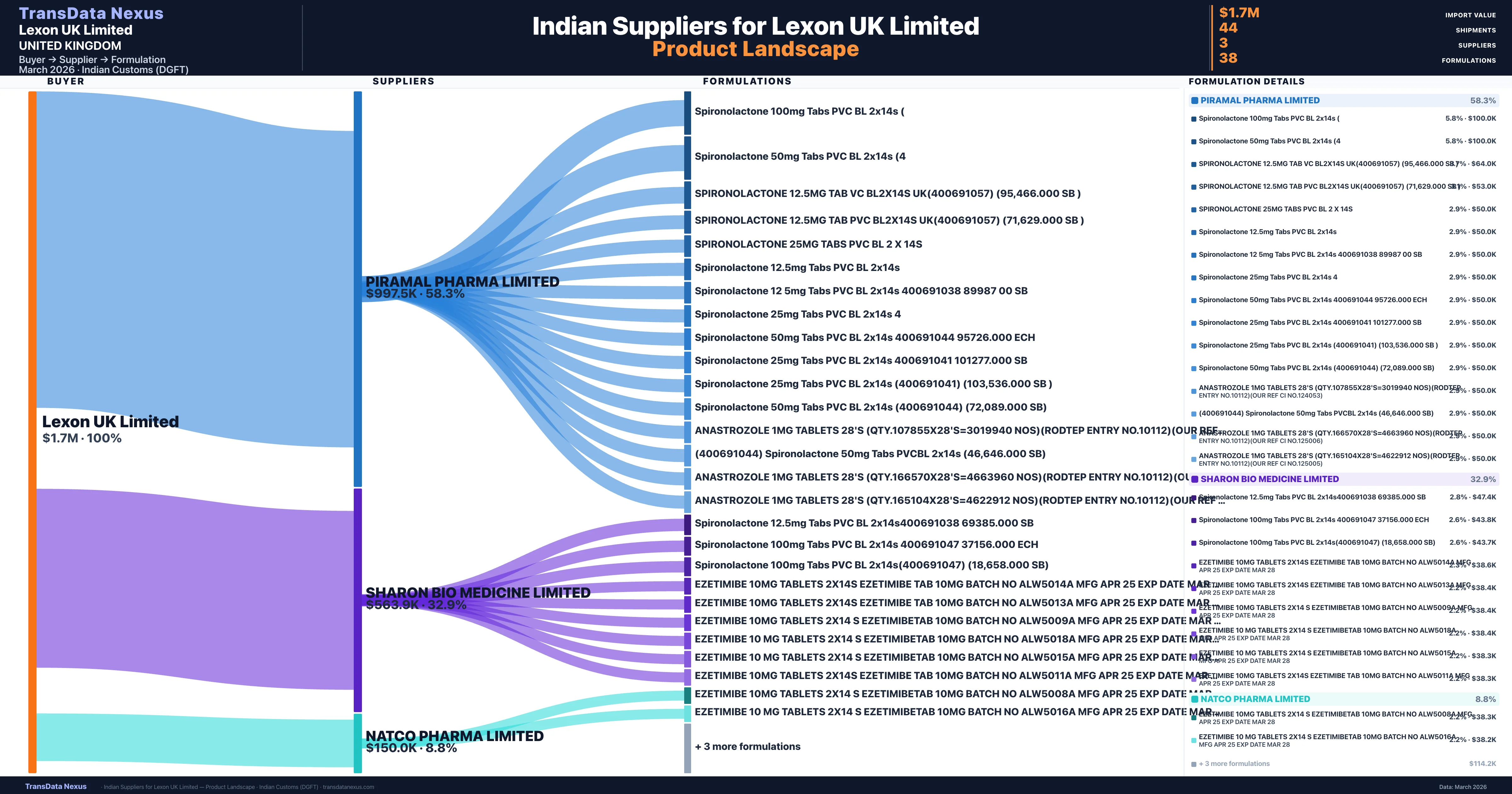
Who Are the Verified Indian Suppliers to Lexon UK Limited?
Customs-verified supplier relationships from Indian DGFT records
| Supplier | Value | Shipments | Share |
|---|---|---|---|
| Piramal Pharma Limited | $997.5K | 26 | 58.3% |
| Sharon Bio Medicine Limited | $563.9K | 15 | 32.9% |
| Natco Pharma Limited | $150.0K | 3 | 8.8% |
Lexon UK Limited sources from 3 verified Indian suppliers across 38 distinct formulations. Total import value: $1.7M across 44 shipments.
What Formulations Does Lexon UK Limited Import?
| Formulation | Value | Ships |
|---|---|---|
| Spironolactone 100MG tabs pvc bl 2x14s | $100.0K | 2 |
| Spironolactone 50MG tabs pvc bl 2x14s | $100.0K | 2 |
| Spironolactone 12.5MG TAB vc bl2x14s uk(400691057) (95,466.000 sb ) | $64.0K | 3 |
| Spironolactone 12.5MG TAB pvc bl2x14s uk(400691057) (71,629.000 sb ) | $53.0K | 3 |
| Spironolactone 25MG tabs pvc bl 2 x 14s | $50.0K | 1 |
| Spironolactone 12.5MG tabs pvc bl 2x14s | $50.0K | 1 |
| Spironolactone 12 5MG tabs pvc bl 2x14s 400691038 89987 00 | $50.0K | 1 |
| Spironolactone 25MG tabs pvc bl 2x14s | $50.0K | 1 |
| Spironolactone 50MG tabs pvc bl 2x14s 400691044 95726.000 | $50.0K | 1 |
| Spironolactone 25MG tabs pvc bl 2x14s 400691041 101277.000 | $50.0K | 1 |
| Spironolactone 25MG tabs pvc bl 2x14s (400691041) (103,536.000 sb ) | $50.0K | 1 |
| Spironolactone 50MG tabs pvc bl 2x14s (400691044) (72,089.000 sb) | $50.0K | 1 |
| Anastrozole 1MG tablets 28's (qty.107855x28's=3019940 nos)(rodtep entry no.10112)(our ref ci no.124053) | $50.0K | 1 |
| (400691044) spironolactone 50MG tabs pvcbl 2x14s (46,646.000 sb) | $50.0K | 1 |
| Anastrozole 1MG tablets 28's (qty.166570x28's=4663960 nos)(rodtep entry no.10112)(our ref ci no.125006) | $50.0K | 1 |
Lexon UK Limited imports 38 distinct pharmaceutical formulations. Showing top 15 by value. For full formulation-level data, contact TransData Nexus.
What Products Does Lexon UK Limited Import?
Top Products by Import Value
Lexon UK Limited Therapeutic Categories — 2 Specializations
Lexon UK Limited imports across 2 therapeutic categories, with Diuretics (89.7%), Advanced Oncology (10.3%), representing the largest segments. The portfolio is concentrated — top 5 products = 100% of total imports.
Diuretics
1 products · 89.7% · $1.3M
Advanced Oncology
1 products · 10.3% · $150.0K
Import Portfolio — Top 2 by Import Value
| # | Product | Category | Value | Ships | Share | Rk |
|---|---|---|---|---|---|---|
| 1 | Spironolactone | Diuretics | $1.3M | 26 | 0.7% | 14 |
| 2 | Anastrozole | Advanced Oncology | $150.0K | 3 | 0.1% | 14 |
Lexon UK Limited imports 2 pharmaceutical products across 2 categories into United Kingdom totaling $1.4M.
Key Metrics
Top Categories
Indian Suppliers
Related Trade Data
Need Detailed Data?
Shipment-level records, supplier connections & pricing for Lexon UK Limited.
Request DemoLexon UK Limited — Corporate Profile & Information
Company type, headquarters, distribution network, and industry role
1Company Overview
Lexon UK Limited, established in 1995, is a prominent pharmaceutical wholesaler headquartered in Redditch, England. The company specializes in the distribution of pharmaceutical products, including both prescription and over-the-counter medications, as well as veterinary medicines. With a workforce of approximately 683 employees, Lexon UK Limited serves a diverse clientele, including retail pharmacies, hospitals, and healthcare providers across the United Kingdom.
In April 2023, Lexon UK Limited was acquired by Well Pharmacy, a subsidiary of the Bestway Group. This acquisition aimed to enhance Well Pharmacy's presence in the pharmaceutical wholesale sector and expand its distribution capabilities. The integration of Lexon UK Limited into Well Pharmacy's operations is expected to strengthen the supply chain and improve service delivery to healthcare providers and patients nationwide. (easterneye.biz)
2Distribution Network
Lexon UK Limited operates a comprehensive distribution network with multiple depots strategically located across the United Kingdom and Ireland. These facilities are situated in Redditch, Leeds, Durham, East Kilbride, and Dublin, enabling the company to effectively serve a wide range of customers. The distribution centers are equipped with advanced logistics capabilities, including temperature-controlled storage and efficient order fulfillment systems, ensuring timely and secure delivery of pharmaceutical products. (easterneye.biz)
The geographic coverage of Lexon UK Limited's distribution network spans the Midlands, northwest, and northeast of England, as well as parts of Ireland. This extensive reach allows the company to cater to the needs of approximately 3,000 retail pharmacy customers and numerous community pharmacies, facilitating access to essential medications across these regions. (easterneye.biz)
3Industry Role
As a pharmaceutical wholesaler, Lexon UK Limited plays a pivotal role in the United Kingdom's pharmaceutical supply chain. The company sources a diverse range of pharmaceutical products from various manufacturers, including generic and over-the-counter medications, and distributes them to retail pharmacies, hospitals, and other healthcare providers. This intermediary function ensures that essential medicines are readily available to meet the demands of the healthcare sector.
In addition to its wholesale operations, Lexon UK Limited has expanded its services to include the operation of community pharmacies under the trading name Knights Pharmacy. This vertical integration allows the company to directly serve patients, providing a comprehensive approach to pharmaceutical distribution and retail. (easterneye.biz)
Supplier Relationship Intelligence — Lexon UK Limited
Sourcing concentration, supply chain resilience, and strategic implications
1Sourcing Concentration Analysis
Lexon UK Limited's sourcing strategy exhibits a notable concentration, with a significant portion of its pharmaceutical imports originating from India. Between 2022 and 2026, the company imported finished pharmaceutical formulations valued at $1.4 million USD from India, encompassing 29 shipments and 38 unique formulations. The top two products imported were Spironolactone ($1.3 million, 26 shipments) and Anastrozole ($150,000, 3 shipments). These imports were sourced from three Indian suppliers: Piramal Pharma Limited, Sharon Bio Medicine Limited, and Natco Pharma Limited.
The high concentration of imports from a limited number of suppliers indicates a strategic sourcing approach, potentially leveraging established relationships and favorable terms. However, this dependency also introduces risks related to supply chain disruptions, such as production delays or regulatory changes in the supplier's country. The stability of these supplier relationships is crucial for maintaining a consistent supply of essential pharmaceutical products.
2Supply Chain Resilience
The resilience of Lexon UK Limited's supply chain, particularly concerning its Indian suppliers, is a critical factor in ensuring uninterrupted access to pharmaceutical products. The company's reliance on three primary suppliers for a substantial portion of its imports suggests a streamlined procurement process but also highlights potential vulnerabilities.
To mitigate risks associated with supplier concentration, Lexon UK Limited may consider diversifying its supplier base by engaging with additional manufacturers in India or other countries. This strategy would enhance supply chain flexibility and reduce the impact of potential disruptions. Additionally, establishing contingency plans and maintaining open communication channels with existing suppliers can further bolster the resilience of the supply chain.
3Strategic Implications
The sourcing pattern of Lexon UK Limited, characterized by a concentrated supplier base, has significant strategic implications. For the company, this approach allows for streamlined procurement and potentially better pricing due to bulk purchasing agreements. However, it also necessitates careful management of supplier relationships to ensure consistent product quality and timely delivery.
For Indian pharmaceutical exporters, the existing relationship between Lexon UK Limited and its current suppliers presents both challenges and opportunities. While entering an established supply chain may require overcoming existing supplier preferences, there is potential to offer competitive advantages such as superior product quality, innovative formulations, or more favorable terms. Understanding the specific needs and preferences of Lexon UK Limited can facilitate the development of tailored proposals to become an alternative supplier.
Importing Pharmaceuticals into United Kingdom — Regulatory Framework
Regulatory authority, GMP requirements, import licensing for United Kingdom
1Regulatory Authority & Framework
In the United Kingdom, the Medicines and Healthcare products Regulatory Agency (MHRA) serves as the primary regulatory authority overseeing the safety, quality, and efficacy of medicines and medical devices. The MHRA is responsible for granting marketing authorizations, conducting inspections, and ensuring compliance with Good Manufacturing Practice (GMP) standards.
The regulatory framework governing pharmaceutical imports into the UK is primarily outlined in the Human Medicines Regulations 2012. These regulations stipulate the requirements for importing, manufacturing, and distributing medicinal products, including the necessity for importers to hold a Wholesale Dealer’s License (WDA) and comply with GMP standards.
2Import Licensing & GMP
Importers of pharmaceutical products into the United Kingdom must obtain a Wholesale Dealer’s License (WDA) from the MHRA. This license authorizes the holder to import, store, and distribute medicinal products within the UK. Additionally, imported medicines must comply with GMP standards, ensuring that they are manufactured and tested in accordance with internationally recognized quality standards.
The MHRA recognizes GMP certificates from various regulatory bodies, including the European Medicines Agency (EMA), the World Health Organization (WHO), and the Pharmaceutical Inspection Co-operation Scheme (PIC/S). This recognition facilitates the importation of medicines from countries with established GMP standards, such as India.
3Quality & Labeling
Imported pharmaceutical products must undergo batch testing to verify their quality, safety, and efficacy. This testing ensures that the products meet the standards set by the MHRA and are safe for patient use. Stability requirements are also enforced to ensure that medicines maintain their quality throughout their shelf life.
Labeling requirements stipulate that all information on pharmaceutical products must be in English, providing clear instructions and safety information to patients and healthcare providers. Serialization mandates may also apply, requiring unique identifiers on packaging to enhance traceability and prevent counterfeit products from entering the supply chain.
4Recent Regulatory Changes
Between 2024 and 2026, several regulatory changes have impacted the importation of pharmaceutical products into the United Kingdom. The MHRA has updated guidelines to align with international standards, including revisions to GMP requirements and the recognition of additional regulatory bodies' certifications. These changes aim to streamline the import process and ensure the continued safety and efficacy of medicines available in the UK market.
Lexon UK Limited — Procurement Pattern Analysis
Product strategy, sourcing profile, and market positioning
1Product Strategy
Lexon UK Limited's focus on importing finished pharmaceutical formulations, particularly in the therapeutic categories of diuretics and advanced oncology, aligns with market demands for essential and specialized medications. The substantial import value of Spironolactone and Anastrozole indicates a strategic emphasis on these high-demand products. This product strategy is driven by the need to provide healthcare providers with reliable access to critical treatments for conditions such as hypertension, heart failure, and various cancers.
The company's sourcing of these products from India reflects a strategic decision to leverage cost-effective manufacturing while maintaining product quality. India's established pharmaceutical industry, with its adherence to international GMP standards, offers a reliable supply of high-quality generic medications.
2Sourcing Profile
Lexon UK Limited's sourcing strategy is centered on importing generic drug formulations, primarily from India. This approach allows the company to offer cost-effective alternatives to branded medications, making essential treatments more accessible to patients. The focus on finished formulations, rather than raw active pharmaceutical ingredients (APIs), indicates a preference for ready-to-market products that can be directly distributed to healthcare providers.
India's role as a key supplier in Lexon UK Limited's procurement strategy underscores the importance of maintaining strong relationships with Indian pharmaceutical manufacturers. Ensuring compliance with international quality standards and regulatory requirements is crucial for sustaining this sourcing model.
Frequently Asked Questions — Lexon UK Limited
What products does Lexon UK Limited import from India?
Lexon UK Limited imports 2 pharmaceutical products across 2 categories. Top imports: Spironolactone ($1.3M), Anastrozole ($150.0K).
Who supplies pharmaceuticals to Lexon UK Limited from India?
Lexon UK Limited sources from 3 verified Indian suppliers. The primary supplier is Piramal Pharma Limited (58.3% of imports, $997.5K).
What is Lexon UK Limited's total pharmaceutical import value?
Lexon UK Limited's total pharmaceutical import value from India is $1.4M, based on 29 verified shipments in Indian Customs (DGFT) data.
What therapeutic categories does Lexon UK Limited focus on?
Lexon UK Limited imports across 2 categories. The largest: Diuretics (89.7%), Advanced Oncology (10.3%).
Get Full Lexon UK Limited Import Intelligence
Access shipment-level details, supplier connections, pricing data, and competitive analysis. TransData Nexus provides verified Indian Customs (DGFT) data trusted by pharmaceutical trade professionals worldwide.
Official References & Regulatory Resources
Verify import regulations and drug registration requirements with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Buyer Matching: Lexon UK Limited identified across shipments using consignee name normalization.
- 2.Statistical Normalization: Shipment values are statistically normalized to ensure accurate market share representation.
- 3.Market Share: Calculated per product as Lexon UK Limited's capped value divided by total Indian exports for that product.
- 4.Shipment Count: Based on 29 individual customs records matching Lexon UK Limited.
- 5.Supplier Verification: Lexon UK Limited sources from 3 verified Indian suppliers across 38 formulations, confirmed from customs records.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
2 Products Tracked
2 therapeutic categories
Expert-Reviewed
By pharmaceutical trade specialists
Data Source & Methodology
Trade data sourced from Indian Customs (DGFT) export shipment records. Values represent FOB export value in USD. For current shipment-level data, contact TransData Nexus.