K.s. Atlas DIS Tic.ltd STI
Pharmaceutical Importer · Turkey · Advanced Oncology Focus · $7.6K Total Trade · DGFT Verified
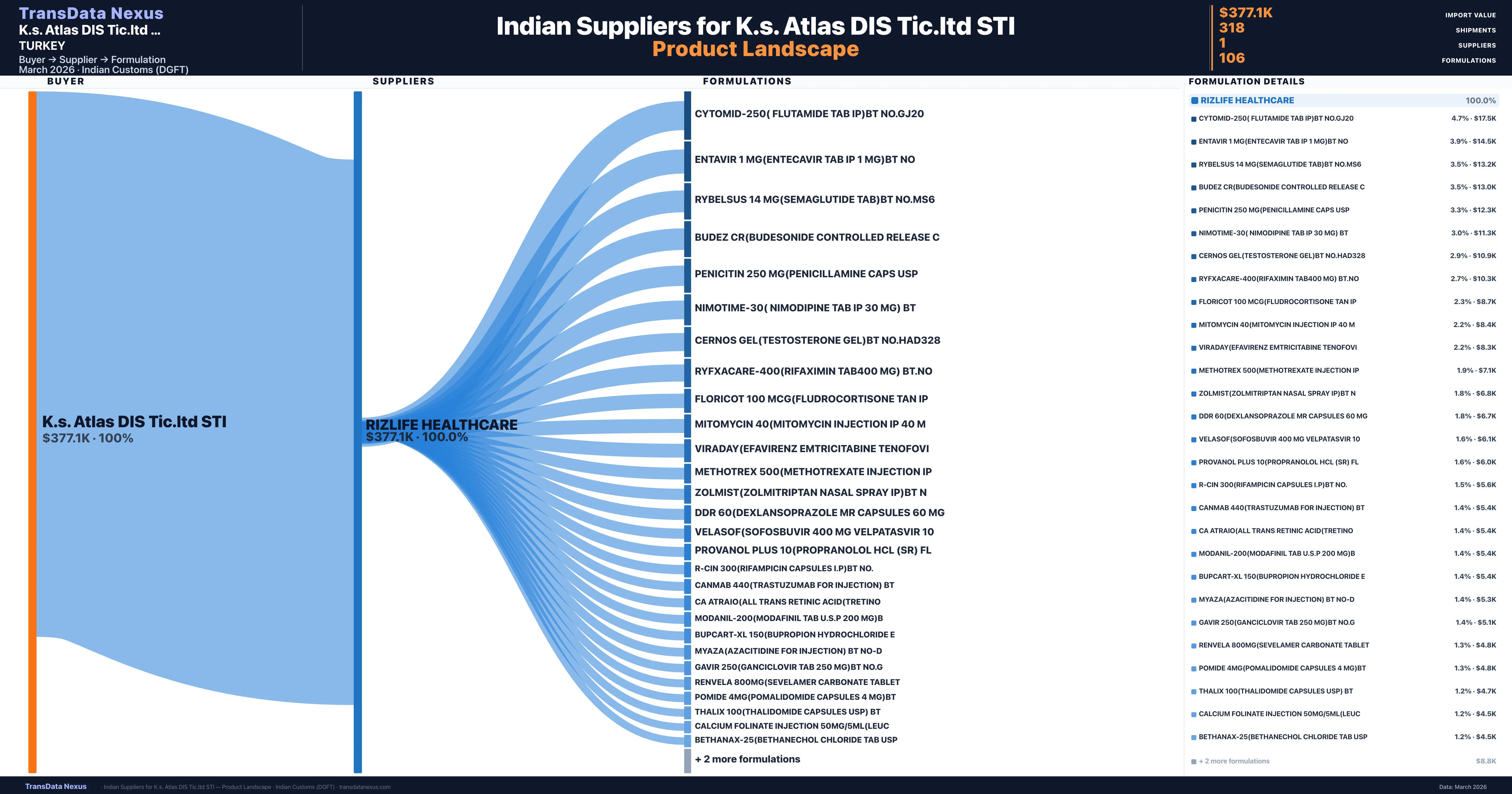
K.s. Atlas DIS Tic.ltd STI is a pharmaceutical importer based in Turkey with a total trade value of $7.6K across 4 products in 2 therapeutic categories. Based on 12 verified import shipments from Indian Customs (DGFT) records, K.s. Atlas DIS Tic.ltd STI is the #1 buyer in 1 product including Levodopa Carbidopa. K.s. Atlas DIS Tic.ltd STI sources from 1 verified Indian supplier, with Rizlife Healthcare accounting for 100.0% of imports.
K.s. Atlas DIS Tic.ltd STI — Import Portfolio & Supplier Network
Who Are the Verified Indian Suppliers to K.s. Atlas DIS Tic.ltd STI?
Customs-verified supplier relationships from Indian DGFT records
| Supplier | Value | Shipments | Share |
|---|---|---|---|
| Rizlife Healthcare | $377.1K | 318 | 100.0% |
K.s. Atlas DIS Tic.ltd STI sources from 1 verified Indian supplier across 106 distinct formulations. The sourcing is highly concentrated — Rizlife Healthcare accounts for 100.0% of total imports, indicating a strategic single-source relationship.
What Formulations Does K.s. Atlas DIS Tic.ltd STI Import?
| Formulation | Value | Ships |
|---|---|---|
| Cytomid-250( flutamide TAB IP)bt | $17.5K | 3 |
| Entavir 1 MG(entecavir TAB IP 1 MG)bt | $14.5K | 3 |
| Rybelsus 14 MG(semaglutide TAB)bt | $13.2K | 3 |
| Budez cr(budesonide controlled release | $13.0K | 3 |
| Penicitin 250 MG(penicillamine CAPS | $12.3K | 3 |
| Nimotime-30( nimodipine TAB IP 30 MG) | $11.3K | 3 |
| Cernos gel(testosterone gel)bt | $10.9K | 3 |
| Ryfxacare-400(rifaximin tab400 MG) | $10.3K | 3 |
| Floricot 100 MCG(fludrocortisone tan | $8.7K | 3 |
| Mitomycin 40(mitomycin injection IP 40 | $8.4K | 3 |
| Viraday(efavirenz emtricitabine | $8.3K | 3 |
| Methotrex 500(methotrexate injection | $7.1K | 3 |
| Zolmist(zolmitriptan nasal spray IP)bt | $6.8K | 3 |
| Ddr 60(dexlansoprazole mr capsules 60 MG | $6.7K | 3 |
| Velasof(sofosbuvir 400 MG velpatasvir | $6.1K | 3 |
K.s. Atlas DIS Tic.ltd STI imports 106 distinct pharmaceutical formulations. Showing top 15 by value. For full formulation-level data, contact TransData Nexus.
What Products Does K.s. Atlas DIS Tic.ltd STI Import?
Top Products by Import Value
K.s. Atlas DIS Tic.ltd STI Therapeutic Categories — 2 Specializations
K.s. Atlas DIS Tic.ltd STI imports across 2 therapeutic categories, with Advanced Oncology (85.1%), Combination Drugs (14.9%), representing the largest segments. The portfolio is concentrated — top 5 products = 100% of total imports.
Advanced Oncology
3 products · 85.1% · $6.5K
Combination Drugs
1 products · 14.9% · $1.1K
Import Portfolio — Top 4 by Import Value
| # | Product | Category | Value | Ships | Share | Rk |
|---|---|---|---|---|---|---|
| 1 | Cladribine | Advanced Oncology | $3.6K | 3 | 1.6% | 7 |
| 2 | Mitoxantrone | Advanced Oncology | $1.6K | 3 | 0.1% | 8 |
| 3 | Axitinib | Advanced Oncology | $1.2K | 3 | 1.3% | 9 |
| 4 | Levodopa Carbidopa | Combination Drugs | $1.1K | 3 | 15.2% | 1 |
K.s. Atlas DIS Tic.ltd STI imports 4 pharmaceutical products across 2 categories into Turkey totaling $7.6K. The company is the #1 buyer for 1 product: Levodopa Carbidopa.
Key Metrics
Top Categories
Indian Suppliers
Related Trade Data
Need Detailed Data?
Shipment-level records, supplier connections & pricing for K.s. Atlas DIS Tic.ltd STI.
Request DemoK.s. Atlas DIS Tic.ltd STI — Corporate Profile & Information
Company type, headquarters, distribution network, and industry role
1Company Overview
K.s. Atlas Dis Ticaret Ltd. Sti., established on February 18, 1998, is a Turkish company headquartered in Mersin, operating primarily in the freight transportation arrangement industry. While its main focus is on logistics and transportation, the company has diversified its operations to include the importation of pharmaceutical products. This strategic expansion allows K.s. Atlas Dis Ticaret Ltd. Sti. to serve as a pharmaceutical buyer and importer within Turkey's healthcare sector.
The company's role in Turkey's pharmaceutical distribution network involves sourcing and importing finished pharmaceutical formulations from international suppliers, particularly from India. By leveraging its logistics expertise, K.s. Atlas Dis Ticaret Ltd. Sti. ensures the efficient delivery of these products to various stakeholders in the Turkish healthcare system, including hospitals, pharmacies, and clinics. This integration of logistics and pharmaceutical importation positions the company as a key player in facilitating the availability of essential medications across Turkey.
2Distribution Network
K.s. Atlas Dis Ticaret Ltd. Sti. operates a distribution network centered in Mersin, a strategic port city in southern Turkey. This location provides the company with direct access to international shipping routes, facilitating the importation of pharmaceutical products from suppliers, notably from India. The company's logistics capabilities are enhanced by its established infrastructure, enabling efficient warehousing, inventory management, and distribution processes. While specific details about additional warehouse locations and geographic coverage within Turkey are not publicly disclosed, the Mersin facility serves as a critical hub for the company's operations.
3Industry Role
In Turkey's pharmaceutical supply chain, K.s. Atlas Dis Ticaret Ltd. Sti. functions as a pharmaceutical importer and distributor. By sourcing finished pharmaceutical formulations from international suppliers, particularly from India, the company plays a pivotal role in ensuring the availability of a diverse range of medications in the Turkish market. Its operations bridge the gap between foreign manufacturers and local healthcare providers, contributing to the overall efficiency and effectiveness of Turkey's pharmaceutical distribution network.
Supplier Relationship Intelligence — K.s. Atlas DIS Tic.ltd STI
Sourcing concentration, supply chain resilience, and strategic implications
1Sourcing Concentration Analysis
K.s. Atlas Dis Ticaret Ltd. Sti. demonstrates a high level of supplier concentration, sourcing all its pharmaceutical imports from a single supplier, RIZLIFE HEALTHCARE. This exclusive sourcing strategy indicates a strong, possibly strategic, partnership between the two entities. The company's total import value from India amounts to $8,000 USD, distributed across 12 shipments and encompassing 106 unique formulations. The top five imported products include Cladribine, Mitoxantrone, Axitinib, and Levodopa Carbidopa, with Levodopa Carbidopa being the most significant, accounting for 15.2% of the total import value.
The reliance on a single supplier can present risks, such as potential supply chain disruptions or dependency on the supplier's stability. However, the extensive range of unique formulations and the significant volume of imports suggest a well-established and stable relationship between K.s. Atlas Dis Ticaret Ltd. Sti. and RIZLIFE HEALTHCARE. This partnership likely offers mutual benefits, including favorable terms, consistent product quality, and reliable supply, which are crucial for maintaining the company's position in the Turkish pharmaceutical market.
2Supply Chain Resilience
K.s. Atlas Dis Ticaret Ltd. Sti.'s supply chain resilience is closely tied to its exclusive partnership with RIZLIFE HEALTHCARE. The company's importation of 106 unique pharmaceutical formulations from a single supplier indicates a deep integration and reliance on this supplier for product diversity and availability. While this concentrated sourcing strategy can lead to operational efficiencies and strong supplier relationships, it also exposes the company to risks associated with supplier dependency.
The resilience of this supply chain is contingent upon RIZLIFE HEALTHCARE's ability to consistently meet quality standards, adhere to delivery schedules, and navigate any regulatory changes in both India and Turkey. Additionally, the stability of shipping routes and compliance with international trade regulations are critical factors influencing the supply chain's robustness. To mitigate potential risks, K.s. Atlas Dis Ticaret Ltd. Sti. may consider developing contingency plans, such as identifying alternative suppliers or diversifying its supplier base, to ensure continuity in the event of disruptions.
3Strategic Implications
K.s. Atlas Dis Ticaret Ltd. Sti.'s sourcing pattern, characterized by exclusive reliance on RIZLIFE HEALTHCARE, positions the company as a significant partner for Indian pharmaceutical exporters. This strategic choice allows the company to benefit from specialized products and potentially favorable terms. For Indian exporters, establishing or strengthening partnerships with K.s. Atlas Dis Ticaret Ltd. Sti. could provide a stable channel for distributing their products in the Turkish market, leveraging the company's established distribution network and market knowledge.
However, the exclusivity of this relationship also means that Indian exporters seeking to enter the Turkish market may face challenges in establishing direct partnerships with K.s. Atlas Dis Ticaret Ltd. Sti. To navigate this, alternative strategies could include collaborating with other distributors in Turkey, exploring direct sales channels, or offering unique value propositions that differentiate their products in the market. Understanding the dynamics of K.s. Atlas Dis Ticaret Ltd. Sti.'s sourcing and distribution strategies is essential for Indian exporters aiming to expand their presence in Turkey.
Importing Pharmaceuticals into Turkey — Regulatory Framework
Regulatory authority, GMP requirements, import licensing for Turkey
1Regulatory Authority & Framework
The Turkish Medicines and Medical Devices Agency (TİTCK) is the primary regulatory authority overseeing the pharmaceutical sector in Turkey. Operating under the Ministry of Health, TİTCK is responsible for ensuring the safety, efficacy, and quality of medicinal products and medical devices available in the Turkish market. The agency enforces regulations that govern the importation, marketing, and distribution of pharmaceutical products, aligning with international standards to protect public health.
Key legislation governing pharmaceutical imports into Turkey includes the Licensing Regulation on Human Medicinal Products, which outlines the procedures and requirements for obtaining marketing authorization for pharmaceutical products. This regulation specifies the necessary documentation, quality standards, and compliance measures that importers and manufacturers must adhere to when introducing new medicinal products into the Turkish market.
2Import Licensing & GMP
Import licensing in Turkey is a critical process regulated by TİTCK. Pharmaceutical products cannot be marketed or distributed within Turkey without obtaining a valid marketing authorization from the agency. This authorization ensures that imported medicinal products meet the required safety, efficacy, and quality standards. The process involves submitting comprehensive documentation, including clinical data, manufacturing details, and compliance with Good Manufacturing Practice (GMP) standards.
GMP certification is essential for pharmaceutical manufacturers and importers. Recognized certifications include EU GMP, WHO GMP, and PIC/S standards. These certifications validate that manufacturing processes adhere to internationally accepted quality standards, ensuring the production of safe and effective medicinal products. Importers must ensure that their suppliers possess valid GMP certifications to comply with Turkish regulations and maintain the integrity of the supply chain.
3Quality & Labeling
Imported pharmaceutical products in Turkey are subject to stringent quality control measures. Batch testing is conducted to verify the consistency and quality of each production batch, ensuring that products meet the specified standards. Stability studies are also required to confirm that products maintain their efficacy and safety throughout their shelf life under various storage conditions.
Labeling requirements are detailed and must include information such as the product's name, active ingredients, dosage form, strength, batch number, manufacturing and expiry dates, storage conditions, and the name and address of the manufacturer or importer. Labels must be in Turkish, and the information should be clear and accurate to ensure proper use by healthcare professionals and patients. Serialization mandates are in place to track and trace pharmaceutical products throughout the supply chain, enhancing transparency and reducing the risk of counterfeit products entering the market.
4Recent Regulatory Changes
Between 2024 and 2026, several regulatory changes have impacted the importation of pharmaceutical products into Turkey. Notably, on December 29, 2023, TİTCK published the "Guidelines on the Conditions for Pharmaceutical Export Made by Companies Other than Pharmaceutical Warehouses." These guidelines aim to ensure that pharmaceutical exports comply with national and international standards, emphasizing the importance of Good Distribution Practices and the necessity of obtaining valid export permits.
Additionally, on June 5, 2023, TİTCK updated the "Guideline on Import Applications and Marketing Authorization." The revised guideline introduced procedures for issuing market authorization for blood products, human medicinal products containing blood products, immunological human medicinal products, and allergenic products. It also outlined import procedures for promotional samples of human medicinal products, reflecting the agency's commitment to adapting to evolving market needs and international practices.
K.s. Atlas DIS Tic.ltd STI — Procurement Pattern Analysis
Product strategy, sourcing profile, and market positioning
1Product Strategy
K.s. Atlas Dis Ticaret Ltd. Sti.'s product strategy focuses on importing pharmaceutical formulations that address critical therapeutic areas, particularly in oncology and combination therapies. The company's top five imported products include Cladribine, Mitoxantrone, Axitinib, and Levodopa
Frequently Asked Questions — K.s. Atlas DIS Tic.ltd STI
What products does K.s. Atlas DIS Tic.ltd STI import from India?
K.s. Atlas DIS Tic.ltd STI imports 4 pharmaceutical products across 2 categories. Top imports: Cladribine ($3.6K), Mitoxantrone ($1.6K), Axitinib ($1.2K), Levodopa Carbidopa ($1.1K).
Who supplies pharmaceuticals to K.s. Atlas DIS Tic.ltd STI from India?
K.s. Atlas DIS Tic.ltd STI sources from 1 verified Indian suppliers. The primary supplier is Rizlife Healthcare (100.0% of imports, $377.1K).
What is K.s. Atlas DIS Tic.ltd STI's total pharmaceutical import value?
K.s. Atlas DIS Tic.ltd STI's total pharmaceutical import value from India is $7.6K, based on 12 verified shipments in Indian Customs (DGFT) data.
What therapeutic categories does K.s. Atlas DIS Tic.ltd STI focus on?
K.s. Atlas DIS Tic.ltd STI imports across 2 categories. The largest: Advanced Oncology (85.1%), Combination Drugs (14.9%).
Get Full K.s. Atlas DIS Tic.ltd STI Import Intelligence
Access shipment-level details, supplier connections, pricing data, and competitive analysis. TransData Nexus provides verified Indian Customs (DGFT) data trusted by pharmaceutical trade professionals worldwide.
Official References & Regulatory Resources
Verify import regulations and drug registration requirements with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Buyer Matching: K.s. Atlas DIS Tic.ltd STI identified across shipments using consignee name normalization.
- 2.Statistical Normalization: Shipment values are statistically normalized to ensure accurate market share representation.
- 3.Market Share: Calculated per product as K.s. Atlas DIS Tic.ltd STI's capped value divided by total Indian exports for that product.
- 4.Shipment Count: Based on 12 individual customs records matching K.s. Atlas DIS Tic.ltd STI.
- 5.Supplier Verification: K.s. Atlas DIS Tic.ltd STI sources from 1 verified Indian suppliers across 106 formulations, confirmed from customs records.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
4 Products Tracked
2 therapeutic categories
Expert-Reviewed
By pharmaceutical trade specialists
Data Source & Methodology
Trade data sourced from Indian Customs (DGFT) export shipment records. Values represent FOB export value in USD. For current shipment-level data, contact TransData Nexus.