I.e. Medica Inc.
Pharmaceutical Importer · Philippines · Diabetes & Endocrine Focus · $4.9M Total Trade · DGFT Verified
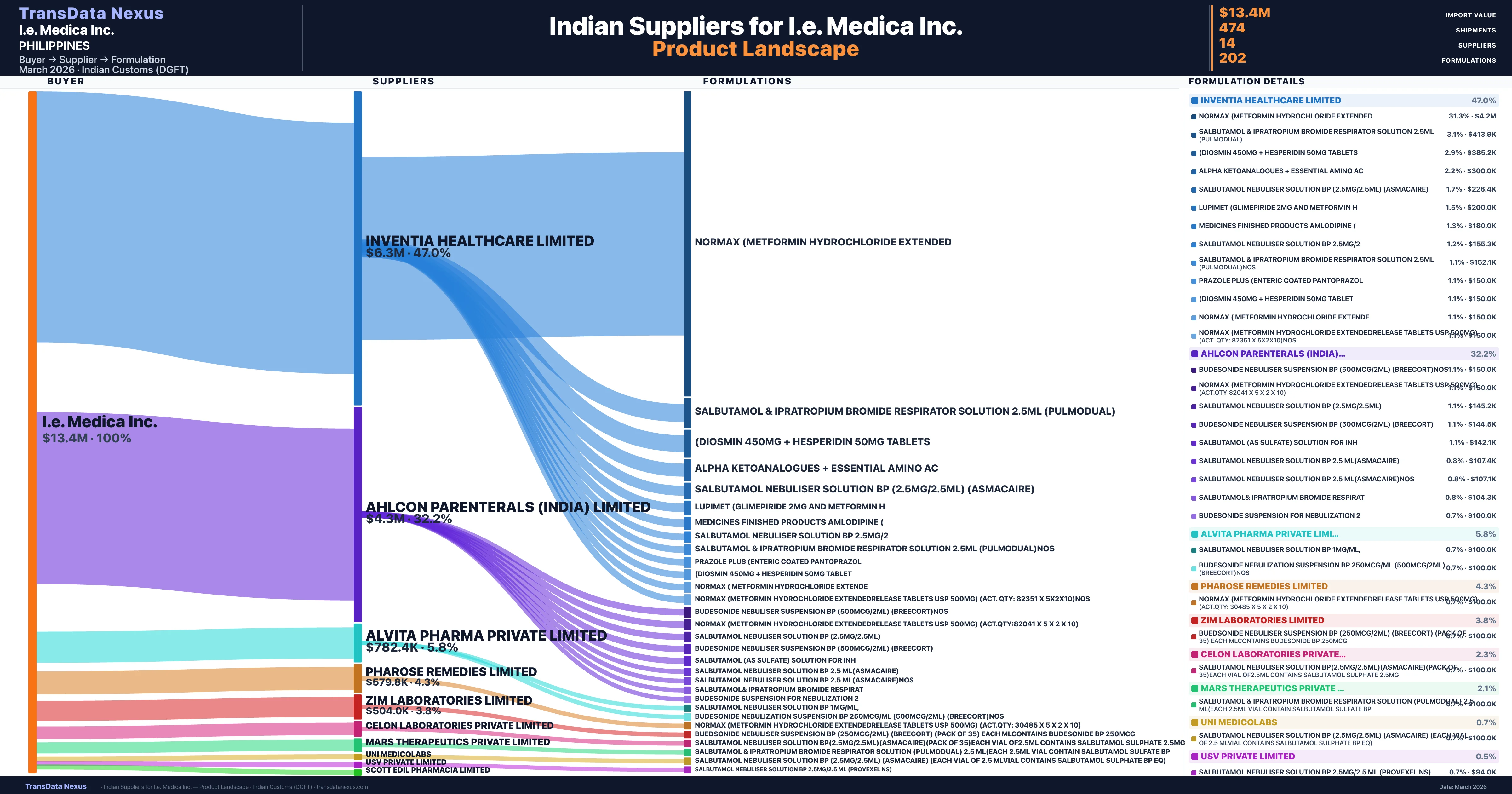
I.e. Medica Inc. is a pharmaceutical importer based in Philippines with a total trade value of $4.9M across 3 products in 2 therapeutic categories. Based on 102 verified import shipments from Indian Customs (DGFT) records, the company actively imports across multiple product segments. I.e. Medica Inc. sources from 14 verified Indian suppliers, with Inventia Healthcare Limited accounting for 47.0% of imports.
I.e. Medica Inc. — Import Portfolio & Supplier Network
Who Are the Verified Indian Suppliers to I.e. Medica Inc.?
Customs-verified supplier relationships from Indian DGFT records
| Supplier | Value | Shipments | Share |
|---|---|---|---|
| Inventia Healthcare Limited | $6.3M | 158 | 47.0% |
| Ahlcon Parenterals (india) Limited | $4.3M | 172 | 32.2% |
| Alvita Pharma Private Limited | $782.4K | 29 | 5.8% |
| Pharose Remedies Limited | $579.8K | 13 | 4.3% |
| Zim Laboratories Limited | $504.0K | 23 | 3.8% |
| Celon Laboratories Private Limited | $313.6K | 35 | 2.3% |
| Mars Therapeutics Private Limited | $277.4K | 18 | 2.1% |
| Uni Medicolabs | $89.8K | 10 | 0.7% |
| Usv Private Limited | $67.4K | 2 | 0.5% |
| Scott Edil Pharmacia Limited | $59.9K | 6 | 0.4% |
| Scott-edil Advance Research Laboratories & Educati | $43.0K | 3 | 0.3% |
| Piramal Pharma Limited | $29.9K | 1 | 0.2% |
| Wiscon Pharmaceuticals Private Limited | $21.6K | 3 | 0.2% |
| Indeus Life Sciences Private Limited | $17.9K | 1 | 0.1% |
I.e. Medica Inc. sources from 14 verified Indian suppliers across 202 distinct formulations. The supply base is diversified across 14 suppliers, reducing single-source dependency risk.
What Formulations Does I.e. Medica Inc. Import?
| Formulation | Value | Ships |
|---|---|---|
| Normax (metformin hydrochloride | $4.2M | 84 |
| Salbutamol & ipratropium bromide respirator solution 2.5ML (pulmodual) | $413.9K | 11 |
| (diosmin 450MG + hesperidin 50MG | $385.2K | 13 |
| Alpha ketoanalogues + essential amino | $300.0K | 7 |
| Salbutamol nebuliser solution BP (2.5MG/2.5ML) (asmacaire) | $226.4K | 6 |
| Lupimet (glimepiride 2MG and metformin | $200.0K | 4 |
| Medicines finished products amlodipine | $180.0K | 8 |
| Salbutamol nebuliser solution BP | $155.3K | 5 |
| Salbutamol & ipratropium bromide respirator solution 2.5ML (pulmodual | $152.1K | 6 |
| Prazole plus (enteric coated | $150.0K | 3 |
| (diosmin 450MG + hesperidin 50MG | $150.0K | 3 |
| Normax ( metformin hydrochloride | $150.0K | 3 |
| Normax (metformin hydrochloride extendedrelease tablets USP 500MG) (act. qty: 82351 x 5x2x10 | $150.0K | 3 |
| Budesonide nebuliser suspension BP (500mcg/2ML) (breecort | $150.0K | 3 |
| Normax (metformin hydrochloride extendedrelease tablets USP 500MG) (act.qty:82041 x 5 x 2 x 10) | $150.0K | 3 |
I.e. Medica Inc. imports 202 distinct pharmaceutical formulations. Showing top 15 by value. For full formulation-level data, contact TransData Nexus.
What Products Does I.e. Medica Inc. Import?
Top Products by Import Value
I.e. Medica Inc. Therapeutic Categories — 2 Specializations
I.e. Medica Inc. imports across 2 therapeutic categories, with Diabetes & Endocrine (97.0%), Respiratory (3.0%), representing the largest segments. The portfolio is concentrated — top 5 products = 100% of total imports.
Diabetes & Endocrine
2 products · 97.0% · $4.8M
Respiratory
1 products · 3.0% · $150.0K
Import Portfolio — Top 3 by Import Value
| # | Product | Category | Value | Ships | Share | Rk |
|---|---|---|---|---|---|---|
| 1 | Metformin | Diabetes & Endocrine | $4.2M | 84 | 0.9% | 11 |
| 2 | Glimepiride | Diabetes & Endocrine | $574.6K | 15 | 0.8% | 10 |
| 3 | Ipratropium | Respiratory | $150.0K | 3 | 0.3% | 16 |
I.e. Medica Inc. imports 3 pharmaceutical products across 2 categories into Philippines totaling $4.9M.
Key Metrics
Top Categories
Indian Suppliers
Related Trade Data
Need Detailed Data?
Shipment-level records, supplier connections & pricing for I.e. Medica Inc..
Request DemoI.e. Medica Inc. — Corporate Profile & Information
Company type, headquarters, distribution network, and industry role
1Company Overview
I.E. Medica Inc. is a Philippine-based pharmaceutical company established in 2007, specializing in the global sourcing, importation, registration, and supply of high-quality pharmaceutical products across various therapeutic areas. The company is headquartered at 5/F RFM Corporate Center, Pioneer St., Mandaluyong City, Philippines. I.E. Medica operates as a pharmaceutical importer and distributor, playing a pivotal role in enhancing healthcare accessibility and affordability in the Philippines. (iemedica.com.ph)
In 2019, I.E. Medica became part of AC Health, the healthcare subsidiary of Ayala Corporation, one of the country's leading conglomerates. This strategic partnership has bolstered I.E. Medica's supply chain capabilities, enabling the company to deliver a broader range of affordable medicines to Filipinos nationwide. (iemedica.com.ph)
2Distribution Network
I.E. Medica's distribution network is strategically located in the heart of Metro Manila, facilitating fast and efficient distribution across the Philippines. The company's warehouses adhere to good warehousing practices, ensuring that products are stored according to specifications. Temperature controls and cold-chain monitors are strictly managed to maintain product integrity. (iemedica.com.ph)
The distribution network spans major cities and regions, including Manila, Davao City, Makati, Cebu City, Iloilo City, Quezon City, Cagayan de Oro, Alaminos, and Canlaon City. This extensive coverage ensures that I.E. Medica's products are accessible to a wide economic spectrum of patients across the archipelago. (iemedica.com.ph)
3Industry Role
I.E. Medica Inc. serves as a pharmaceutical importer and distributor in the Philippines, sourcing and supplying a diverse range of pharmaceutical products to various marketing and distribution partners. The company is not a primary wholesaler, parallel importer, hospital supplier, or logistics intermediary; instead, it focuses on the importation and distribution of pharmaceutical products sourced from global manufacturers. (iemedica.com.ph)
Supplier Relationship Intelligence — I.e. Medica Inc.
Sourcing concentration, supply chain resilience, and strategic implications
1Sourcing Concentration Analysis
I.E. Medica's sourcing strategy exhibits a high degree of concentration, with a significant portion of its imports originating from a select group of Indian suppliers. The top five suppliers—Inventia Healthcare Limited, Ahlcon Parenterals (India) Limited, Alvita Pharma Private Limited, Pharose Remedies Limited, and Zim Laboratories Limited—account for a substantial share of the company's total import value. This concentration suggests a strategic choice to maintain strong, stable relationships with key suppliers, ensuring consistent product quality and supply reliability.
The shipment data indicates a stable relationship with these suppliers, as evidenced by the consistent number of shipments over the years. However, the lack of diversification in sourcing could pose risks related to supply chain disruptions, currency fluctuations, and geopolitical factors affecting trade relations. To mitigate these risks, I.E. Medica may consider diversifying its supplier base and exploring partnerships with additional manufacturers.
2Supply Chain Resilience
I.E. Medica's supply chain resilience is closely tied to its reliance on a limited number of Indian suppliers. While this focus allows for streamlined operations and strong supplier relationships, it also exposes the company to potential risks such as supply chain disruptions, currency fluctuations, and geopolitical tensions. The company's current sourcing strategy may benefit from diversification to enhance resilience.
I.E. Medica's suppliers are required to comply with current Good Manufacturing Practices (cGMP) and international standards such as UK-MHRA, US-FDA, WHO-GMP, and PIC/S. This adherence ensures that the imported pharmaceutical products meet stringent quality and safety requirements. (iemedica.com.ph)
3Strategic Implications
I.E. Medica's concentrated sourcing strategy positions the company to leverage strong, stable relationships with key suppliers, ensuring consistent product quality and supply reliability. This approach may provide a competitive edge in the Philippine market by offering a curated selection of high-quality pharmaceutical products.
For Indian exporters, the existing supplier relationships with I.E. Medica present an opportunity to introduce alternative products or formulations that complement the current portfolio. By aligning with I.E. Medica's quality standards and regulatory requirements, Indian exporters can establish partnerships that enhance their presence in the Philippine market.
Importing Pharmaceuticals into Philippines — Regulatory Framework
Regulatory authority, GMP requirements, import licensing for Philippines
1Regulatory Authority & Framework
The Food and Drug Administration (FDA) of the Philippines is the primary regulatory body overseeing the importation, registration, and distribution of pharmaceutical products in the country. The FDA ensures that all imported medicines meet the required safety, efficacy, and quality standards before they are made available to the public.
Key legislation governing pharmaceutical imports includes the Food, Drug, and Cosmetic Act (Republic Act No. 3720) and its subsequent amendments. These laws establish the regulatory framework for the importation, registration, and distribution of pharmaceutical products in the Philippines. The FDA's guidelines and circulars provide detailed procedures for compliance with these regulations.
The marketing authorization pathway for Indian generics involves obtaining product registration from the FDA. This process includes submitting comprehensive documentation, such as product dossiers, clinical trial data, and manufacturing information, to demonstrate the product's safety, efficacy, and quality. The FDA evaluates these submissions to grant marketing authorization for the product in the Philippine market.
2Import Licensing & GMP
Import licensing requirements for pharmaceutical products in the Philippines mandate that importers possess a valid License to Operate (LTO) and Drug Distributor-Importer authorization from the FDA. These licenses ensure that importers comply with regulatory standards and are authorized to import and distribute pharmaceutical products.
Good Manufacturing Practice (GMP) certification is recognized by the FDA to ensure that pharmaceutical products are consistently produced and controlled according to quality standards. Certifications from reputable authorities such as the European Medicines Agency (EMA), World Health Organization (WHO), and Pharmaceutical Inspection Co-operation Scheme (PIC/S) are acknowledged by the FDA. Importers must ensure that their suppliers hold valid GMP certifications from recognized bodies to meet regulatory requirements.
3Quality & Labeling
Imported pharmaceutical products are subject to batch testing and stability studies to ensure their safety, efficacy, and quality. The FDA may require these tests as part of the product registration process.
Labeling requirements stipulate that product labels be in English and/or Filipino, providing essential information such as the product's name, active ingredients, dosage form, strength, batch number, manufacturing and expiration dates, storage conditions, and the name and address of the manufacturer or distributor. Serialization mandates may apply to facilitate traceability and prevent counterfeit products.
4Recent Regulatory Changes
Between 2024 and 2026, the Philippines FDA has implemented several policy changes affecting pharmaceutical imports. These include updates to the product registration process, stricter enforcement of GMP compliance, and enhanced requirements for labeling and packaging to improve product traceability and safety. Importers and suppliers must stay informed about these regulatory changes to ensure continued compliance and uninterrupted market access.
I.e. Medica Inc. — Procurement Pattern Analysis
Product strategy, sourcing profile, and market positioning
1Product Strategy
I.E. Medica's product strategy focuses on importing pharmaceutical products across various therapeutic areas, with a notable emphasis on diabetes and endocrine treatments. The company's portfolio includes products like metformin and glimepiride, which are essential in managing diabetes. This focus aligns with the growing prevalence of diabetes in the Philippines and the increasing demand for effective treatments.
The inclusion of respiratory products, such as ipratropium, addresses the needs of patients with respiratory conditions, reflecting I.E. Medica's commitment to providing comprehensive healthcare solutions. By sourcing these products from reputable manufacturers, I.E. Medica ensures the availability of high-quality medicines to meet the diverse needs of the Filipino population.
2Sourcing Profile
I.E. Medica's sourcing strategy is centered on importing finished pharmaceutical formulations from India, focusing on generic drugs and unique fixed-dose combinations. The company partners with manufacturers that comply with current Good Manufacturing Practices (cGMP) and international standards such as UK-MHRA, US-FDA, WHO-GMP, and PIC/S. This approach ensures that the imported products meet stringent quality and safety requirements. (iemedica.com.ph)
India's well-established pharmaceutical industry, characterized by a large number of cGMP-compliant manufacturers, makes it a strategic sourcing destination for I.E. Medica. By leveraging India's manufacturing capabilities, I.E. Medica can offer a diverse range of high-quality pharmaceutical products to the Philippine market.
3Market Positioning
I.E. Medica serves the wholesale distribution segment of the Philippine pharmaceutical market. By importing and supplying a wide range of pharmaceutical products, the company supports various marketing and distribution partners, including retail pharmacies, hospitals, and government healthcare facilities. This positioning enables I.E. Medica to contribute significantly to the nation's healthcare system by ensuring the availability of essential medicines across the archipelago.
Seller's Guide — How to Become a Supplier to I.e. Medica Inc.
Opportunity assessment, qualifications, and practical approach strategy
1Opportunity Assessment
There is a realistic opportunity for new Indian suppliers to partner with I.E. Medica, especially if they
Frequently Asked Questions — I.e. Medica Inc.
What products does I.e. Medica Inc. import from India?
I.e. Medica Inc. imports 3 pharmaceutical products across 2 categories. Top imports: Metformin ($4.2M), Glimepiride ($574.6K), Ipratropium ($150.0K).
Who supplies pharmaceuticals to I.e. Medica Inc. from India?
I.e. Medica Inc. sources from 14 verified Indian suppliers. The primary supplier is Inventia Healthcare Limited (47.0% of imports, $6.3M).
What is I.e. Medica Inc.'s total pharmaceutical import value?
I.e. Medica Inc.'s total pharmaceutical import value from India is $4.9M, based on 102 verified shipments in Indian Customs (DGFT) data.
What therapeutic categories does I.e. Medica Inc. focus on?
I.e. Medica Inc. imports across 2 categories. The largest: Diabetes & Endocrine (97.0%), Respiratory (3.0%).
Get Full I.e. Medica Inc. Import Intelligence
Access shipment-level details, supplier connections, pricing data, and competitive analysis. TransData Nexus provides verified Indian Customs (DGFT) data trusted by pharmaceutical trade professionals worldwide.
Official References & Regulatory Resources
Verify import regulations and drug registration requirements with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Buyer Matching: I.e. Medica Inc. identified across shipments using consignee name normalization, aggregating 2 name variants.
- 2.Statistical Normalization: Shipment values are statistically normalized to ensure accurate market share representation.
- 3.Market Share: Calculated per product as I.e. Medica Inc.'s capped value divided by total Indian exports for that product.
- 4.Shipment Count: Based on 102 individual customs records matching I.e. Medica Inc..
- 5.Supplier Verification: I.e. Medica Inc. sources from 14 verified Indian suppliers across 202 formulations, confirmed from customs records.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
3 Products Tracked
2 therapeutic categories
Expert-Reviewed
By pharmaceutical trade specialists
Data Source & Methodology
Trade data sourced from Indian Customs (DGFT) export shipment records. Values represent FOB export value in USD. Profile aggregates 2 company name variants from customs records. For current shipment-level data, contact TransData Nexus.