Glenmark Pharmaceuticals Inc
Pharmaceutical Importer · United States · Immunosuppressants Focus · $2.3M Total Trade · DGFT Verified
Glenmark Pharmaceuticals Inc is a pharmaceutical importer based in United States with a total trade value of $2.3M across 5 products in 4 therapeutic categories. Based on 52 verified import shipments from Indian Customs (DGFT) records, the company actively imports across multiple product segments. Glenmark Pharmaceuticals Inc sources from 7 verified Indian suppliers, with Glenmark Pharmaceuticals Limited accounting for 97.9% of imports.
Glenmark Pharmaceuticals Inc — Import Portfolio & Supplier Network
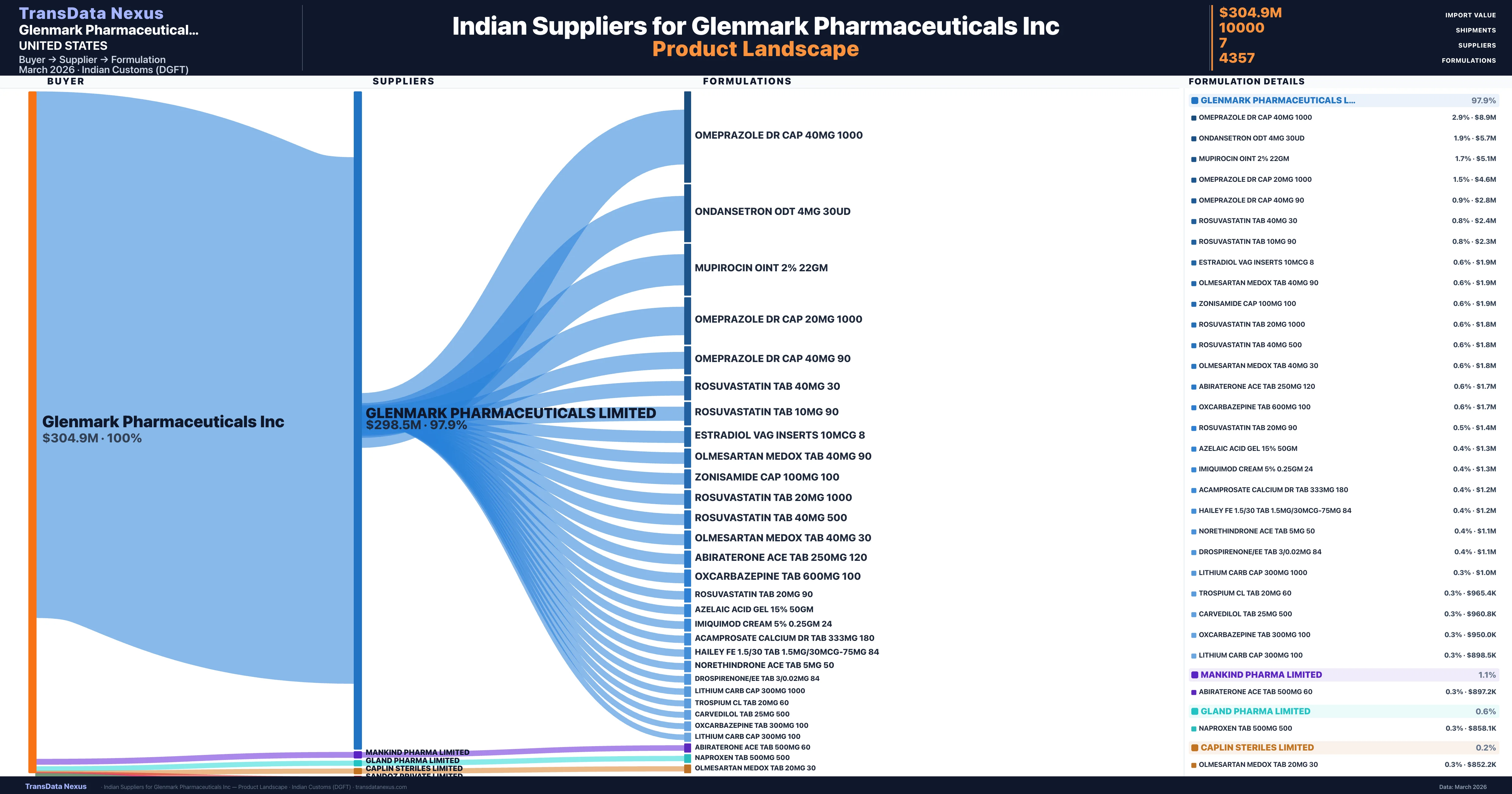
Who Are the Verified Indian Suppliers to Glenmark Pharmaceuticals Inc?
Customs-verified supplier relationships from Indian DGFT records
| Supplier | Value | Shipments | Share |
|---|---|---|---|
| Glenmark Pharmaceuticals Limited | $298.5M | 9,838 | 97.9% |
| Mankind Pharma Limited | $3.3M | 89 | 1.1% |
| Gland Pharma Limited | $2.0M | 42 | 0.6% |
| Caplin Steriles Limited | $515.1K | 12 | 0.2% |
| Sandoz Private Limited | $436.6K | 15 | 0.1% |
| Graviti Pharmaceuticals Private Limited | $131.5K | 3 | 0.0% |
| Immacule Lifesciences Private Limited | $50.0K | 1 | 0.0% |
Glenmark Pharmaceuticals Inc sources from 7 verified Indian suppliers across 4,357 distinct formulations. The sourcing is highly concentrated — Glenmark Pharmaceuticals Limited accounts for 97.9% of total imports, indicating a strategic single-source relationship.
What Formulations Does Glenmark Pharmaceuticals Inc Import?
| Formulation | Value | Ships |
|---|---|---|
| Omeprazole dr cap 40MG | $8.9M | 243 |
| Ondansetron odt 4MG | $5.7M | 220 |
| Mupirocin oint 2% 22GM | $5.1M | 274 |
| Omeprazole dr cap 20MG | $4.6M | 137 |
| Omeprazole dr cap 40MG | $2.8M | 79 |
| Rosuvastatin TAB 40MG | $2.4M | 56 |
| Rosuvastatin TAB 10MG | $2.3M | 55 |
| Estradiol vag inserts 10mcg | $1.9M | 54 |
| Olmesartan medox TAB 40MG | $1.9M | 40 |
| Zonisamide cap 100MG | $1.9M | 47 |
| Rosuvastatin TAB 20MG | $1.8M | 46 |
| Rosuvastatin TAB 40MG | $1.8M | 44 |
| Olmesartan medox TAB 40MG | $1.8M | 42 |
| Abiraterone ace TAB 250MG | $1.7M | 34 |
| Oxcarbazepine TAB 600MG | $1.7M | 34 |
Glenmark Pharmaceuticals Inc imports 4,357 distinct pharmaceutical formulations. Showing top 15 by value. For full formulation-level data, contact TransData Nexus.
What Products Does Glenmark Pharmaceuticals Inc Import?
Top Products by Import Value
Glenmark Pharmaceuticals Inc Therapeutic Categories — 4 Specializations
Glenmark Pharmaceuticals Inc imports across 4 therapeutic categories, with Immunosuppressants (64.7%), Cardiovascular (29.6%), Analgesics & Antipyretics (3.6%) representing the largest segments. The portfolio is concentrated — top 5 products = 100% of total imports.
Immunosuppressants
2 products · 64.7% · $1.5M
Cardiovascular
1 products · 29.6% · $666.5K
Analgesics & Antipyretics
1 products · 3.6% · $81.9K
Advanced Diabetes Medications
1 products · 2.0% · $45.2K
Import Portfolio — Top 5 by Import Value
| # | Product | Category | Value | Ships | Share | Rk |
|---|---|---|---|---|---|---|
| 1 | Tacrolimus | Immunosuppressants | $1.3M | 26 | 0.6% | 13 |
| 2 | Carvedilol | Cardiovascular | $666.5K | 18 | 0.7% | 20 |
| 3 | Sirolimus | Immunosuppressants | $156.9K | 4 | 1.1% | 8 |
| 4 | Indomethacin | Analgesics & Antipyretics | $81.9K | 2 | 0.9% | 9 |
| 5 | Saxagliptin | Advanced Diabetes Medications | $45.2K | 2 | 1.1% | 4 |
Glenmark Pharmaceuticals Inc imports 5 pharmaceutical products across 4 categories into United States totaling $2.3M.
Key Metrics
Top Categories
Indian Suppliers
Related Trade Data
Need Detailed Data?
Shipment-level records, supplier connections & pricing for Glenmark Pharmaceuticals Inc.
Request DemoGlenmark Pharmaceuticals Inc — Corporate Profile & Information
Company type, headquarters, distribution network, and industry role
1Company Overview
Glenmark Pharmaceuticals Inc., USA, is a subsidiary of Glenmark Pharmaceuticals Ltd., a global, research-driven pharmaceutical company headquartered in Mumbai, India. Established in 2003, Glenmark Pharmaceuticals Inc. serves as the North American arm, focusing on the development, manufacturing, and marketing of generic and over-the-counter (OTC) pharmaceutical products. The U.S. headquarters are located at 619 River Drive, Suite 400-A, Elmwood Park, NJ 07407.
In the United States, Glenmark Pharmaceuticals Inc. plays a significant role in the pharmaceutical distribution network, offering a diverse portfolio that includes generics, branded generics, and OTC products. The company has expanded its presence by acquiring approved Abbreviated New Drug Applications (ANDAs) from Wockhardt Limited, thereby enhancing its OTC portfolio. Additionally, Glenmark Therapeutics Inc., a wholly owned subsidiary, focuses on the branded dermatology segment, marking the company's strategic move into specialized therapeutic areas.
2Distribution Network
Glenmark Pharmaceuticals Inc. operates a robust distribution network within the United States, with its primary distribution center and warehouse situated in Mahwah, New Jersey. This strategic location facilitates efficient logistics and timely delivery of pharmaceutical products across the country. The company's manufacturing facility in Monroe, North Carolina, further supports its distribution capabilities, ensuring a steady supply of products to meet market demand.
3Industry Role
In the U.S. pharmaceutical supply chain, Glenmark Pharmaceuticals Inc. functions as a primary wholesaler and distributor. The company imports a range of pharmaceutical formulations from India, including immunosuppressants, cardiovascular drugs, and analgesics. By leveraging its manufacturing facilities and distribution network, Glenmark ensures the availability of these products to various stakeholders, including healthcare providers, pharmacies, and patients.
Supplier Relationship Intelligence — Glenmark Pharmaceuticals Inc
Sourcing concentration, supply chain resilience, and strategic implications
1Sourcing Concentration Analysis
Glenmark Pharmaceuticals Inc. imports finished pharmaceutical formulations from seven verified Indian suppliers, with Glenmark Pharmaceuticals Limited accounting for 97.9% of the total import value. This significant concentration indicates a strategic partnership aimed at ensuring product quality and supply chain efficiency. However, such dependency on a single supplier could pose risks related to supply disruptions or changes in supplier dynamics. The stability of this relationship is crucial for maintaining uninterrupted product availability in the U.S. market.
2Supply Chain Resilience
The resilience of Glenmark Pharmaceuticals Inc.'s supply chain is bolstered by its diversified sourcing from multiple Indian suppliers, reducing the impact of potential disruptions from any single source. The company's manufacturing facilities in the U.S. provide an additional layer of security, enabling it to mitigate supply chain challenges effectively. Ensuring that all suppliers comply with Good Manufacturing Practices (GMP) and maintaining robust quality control measures are essential for sustaining supply chain integrity.
3Strategic Implications
Glenmark Pharmaceuticals Inc.'s sourcing strategy positions it to offer a diverse range of pharmaceutical products in the U.S. market, catering to various therapeutic needs. The company's focus on high-demand categories such as immunosuppressants and cardiovascular drugs aligns with market requirements, enhancing its competitive edge. For Indian exporters, establishing partnerships with Glenmark could provide a pathway to enter the U.S. market, leveraging Glenmark's established distribution channels and regulatory expertise.
Importing Pharmaceuticals into United States — Regulatory Framework
Regulatory authority, GMP requirements, import licensing for United States
1Regulatory Authority & Framework
In the United States, the Food and Drug Administration (FDA) is the primary regulatory body overseeing the importation and marketing of pharmaceutical products. The Federal Food, Drug, and Cosmetic Act (FD&C Act) provides the legal framework governing these activities. Pharmaceutical imports must comply with FDA standards to ensure safety, efficacy, and quality. The FDA's Regulatory Procedures Manual outlines the processes for the importation of human drugs, including the examination and analysis of samples to verify compliance with applicable standards.
2Import Licensing & GMP
Importers of pharmaceutical products into the U.S. must ensure that their foreign suppliers are registered with the FDA and that their products are listed in the FDA's database. This registration process involves providing detailed information about the manufacturing facility and the products intended for import. Additionally, all imported drugs must be manufactured in accordance with Good Manufacturing Practices (GMP) as recognized by the FDA. While the FDA does not have a formal recognition process for foreign GMP certifications, it evaluates foreign manufacturing facilities during inspections to ensure compliance with U.S. standards.
3Quality & Labeling
Imported pharmaceutical products must adhere to FDA labeling requirements, which include accurate and informative labeling in English. The FDA reviews drug labeling at the time of import to verify compliance with regulations. Specific labeling requirements depend on the type of drug product, such as prescription drugs, over-the-counter drugs, or investigational drugs. Additionally, imported drugs are subject to batch testing and stability requirements to ensure their safety and efficacy.
4Recent Regulatory Changes
Between 2024 and 2026, the FDA has implemented several policy changes affecting the importation of pharmaceutical products from India. These changes include stricter enforcement of GMP compliance, enhanced scrutiny of drug labeling, and updated procedures for the importation of unapproved drugs intended for further processing or export. Importers must stay informed about these regulatory updates to ensure continued compliance and avoid potential disruptions in their supply chains.
Glenmark Pharmaceuticals Inc — Procurement Pattern Analysis
Product strategy, sourcing profile, and market positioning
1Product Strategy
Glenmark Pharmaceuticals Inc.'s focus on importing immunosuppressants, cardiovascular drugs, and analgesics aligns with the therapeutic needs of the U.S. market. The high demand for these products, driven by chronic health conditions prevalent in the population, presents a strategic opportunity for Glenmark to strengthen its market position. By sourcing these products from India, Glenmark leverages cost-effective manufacturing while ensuring access to essential medications for patients.
2Sourcing Profile
Glenmark Pharmaceuticals Inc. employs a sourcing strategy that emphasizes the importation of finished pharmaceutical formulations from India. This approach allows the company to offer a diverse product portfolio without the complexities associated with raw material sourcing. India's established pharmaceutical manufacturing capabilities and adherence to international quality standards make it a preferred sourcing destination for Glenmark.
3Market Positioning
Through its diverse product mix, Glenmark Pharmaceuticals Inc. serves multiple segments of the U.S. pharmaceutical market, including retail pharmacies, hospitals, and government tenders. The company's ability to provide both generic and branded products enables it to meet the needs of various healthcare providers and patients, positioning it as a versatile player in the distribution network.
Seller's Guide — How to Become a Supplier to Glenmark Pharmaceuticals Inc
Opportunity assessment, qualifications, and practical approach strategy
1Opportunity Assessment
There is a realistic opportunity for new Indian suppliers to partner with Glenmark Pharmaceuticals Inc., especially in therapeutic areas where Glenmark seeks to expand its portfolio. Identifying gaps in Glenmark's current sourcing, such as emerging therapeutic categories or innovative formulations, could present avenues for collaboration. Prospective suppliers should demonstrate compliance with FDA regulations and a commitment to quality to establish a successful partnership.
2Requirements & Qualifications
Indian exporters aiming to supply Glenmark Pharmaceuticals Inc. and the U.S. market must ensure that their manufacturing facilities are registered with the FDA and that their products are listed in the FDA's database. Compliance with GMP standards recognized by the FDA is essential. Additionally, products must meet FDA labeling requirements, including accurate and informative labeling in English. Maintaining up-to-date certifications and quality standards is crucial for establishing and sustaining business relationships.
3How to Approach
Indian exporters should initiate contact with Glenmark Pharmaceuticals Inc. by providing detailed information about their manufacturing capabilities, product portfolios, and compliance with FDA regulations. Participating in industry trade shows and networking events can facilitate introductions. Developing a comprehensive regulatory filing strategy, including obtaining necessary approvals and certifications, is vital. Establishing clear communication channels and understanding Glenmark's specific requirements will aid in building a successful partnership.
Frequently Asked Questions — Glenmark Pharmaceuticals Inc
What products does Glenmark Pharmaceuticals Inc import from India?
Glenmark Pharmaceuticals Inc imports 5 pharmaceutical products across 4 categories. Top imports: Tacrolimus ($1.3M), Carvedilol ($666.5K), Sirolimus ($156.9K), Indomethacin ($81.9K), Saxagliptin ($45.2K).
Who supplies pharmaceuticals to Glenmark Pharmaceuticals Inc from India?
Glenmark Pharmaceuticals Inc sources from 7 verified Indian suppliers. The primary supplier is Glenmark Pharmaceuticals Limited (97.9% of imports, $298.5M).
What is Glenmark Pharmaceuticals Inc's total pharmaceutical import value?
Glenmark Pharmaceuticals Inc's total pharmaceutical import value from India is $2.3M, based on 52 verified shipments in Indian Customs (DGFT) data.
What therapeutic categories does Glenmark Pharmaceuticals Inc focus on?
Glenmark Pharmaceuticals Inc imports across 4 categories. The largest: Immunosuppressants (64.7%), Cardiovascular (29.6%), Analgesics & Antipyretics (3.6%).
Get Full Glenmark Pharmaceuticals Inc Import Intelligence
Access shipment-level details, supplier connections, pricing data, and competitive analysis. TransData Nexus provides verified Indian Customs (DGFT) data trusted by pharmaceutical trade professionals worldwide.
Official References & Regulatory Resources
Verify import regulations and drug registration requirements with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Buyer Matching: Glenmark Pharmaceuticals Inc identified across shipments using consignee name normalization.
- 2.Statistical Normalization: Shipment values are statistically normalized to ensure accurate market share representation.
- 3.Market Share: Calculated per product as Glenmark Pharmaceuticals Inc's capped value divided by total Indian exports for that product.
- 4.Shipment Count: Based on 52 individual customs records matching Glenmark Pharmaceuticals Inc.
- 5.Supplier Verification: Glenmark Pharmaceuticals Inc sources from 7 verified Indian suppliers across 4,357 formulations, confirmed from customs records.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
5 Products Tracked
4 therapeutic categories
Expert-Reviewed
By pharmaceutical trade specialists
Data Source & Methodology
Trade data sourced from Indian Customs (DGFT) export shipment records. Values represent FOB export value in USD. For current shipment-level data, contact TransData Nexus.