Fresenius Kabi Canada Ltd
Pharmaceutical Importer · Canada · Cardiovascular Focus · $8.3M Total Trade · DGFT Verified
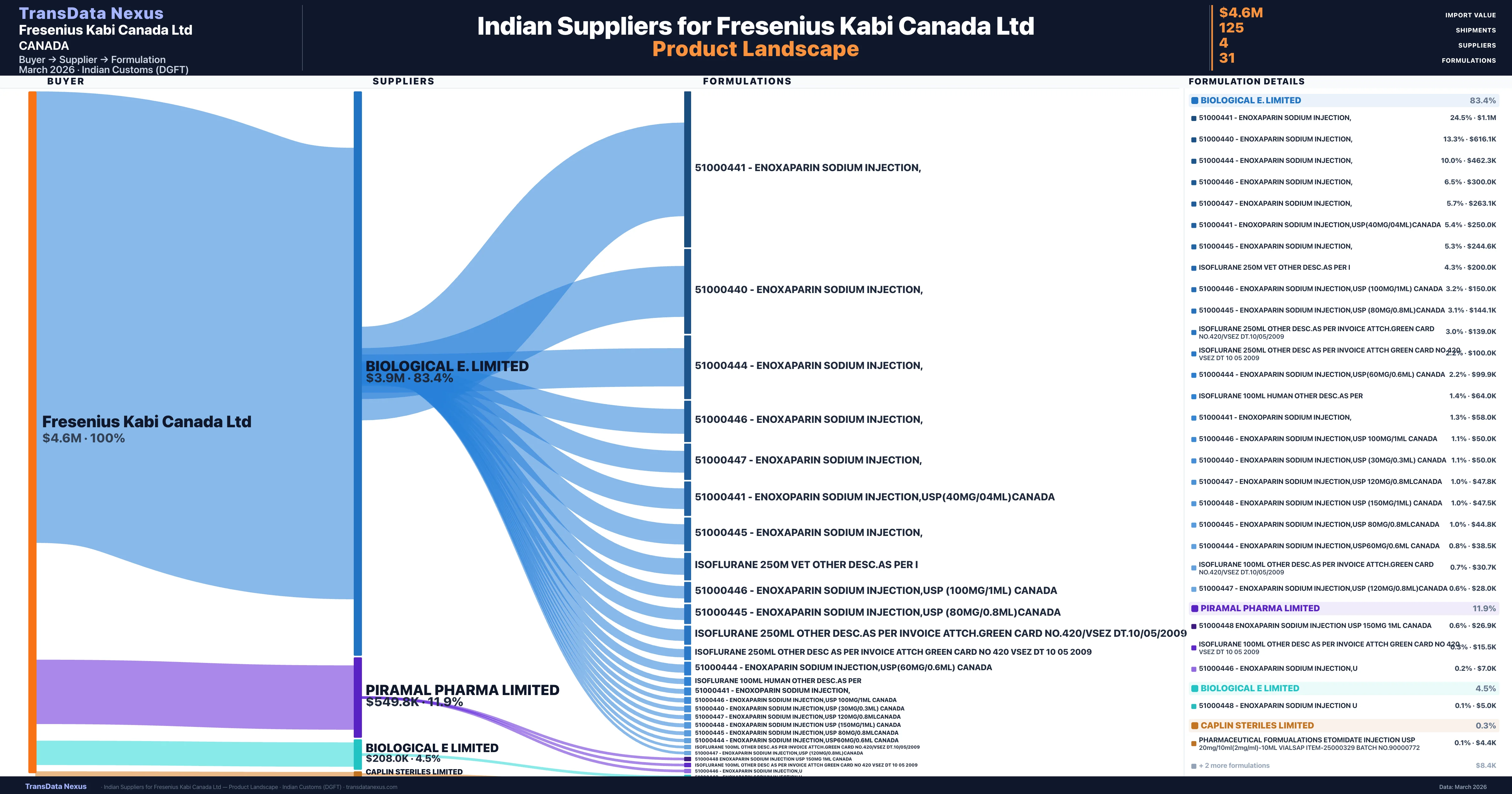
Fresenius Kabi Canada Ltd is a pharmaceutical importer based in Canada with a total trade value of $8.3M across 2 products in 2 therapeutic categories. Based on 167 verified import shipments from Indian Customs (DGFT) records, the company actively imports across multiple product segments. Fresenius Kabi Canada Ltd sources from 4 verified Indian suppliers, with Biological E. Limited accounting for 83.4% of imports.
Fresenius Kabi Canada Ltd — Import Portfolio & Supplier Network
Who Are the Verified Indian Suppliers to Fresenius Kabi Canada Ltd?
Customs-verified supplier relationships from Indian DGFT records
| Supplier | Value | Shipments | Share |
|---|---|---|---|
| Biological E. Limited | $3.9M | 96 | 83.4% |
| Piramal Pharma Limited | $549.8K | 21 | 11.9% |
| Biological E Limited | $208.0K | 5 | 4.5% |
| Caplin Steriles Limited | $12.7K | 3 | 0.3% |
Fresenius Kabi Canada Ltd sources from 4 verified Indian suppliers across 31 distinct formulations. The sourcing is highly concentrated — Biological E. Limited accounts for 83.4% of total imports, indicating a strategic single-source relationship.
What Formulations Does Fresenius Kabi Canada Ltd Import?
| Formulation | Value | Ships |
|---|---|---|
| 51000441 - enoxaparin sodium injection | $1.1M | 24 |
| 51000440 - enoxaparin sodium injection | $616.1K | 14 |
| 51000444 - enoxaparin sodium injection | $462.3K | 14 |
| 51000446 - enoxaparin sodium injection | $300.0K | 6 |
| 51000447 - enoxaparin sodium injection | $263.1K | 8 |
| 51000441 - enoxoparin sodium injection,USP(40MG/04ML | $250.0K | 5 |
| 51000445 - enoxaparin sodium injection | $244.6K | 8 |
| Isoflurane 250m vet other desc.as per | $200.0K | 4 |
| 51000446 - enoxaparin sodium injection,USP (100MG/1ML) | $150.0K | 3 |
| 51000445 - enoxaparin sodium injection,USP (80MG/0.8ML | $144.1K | 3 |
| Isoflurane 250ML other desc.as per invoice attch.green card no.420/vsez | $139.0K | 4 |
| Isoflurane 250ML other desc as per invoice attch green card no 420 vsez dt 10 05 | $100.0K | 2 |
| 51000444 - enoxaparin sodium injection,USP(60MG/0.6ML) | $99.9K | 2 |
| Isoflurane 100ML human other desc.as | $64.0K | 7 |
| 51000441 - enoxoparin sodium injection | $58.0K | 2 |
Fresenius Kabi Canada Ltd imports 31 distinct pharmaceutical formulations. Showing top 15 by value. For full formulation-level data, contact TransData Nexus.
What Products Does Fresenius Kabi Canada Ltd Import?
Top Products by Import Value
Fresenius Kabi Canada Ltd Therapeutic Categories — 2 Specializations
Fresenius Kabi Canada Ltd imports across 2 therapeutic categories, with Cardiovascular (52.1%), Nutritional Supplements (47.9%), representing the largest segments. The portfolio is concentrated — top 5 products = 100% of total imports.
Cardiovascular
1 products · 52.1% · $4.3M
Nutritional Supplements
1 products · 47.9% · $4.0M
Import Portfolio — Top 2 by Import Value
| # | Product | Category | Value | Ships | Share | Rk |
|---|---|---|---|---|---|---|
| 1 | Enoxaparin | Cardiovascular | $4.3M | 87 | 2.1% | 8 |
| 2 | Sodium | Nutritional Supplements | $4.0M | 80 | 1.1% | 15 |
Fresenius Kabi Canada Ltd imports 2 pharmaceutical products across 2 categories into Canada totaling $8.3M.
Key Metrics
Top Categories
Indian Suppliers
Related Trade Data
Need Detailed Data?
Shipment-level records, supplier connections & pricing for Fresenius Kabi Canada Ltd.
Request DemoFresenius Kabi Canada Ltd — Corporate Profile & Information
Company type, headquarters, distribution network, and industry role
1Company Overview
Fresenius Kabi Canada Ltd is a Canadian subsidiary of the global healthcare company Fresenius Kabi, specializing in lifesaving medicines and technologies for infusion, transfusion, and clinical nutrition. The company is headquartered in Toronto, Ontario, and is dedicated to supplying high-quality, cost-effective pharmaceutical products and services to Canadians. Their extensive product portfolio includes intravenous specialty and generic medicines, transfusion technologies, infusion therapies, parenteral nutrition, biosimilars, complex formulations, and related medical devices.
Fresenius Kabi Canada plays a significant role in the Canadian pharmaceutical distribution network by providing essential medicines and technologies to healthcare providers across the country. Their commitment to improving patient care is evident through their diverse product offerings and continuous investment in advanced manufacturing and distribution facilities. For instance, in October 2018, the company opened a state-of-the-art compounding center in Mississauga, Ontario, to meet the increasing demand for large batches of compounded medications in Canadian hospitals.
2Distribution Network
Fresenius Kabi Canada Ltd operates a robust distribution network to ensure the timely delivery of its products across Canada. The company's Canadian headquarters are located at 165 Galaxy Blvd. Suite 100, Toronto, Ontario M9W 0C8. Additionally, in October 2018, Fresenius Kabi opened a new 35,000-square-foot compounding center in Mississauga, Ontario, to enhance its manufacturing capabilities and meet the growing demand for compounded medications in Canadian hospitals.
While specific details about other warehouse locations and logistics capabilities are not publicly disclosed, Fresenius Kabi's global presence and integrated supply chain network suggest a well-established infrastructure to support its operations in Canada. The company's commitment to investing in North American manufacturing and technology further underscores its dedication to meeting the needs of the Canadian market.
3Industry Role
Fresenius Kabi Canada Ltd serves as a pharmaceutical importer and distributor within Canada's healthcare sector. The company imports finished pharmaceutical formulations from various suppliers, including those in India, to meet the needs of Canadian healthcare providers. Their product portfolio encompasses intravenous specialty and generic medicines, transfusion technologies, infusion therapies, parenteral nutrition, biosimilars, complex formulations, and related medical devices.
By importing and distributing these products, Fresenius Kabi Canada plays a crucial role in ensuring the availability of essential medicines and technologies for critically and chronically ill patients across the country. Their operations contribute to the overall efficiency and effectiveness of Canada's pharmaceutical supply chain, supporting healthcare providers in delivering quality care to patients.
Supplier Relationship Intelligence — Fresenius Kabi Canada Ltd
Sourcing concentration, supply chain resilience, and strategic implications
1Sourcing Concentration Analysis
Fresenius Kabi Canada Ltd's sourcing strategy reveals a significant concentration in its supplier base, particularly from India. The company has imported a total of $8.3 million USD worth of finished pharmaceutical formulations from India across 167 shipments, encompassing 31 unique formulations. The top two products imported are Enoxaparin ($4.3 million, 2.1% share) and Sodium ($4.0 million, 1.1% share), indicating a heavy reliance on these specific products.
This sourcing pattern suggests a strategic choice to focus on high-demand, high-value products that align with the company's therapeutic focus areas. However, the heavy reliance on a limited number of products and suppliers may pose risks related to supply chain disruptions, price fluctuations, and regulatory changes in the supplier's country. The stability of the relationship with Indian suppliers, particularly BIOLOGICAL E. LIMITED, which accounts for 83.4% of the shipments, is crucial for maintaining a consistent supply of these key products.
2Supply Chain Resilience
Fresenius Kabi Canada Ltd's supply chain resilience is influenced by its sourcing strategy, which heavily depends on a limited number of suppliers and products. The company's reliance on BIOLOGICAL E. LIMITED for a significant portion of its imports indicates a concentrated supply chain, which may be vulnerable to disruptions such as production issues, regulatory changes, or geopolitical events in India.
To enhance supply chain resilience, it would be prudent for Fresenius Kabi Canada to diversify its supplier base and product portfolio. Engaging with additional suppliers and incorporating a broader range of formulations can mitigate risks associated with over-reliance on specific sources. Additionally, establishing contingency plans and maintaining open communication channels with suppliers can further strengthen the supply chain's robustness.
3Strategic Implications
Fresenius Kabi Canada Ltd's current sourcing pattern, characterized by a concentrated supplier base and product portfolio, has several strategic implications. While focusing on high-demand products can lead to cost efficiencies and streamlined operations, the heavy reliance on a limited number of suppliers and products may expose the company to supply chain risks.
For Indian exporters seeking to become alternative suppliers, this presents an opportunity to diversify their customer base and reduce dependency on a single client. By offering a broader range of products or formulations not currently sourced by Fresenius Kabi Canada, Indian exporters can position themselves as valuable partners in enhancing the company's supply chain resilience.
Importing Pharmaceuticals into Canada — Regulatory Framework
Regulatory authority, GMP requirements, import licensing for Canada
1Regulatory Authority & Framework
In Canada, the primary regulatory authority overseeing pharmaceutical imports is Health Canada, which is responsible for ensuring that all pharmaceutical products meet the necessary safety, efficacy, and quality standards before they are authorized for sale. The Food and Drugs Act and its associated regulations govern the importation, distribution, and sale of pharmaceutical products in Canada.
For Indian generics to be marketed in Canada, they must undergo a rigorous evaluation process by Health Canada. This process includes the submission of a New Drug Submission (NDS) or Abbreviated New Drug Submission (ANDS), depending on the product's nature. The evaluation assesses the product's safety, efficacy, quality, and compliance with Canadian standards. Once approved, the product is granted a Notice of Compliance (NOC), allowing it to be marketed in Canada.
2Import Licensing & GMP
Import licensing requirements in Canada are managed by Health Canada. Companies importing pharmaceutical products must obtain a Drug Establishment License (DEL), which authorizes them to import, distribute, and sell drugs in Canada. This license ensures that the importer complies with all regulatory requirements and maintains appropriate records.
Good Manufacturing Practice (GMP) certification is essential for pharmaceutical products imported into Canada. Health Canada recognizes GMP certifications from various regulatory bodies, including the European Union (EU) GMP, World Health Organization (WHO) GMP, and Pharmaceutical Inspection Co-operation Scheme (PIC/S). Importers must ensure that their suppliers hold valid GMP certifications from recognized authorities to guarantee product quality and safety.
3Quality & Labeling
Imported pharmaceutical products must undergo batch testing to confirm their quality, safety, and efficacy. Health Canada may require samples to be submitted for analysis before granting market authorization. Additionally, products must meet stability requirements to ensure they remain effective throughout their shelf life.
Labeling requirements in Canada stipulate that all information be provided in both English and French. Labels must include essential details such as product name, dosage form, strength, route of administration, storage conditions, and expiry date. Serialization mandates may apply to certain products to enhance traceability and prevent counterfeit drugs from entering the market.
4Recent Regulatory Changes
Between 2024 and 2026, several policy changes have impacted the importation of Indian pharmaceutical products into Canada. Health Canada has implemented stricter guidelines for GMP certification recognition, now requiring that all foreign manufacturers hold certifications from PIC/S member countries. This change aims to enhance product quality and safety standards.
Additionally, Health Canada has introduced more rigorous batch testing protocols, necessitating that all imported pharmaceutical products undergo comprehensive stability and efficacy testing before market authorization. These measures are designed to ensure that imported drugs meet Canadian standards and are safe for patient use.
Fresenius Kabi Canada Ltd — Procurement Pattern Analysis
Product strategy, sourcing profile, and market positioning
1Product Strategy
Fresenius Kabi Canada Ltd's product strategy focuses on two primary therapeutic areas: cardiovascular and nutritional supplements. The company's imports from India align with these areas, with Enoxaparin and Sodium being the top two products sourced. This strategic focus indicates a commitment to addressing critical health conditions prevalent in the Canadian population.
The demand for these products is driven by the high incidence of cardiovascular diseases and the need for nutritional support in critically ill patients. By concentrating on these therapeutic areas, Fresenius Kabi Canada aims to meet the specific healthcare needs of Canadians and enhance patient outcomes.
2Sourcing Profile
Fresenius Kabi Canada Ltd's sourcing strategy emphasizes the procurement of generic drugs, particularly in the cardiovascular and nutritional supplement categories. The company imports finished pharmaceutical formulations from India, focusing on products that align with its therapeutic areas of interest. This approach allows Fresenius Kabi Canada to offer cost-effective solutions while maintaining product quality.
India's well-established pharmaceutical manufacturing industry, with its adherence to recognized GMP standards, makes it
Frequently Asked Questions — Fresenius Kabi Canada Ltd
What products does Fresenius Kabi Canada Ltd import from India?
Fresenius Kabi Canada Ltd imports 2 pharmaceutical products across 2 categories. Top imports: Enoxaparin ($4.3M), Sodium ($4.0M).
Who supplies pharmaceuticals to Fresenius Kabi Canada Ltd from India?
Fresenius Kabi Canada Ltd sources from 4 verified Indian suppliers. The primary supplier is Biological E. Limited (83.4% of imports, $3.9M).
What is Fresenius Kabi Canada Ltd's total pharmaceutical import value?
Fresenius Kabi Canada Ltd's total pharmaceutical import value from India is $8.3M, based on 167 verified shipments in Indian Customs (DGFT) data.
What therapeutic categories does Fresenius Kabi Canada Ltd focus on?
Fresenius Kabi Canada Ltd imports across 2 categories. The largest: Cardiovascular (52.1%), Nutritional Supplements (47.9%).
Get Full Fresenius Kabi Canada Ltd Import Intelligence
Access shipment-level details, supplier connections, pricing data, and competitive analysis. TransData Nexus provides verified Indian Customs (DGFT) data trusted by pharmaceutical trade professionals worldwide.
Official References & Regulatory Resources
Verify import regulations and drug registration requirements with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Buyer Matching: Fresenius Kabi Canada Ltd identified across shipments using consignee name normalization.
- 2.Statistical Normalization: Shipment values are statistically normalized to ensure accurate market share representation.
- 3.Market Share: Calculated per product as Fresenius Kabi Canada Ltd's capped value divided by total Indian exports for that product.
- 4.Shipment Count: Based on 167 individual customs records matching Fresenius Kabi Canada Ltd.
- 5.Supplier Verification: Fresenius Kabi Canada Ltd sources from 4 verified Indian suppliers across 31 formulations, confirmed from customs records.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
2 Products Tracked
2 therapeutic categories
Expert-Reviewed
By pharmaceutical trade specialists
Data Source & Methodology
Trade data sourced from Indian Customs (DGFT) export shipment records. Values represent FOB export value in USD. For current shipment-level data, contact TransData Nexus.