Eurimex Pharma
Pharmaceutical Importer · France · Antimalarial & Antiparasitic Focus · $1.5M Total Trade · DGFT Verified
Eurimex Pharma is a pharmaceutical importer based in France with a total trade value of $1.5M across 5 products in 4 therapeutic categories. Based on 64 verified import shipments from Indian Customs (DGFT) records, the company actively imports across multiple product segments. Eurimex Pharma sources from 17 verified Indian suppliers, with Cadila Pharmaceuticals Limited accounting for 33.0% of imports.
Eurimex Pharma — Import Portfolio & Supplier Network
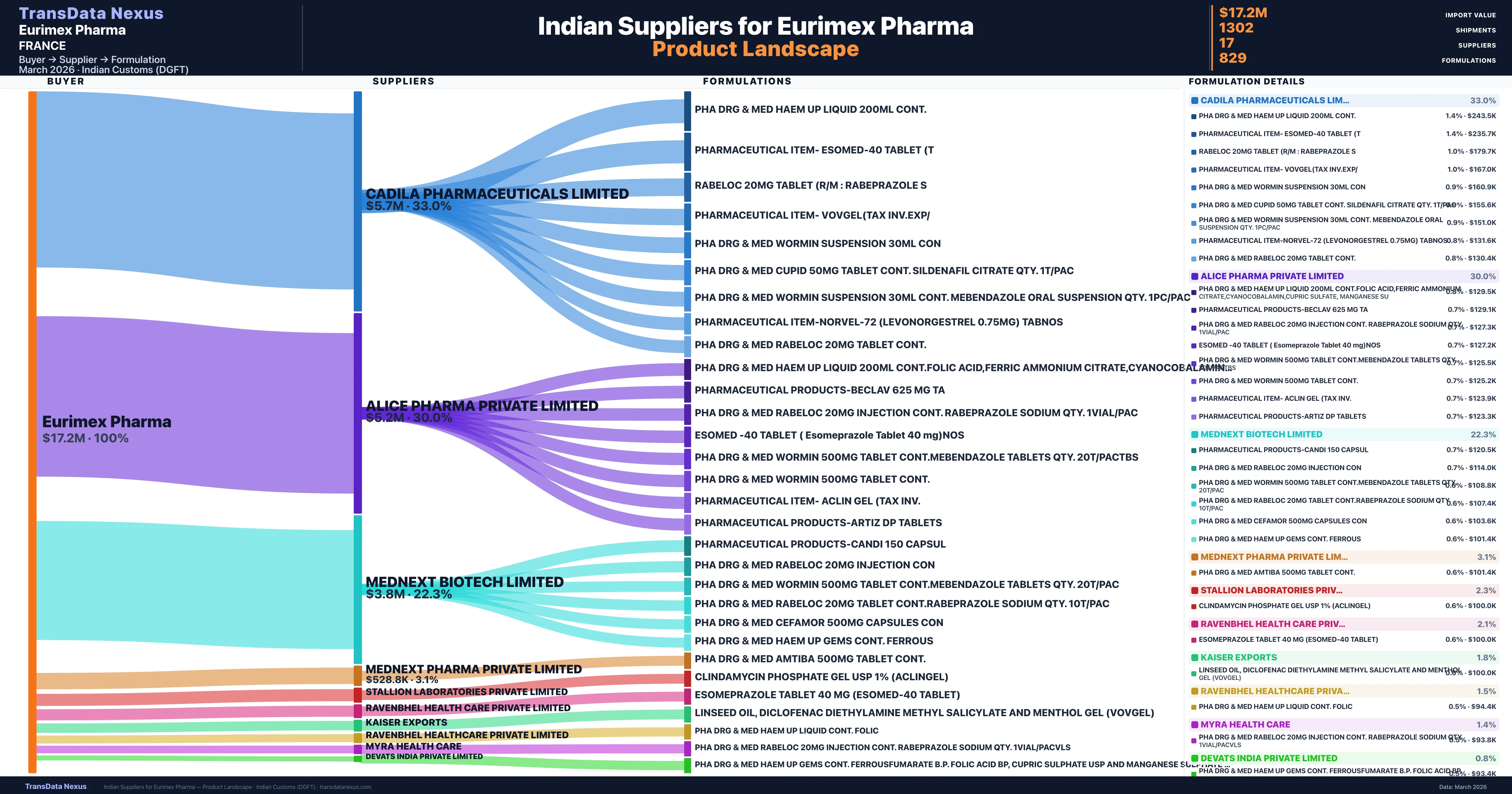
Who Are the Verified Indian Suppliers to Eurimex Pharma?
Customs-verified supplier relationships from Indian DGFT records
| Supplier | Value | Shipments | Share |
|---|---|---|---|
| Cadila Pharmaceuticals Limited | $5.7M | 312 | 33.0% |
| Alice Pharma Private Limited | $5.2M | 480 | 30.0% |
| Mednext Biotech Limited | $3.8M | 214 | 22.3% |
| Mednext Pharma Private Limited | $528.8K | 27 | 3.1% |
| Stallion Laboratories Private Limited | $389.6K | 75 | 2.3% |
| Ravenbhel Health Care Private Limited | $357.6K | 36 | 2.1% |
| Kaiser Exports | $306.5K | 56 | 1.8% |
| Ravenbhel Healthcare Private Limited | $257.4K | 15 | 1.5% |
| Myra Health Care | $244.7K | 20 | 1.4% |
| Devats India Private Limited | $134.3K | 5 | 0.8% |
| Careth Corporation | $110.5K | 15 | 0.6% |
| Ciel Pharma Private Limited | $83.3K | 26 | 0.5% |
| London United Medimpex Private Limited | $44.6K | 10 | 0.3% |
| Bliss Gvs Pharma Limited | $33.2K | 1 | 0.2% |
| Lob International | $22.0K | 2 | 0.1% |
| Psa Chemicals And Pharmaceuticals Private Limited | $16.9K | 6 | 0.1% |
| Kausikh Therapeutics Private Limited | $5.3K | 2 | 0.0% |
Eurimex Pharma sources from 17 verified Indian suppliers across 829 distinct formulations. The supply base is diversified across 17 suppliers, reducing single-source dependency risk.
What Formulations Does Eurimex Pharma Import?
| Formulation | Value | Ships |
|---|---|---|
| Pha drg & med haem up liquid 200ML cont. | $243.5K | 6 |
| Pharmaceutical item- esomed-40 tablet | $235.7K | 5 |
| Rabeloc 20MG tablet (r/m : rabeprazole | $179.7K | 5 |
| Pharmaceutical item- vovgel(tax | $167.0K | 4 |
| Pha drg & med wormin suspension 30ML | $160.9K | 6 |
| Pha drg & med cupid 50MG tablet cont. sildenafil citrate qty. | $155.6K | 7 |
| Pha drg & med wormin suspension 30ML cont. mebendazole oral suspension qty. | $151.0K | 4 |
| Pharmaceutical item-norvel-72 (levonorgestrel 0.75MG) | $131.6K | 3 |
| Pha drg & med rabeloc 20MG tablet cont. | $130.4K | 5 |
| Pha drg & med haem up liquid 200ML cont.folic acid,ferric ammonium citrate,cyanocobalamin,cupric sulfate, manganese | $129.5K | 3 |
| Pharmaceutical products-beclav 625 MG | $129.1K | 7 |
| Pha drg & med rabeloc 20MG injection cont. rabeprazole sodium qty. | $127.3K | 3 |
| Esomed -40 tablet ( esomeprazole tablet 40 MG | $127.2K | 3 |
| Pha drg & med wormin 500MG tablet cont.mebendazole tablets qty. | $125.5K | 6 |
| Pha drg & med wormin 500MG tablet cont. | $125.2K | 4 |
Eurimex Pharma imports 829 distinct pharmaceutical formulations. Showing top 15 by value. For full formulation-level data, contact TransData Nexus.
What Products Does Eurimex Pharma Import?
Top Products by Import Value
Eurimex Pharma Therapeutic Categories — 4 Specializations
Eurimex Pharma imports across 4 therapeutic categories, with Antimalarial & Antiparasitic (45.8%), Nutritional Supplements (33.5%), Advanced Antibiotics (12.3%) representing the largest segments. The portfolio is concentrated — top 5 products = 100% of total imports.
Antimalarial & Antiparasitic
1 products · 45.8% · $694.5K
Nutritional Supplements
2 products · 33.5% · $507.1K
Advanced Antibiotics
1 products · 12.3% · $187.1K
Gastrointestinal
1 products · 8.3% · $126.2K
Import Portfolio — Top 5 by Import Value
| # | Product | Category | Value | Ships | Share | Rk |
|---|---|---|---|---|---|---|
| 1 | Mebendazole | Antimalarial & Antiparasitic | $694.5K | 25 | 3.6% | 3 |
| 2 | Folic | Nutritional Supplements | $465.3K | 20 | 0.8% | 14 |
| 3 | Secnidazole | Advanced Antibiotics | $187.1K | 10 | 0.6% | 12 |
| 4 | Ranitidine | Gastrointestinal | $126.2K | 6 | 1.0% | 16 |
| 5 | Collagen | Nutritional Supplements | $41.8K | 3 | 1.8% | 8 |
Eurimex Pharma imports 5 pharmaceutical products across 4 categories into France totaling $1.5M.
Key Metrics
Top Categories
Indian Suppliers
Related Trade Data
Need Detailed Data?
Shipment-level records, supplier connections & pricing for Eurimex Pharma.
Request DemoEurimex Pharma — Corporate Profile & Information
Company type, headquarters, distribution network, and industry role
1Company Overview
Eurimex Pharma is an independent French pharmaceutical wholesaler and distributor, specializing in the export of human medicines. Established in 1994, the company is headquartered in Vitré, Brittany, France. It operates as a Société par Actions Simplifiée (SAS) with a capital of €161,952. Eurimex Pharma is not affiliated with any international laboratory subsidiaries or purchasing offices, allowing it to serve both private and public markets without bias.
The company offers comprehensive services, including distribution, storage, and registration of pharmaceutical products. Its primary markets include French overseas territories, such as Guyane, Guadeloupe, La Réunion, Martinique, Madagascar, and Djibouti, as well as French-speaking West Africa. Eurimex Pharma's distribution center in Vitré spans 1,050 m², with a storage capacity of 900 pallets, facilitating efficient logistics and supply chain management. (eurimex-pharma.fr)
2Distribution Network
Eurimex Pharma's distribution network is strategically located to serve both domestic and international markets. The main warehouse in Vitré, situated on the Rennes-Paris highway, is approximately three hours from Paris and Rouen, ensuring timely deliveries across France. The facility covers 1,050 m² and can accommodate 900 pallets, supporting efficient logistics operations. Additionally, the company has expanded its operations to Étrelles, investing €1.7 million in a new building to meet the growing demand for storage and distribution services.
3Industry Role
Eurimex Pharma functions as a pharmaceutical wholesaler and distributor, focusing on the export of human medicines. It is not a parallel importer, hospital supplier, or logistics intermediary. The company's independence from international laboratory subsidiaries or purchasing offices enables it to serve both private and public markets without bias, offering a diverse range of pharmaceutical products to various clients. (eurimex-pharma.fr)
Supplier Relationship Intelligence — Eurimex Pharma
Sourcing concentration, supply chain resilience, and strategic implications
1Sourcing Concentration Analysis
Eurimex Pharma's sourcing strategy is characterized by a high concentration of imports from a select group of Indian pharmaceutical suppliers. The top five suppliers—Cadila Pharmaceuticals Limited, Alice Pharma Private Limited, Mednext Biotech Limited, Mednext Pharma Private Limited, and Stallion Laboratories Private Limited—account for a significant portion of the company's total import value. This concentrated sourcing approach may present risks related to supply chain disruptions, such as production delays or regulatory changes affecting these suppliers. However, the company's diversified product portfolio, with 829 unique formulations, suggests a strategic choice to mitigate potential risks by not relying on a single supplier or product line.
2Supply Chain Resilience
Eurimex Pharma's supply chain resilience is supported by its diversified product portfolio, encompassing 829 unique formulations across various therapeutic categories. This diversity allows the company to adapt to changes in demand and mitigate risks associated with supply chain disruptions. The company's strategic sourcing from multiple Indian suppliers further enhances its resilience, providing alternative options in case of issues with any single supplier. Additionally, the company's investment in expanding its storage and distribution facilities demonstrates a commitment to maintaining a robust and adaptable supply chain.
3Strategic Implications
Eurimex Pharma's sourcing pattern, characterized by a high concentration of imports from a select group of Indian suppliers, positions the company to leverage strong relationships and potentially favorable terms with these suppliers. For Indian exporters, this presents an opportunity to strengthen partnerships with Eurimex Pharma by ensuring consistent product quality, reliable delivery schedules, and compliance with regulatory standards. By aligning with Eurimex Pharma's sourcing strategy, Indian exporters can enhance their market presence in France and French-speaking West Africa, tapping into a network that serves both private and public sectors.
Importing Pharmaceuticals into France — Regulatory Framework
Regulatory authority, GMP requirements, import licensing for France
1Regulatory Authority & Framework
In France, the primary regulatory authority overseeing pharmaceutical imports is the French National Agency for Medicines and Health Products Safety (ANSM). The key legislation governing pharmaceutical imports includes the Public Health Code (Code de la santé publique), which outlines the requirements for marketing authorization, importation, and distribution of medicinal products. The marketing authorization pathway for Indian generics involves obtaining a Marketing Authorization (AMM) from the ANSM, which requires demonstrating the product's quality, safety, and efficacy. Additionally, compliance with Good Manufacturing Practice (GMP) standards, as recognized by the European Medicines Agency (EMA), is mandatory for all imported pharmaceutical products.
2Import Licensing & GMP
Import licensing requirements for pharmaceutical products in France include obtaining a wholesale distribution authorization from the ANSM. This authorization ensures that the importer adheres to the necessary standards for storage, handling, and distribution of medicinal products. GMP certificates recognized by the European Union, such as EU GMP, WHO GMP, and PIC/S, are essential for ensuring product quality and safety. Eurimex Pharma, as a pharmaceutical wholesaler and distributor, must ensure that all imported products comply with these GMP standards to maintain regulatory compliance and product integrity.
3Quality & Labeling
Imported pharmaceutical products must undergo batch testing to verify their quality, safety, and efficacy before being distributed in France. Stability requirements ensure that products maintain their intended quality throughout their shelf life. Labeling must be in French and include all necessary information, such as dosage instructions, side effects, and storage conditions, to ensure clear communication with healthcare professionals and patients. Serialization mandates are in place to track and trace pharmaceutical products throughout the supply chain, enhancing transparency and preventing counterfeit products from entering the market.
4Recent Regulatory Changes
Between 2024 and 2026, France implemented several regulatory changes affecting pharmaceutical imports, including stricter requirements for GMP compliance and enhanced scrutiny of import licenses. These changes aim to ensure higher standards of product quality and safety. Additionally, the ANSM introduced more rigorous batch testing protocols and updated labeling requirements to improve transparency and patient safety. Eurimex Pharma must adapt to these regulatory changes by ensuring that all imported products meet the new standards and by maintaining up-to-date knowledge of regulatory developments.
Eurimex Pharma — Procurement Pattern Analysis
Product strategy, sourcing profile, and market positioning
1Product Strategy
Eurimex Pharma's product strategy focuses on therapeutic areas with high demand, including antimalarial and antiparasitic agents, nutritional supplements, and advanced antibiotics. The company's imports of mebendazole, secnidazole, and ranitidine align with these therapeutic categories, indicating a strategic focus on addressing prevalent health concerns in its target markets. The significant import value of mebendazole and secnidazole suggests a strong market demand for these products, while the inclusion of ranitidine reflects a commitment to providing comprehensive therapeutic options.
2Sourcing Profile
Eurimex Pharma's sourcing strategy emphasizes the importation of finished pharmaceutical formulations from India, focusing on generic drugs. This approach allows the company to offer cost-effective alternatives to branded medications, making essential treatments more accessible in its target markets. The company's preference for finished formulations over raw active pharmaceutical ingredients (APIs) indicates a focus on ready-to-market products that meet regulatory standards and are prepared for immediate distribution.
3Market Positioning
Eurimex Pharma serves a diverse segment of the French pharmaceutical market, including retail pharmacies, hospitals, government tenders, and wholesale distribution. Its extensive product portfolio, encompassing 829 unique formulations across various therapeutic categories, enables the company to meet the needs of different healthcare providers and institutions. The company's focus on both private and public markets, including French overseas territories and French-speaking West Africa, demonstrates its broad market reach and adaptability to various healthcare environments.
Seller's Guide — How to Become a Supplier to Eurimex Pharma
Opportunity assessment, qualifications, and practical approach strategy
1Opportunity Assessment
There is a realistic opportunity for new Indian suppliers to partner with Eurimex Pharma, particularly those offering high-quality generic formulations that align with the company's therapeutic focus areas. Gaps in Eurimex Pharma's current sourcing may include specific formulations or dosage forms not currently imported from India. Indian exporters can explore these opportunities by ensuring compliance with French regulatory standards, maintaining consistent product quality, and demonstrating the ability to meet Eurimex Pharma's logistical requirements.
2Requirements & Qualifications
Indian exporters seeking to supply Eurimex Pharma must ensure that their products comply with French regulatory standards, including obtaining the necessary marketing authorizations from the ANSM. Products must adhere to GMP standards recognized by the European Union, such as EU GMP, WHO GMP, or PIC/S. Additionally, exporters must provide labeling in French that meets all regulatory requirements and ensure that their products undergo batch testing and stability assessments as mandated by French authorities.
3How to Approach
Indian exporters can approach Eurimex Pharma by initiating direct communication to express interest in partnership opportunities. Participating in relevant pharmaceutical trade fairs and industry events can also facilitate introductions. It is essential to understand and comply with the regulatory filing strategy, including obtaining the necessary marketing authorizations and ensuring GMP compliance. Establishing a clear timeline for product registration and distribution is crucial to align with Eurimex Pharma's operational schedules and market demands.
Frequently Asked Questions — Eurimex Pharma
What products does Eurimex Pharma import from India?
Eurimex Pharma imports 5 pharmaceutical products across 4 categories. Top imports: Mebendazole ($694.5K), Folic ($465.3K), Secnidazole ($187.1K), Ranitidine ($126.2K), Collagen ($41.8K).
Who supplies pharmaceuticals to Eurimex Pharma from India?
Eurimex Pharma sources from 17 verified Indian suppliers. The primary supplier is Cadila Pharmaceuticals Limited (33.0% of imports, $5.7M).
What is Eurimex Pharma's total pharmaceutical import value?
Eurimex Pharma's total pharmaceutical import value from India is $1.5M, based on 64 verified shipments in Indian Customs (DGFT) data.
What therapeutic categories does Eurimex Pharma focus on?
Eurimex Pharma imports across 4 categories. The largest: Antimalarial & Antiparasitic (45.8%), Nutritional Supplements (33.5%), Advanced Antibiotics (12.3%).
Get Full Eurimex Pharma Import Intelligence
Access shipment-level details, supplier connections, pricing data, and competitive analysis. TransData Nexus provides verified Indian Customs (DGFT) data trusted by pharmaceutical trade professionals worldwide.
Official References & Regulatory Resources
Verify import regulations and drug registration requirements with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Buyer Matching: Eurimex Pharma identified across shipments using consignee name normalization, aggregating 2 name variants.
- 2.Statistical Normalization: Shipment values are statistically normalized to ensure accurate market share representation.
- 3.Market Share: Calculated per product as Eurimex Pharma's capped value divided by total Indian exports for that product.
- 4.Shipment Count: Based on 64 individual customs records matching Eurimex Pharma.
- 5.Supplier Verification: Eurimex Pharma sources from 17 verified Indian suppliers across 829 formulations, confirmed from customs records.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
5 Products Tracked
4 therapeutic categories
Expert-Reviewed
By pharmaceutical trade specialists
Data Source & Methodology
Trade data sourced from Indian Customs (DGFT) export shipment records. Values represent FOB export value in USD. Profile aggregates 2 company name variants from customs records. For current shipment-level data, contact TransData Nexus.