East Midlands Pharma Ltd
Pharmaceutical Importer · United Kingdom · Ayurvedic & Herbal Products Focus · $866.4K Total Trade · DGFT Verified
East Midlands Pharma Ltd is a pharmaceutical importer based in United Kingdom with a total trade value of $866.4K across 2 products in 1 therapeutic categories. Based on 23 verified import shipments from Indian Customs (DGFT) records, the company actively imports across multiple product segments. East Midlands Pharma Ltd sources from 1 verified Indian supplier, with Stericon Pharma Private Limited accounting for 100.0% of imports.
East Midlands Pharma Ltd — Import Portfolio & Supplier Network
Who Are the Verified Indian Suppliers to East Midlands Pharma Ltd?
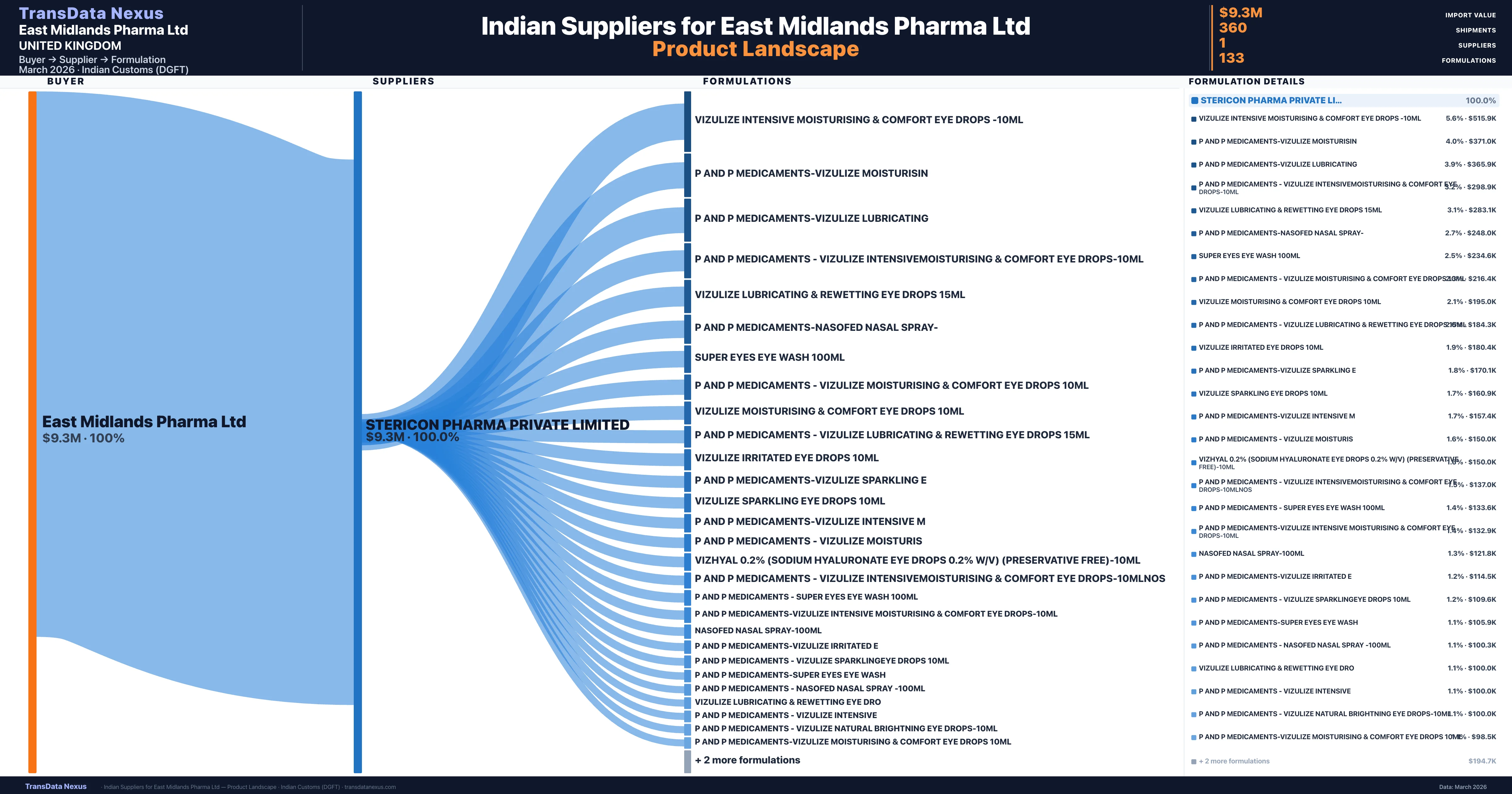
Customs-verified supplier relationships from Indian DGFT records
| Supplier | Value | Shipments | Share |
|---|---|---|---|
| Stericon Pharma Private Limited | $9.3M | 360 | 100.0% |
East Midlands Pharma Ltd sources from 1 verified Indian supplier across 133 distinct formulations. The sourcing is highly concentrated — Stericon Pharma Private Limited accounts for 100.0% of total imports, indicating a strategic single-source relationship.
What Formulations Does East Midlands Pharma Ltd Import?
| Formulation | Value | Ships |
|---|---|---|
| Vizulize intensive moisturising & comfort eye drops -10ML | $515.9K | 12 |
| P and p medicaments-vizulize | $371.0K | 9 |
| P and p medicaments-vizulize | $365.9K | 9 |
| P and p medicaments - vizulize intensivemoisturising & comfort eye drops-10ML | $298.9K | 8 |
| Vizulize lubricating & rewetting eye drops 15ML | $283.1K | 8 |
| P and p medicaments-nasofed nasal | $248.0K | 9 |
| Super eyes eye wash 100ML | $234.6K | 9 |
| P and p medicaments - vizulize moisturising & comfort eye drops 10ML | $216.4K | 7 |
| Vizulize moisturising & comfort eye drops 10ML | $195.0K | 5 |
| P and p medicaments - vizulize lubricating & rewetting eye drops 15ML | $184.3K | 6 |
| Vizulize irritated eye drops 10ML | $180.4K | 4 |
| P and p medicaments-vizulize sparkling | $170.1K | 4 |
| Vizulize sparkling eye drops 10ML | $160.9K | 4 |
| P and p medicaments-vizulize intensive | $157.4K | 4 |
| P and p medicaments - vizulize | $150.0K | 3 |
East Midlands Pharma Ltd imports 133 distinct pharmaceutical formulations. Showing top 15 by value. For full formulation-level data, contact TransData Nexus.
What Products Does East Midlands Pharma Ltd Import?
Top Products by Import Value
East Midlands Pharma Ltd Therapeutic Categories — 1 Specializations
East Midlands Pharma Ltd imports across 1 therapeutic categories, with Ayurvedic & Herbal Products (100.0%), representing the largest segments. The portfolio is concentrated — top 5 products = 100% of total imports.
Ayurvedic & Herbal Products
2 products · 100.0% · $866.4K
Import Portfolio — Top 2 by Import Value
| # | Product | Category | Value | Ships | Share | Rk |
|---|---|---|---|---|---|---|
| 1 | Vati | Ayurvedic & Herbal Products | $500.0K | 10 | 1.9% | 10 |
| 2 | Natural | Ayurvedic & Herbal Products | $366.4K | 13 | 1.4% | 13 |
East Midlands Pharma Ltd imports 2 pharmaceutical products across 1 categories into United Kingdom totaling $866.4K.
Key Metrics
Top Categories
Indian Suppliers
Related Trade Data
Need Detailed Data?
Shipment-level records, supplier connections & pricing for East Midlands Pharma Ltd.
Request DemoEast Midlands Pharma Ltd — Corporate Profile & Information
Company type, headquarters, distribution network, and industry role
1Company Overview
East Midlands Pharma Ltd is a privately held pharmaceutical company based in Leicester, United Kingdom. Established on 15 February 2005, the company specializes in the wholesale distribution of pharmaceutical goods, focusing on the import and supply of finished pharmaceutical formulations. Their product portfolio includes a range of Ayurvedic and herbal products, catering to the growing demand for alternative and complementary medicines in the UK market. The company's registered office is located at Unit 1 Rothley Lodge, Loughborough Road, Rothley, Leicester, LE7 7NL. (find-and-update.company-information.service.gov.uk)
Financially, East Midlands Pharma Ltd has demonstrated consistent growth. In the fiscal year ending 31 March 2025, the company reported a turnover of £22.52 million, marking a 26% increase from the previous year. This growth reflects the company's expanding presence in the pharmaceutical distribution sector. The company's financial statements are audited, with the next accounts due by 31 December 2026.
2Distribution Network
East Midlands Pharma Ltd operates a centralized distribution network from its headquarters in Leicester. The company maintains a warehouse at Unit 1 Rothley Lodge, Loughborough Road, Rothley, Leicester, LE7 7NL, which serves as the primary hub for its logistics operations. While specific details about additional warehouse locations or logistics capabilities are not publicly disclosed, the company's financial growth and increasing turnover suggest an expanding distribution capacity to meet the demands of the UK pharmaceutical market. (find-and-update.company-information.service.gov.uk)
3Industry Role
In the United Kingdom's pharmaceutical supply chain, East Midlands Pharma Ltd functions as a pharmaceutical importer and wholesaler. The company specializes in importing finished pharmaceutical formulations, particularly Ayurvedic and herbal products, from India. By focusing on these specific product categories, East Midlands Pharma Ltd plays a crucial role in diversifying the UK's pharmaceutical offerings and meeting the growing consumer interest in alternative and complementary medicines. (find-and-update.company-information.service.gov.uk)
Supplier Relationship Intelligence — East Midlands Pharma Ltd
Sourcing concentration, supply chain resilience, and strategic implications
1Sourcing Concentration Analysis
East Midlands Pharma Ltd's sourcing strategy exhibits a high degree of concentration, with all 23 shipments from India between 2022 and 2026 originating from a single supplier: STERICON PHARMA PRIVATE LIMITED. This supplier has provided 100% of the imported products, totaling $9.3 million across 360 shipments. Such a concentrated sourcing approach can present risks, including potential supply chain disruptions if the sole supplier faces operational challenges. However, this strategy may also reflect a strategic choice to establish a strong, reliable partnership with a trusted supplier, ensuring consistent product quality and availability. The substantial volume of imports from STERICON PHARMA PRIVATE LIMITED indicates a stable and ongoing relationship, which is essential for maintaining the supply of specialized pharmaceutical products.
2Supply Chain Resilience
The resilience of East Midlands Pharma Ltd's supply chain is closely tied to its exclusive relationship with STERICON PHARMA PRIVATE LIMITED. While this partnership ensures consistency in product quality and supply, it also means that the company is highly dependent on a single supplier. In the event of disruptions—such as production issues, regulatory challenges, or geopolitical factors affecting trade routes—the company's ability to maintain a steady supply of products could be compromised. To enhance supply chain resilience, it would be prudent for East Midlands Pharma Ltd to consider diversifying its supplier base and establishing relationships with additional manufacturers. This strategy would mitigate risks associated with sole-source dependency and provide alternative avenues for sourcing products, thereby strengthening the overall stability of the supply chain.
3Strategic Implications
East Midlands Pharma Ltd's concentrated sourcing strategy positions the company to benefit from deep, long-term partnerships with its suppliers, potentially leading to favorable terms and prioritized access to products. However, this approach also exposes the company to risks associated with supplier dependency, including potential supply chain disruptions. For Indian exporters, the company's focus on Ayurvedic and herbal products presents an opportunity to establish or strengthen partnerships, especially if they can offer products that meet the quality standards and regulatory requirements of the UK market. Diversifying the supplier base could also open avenues for Indian exporters to become alternative suppliers, thereby reducing the risk of sole-source dependency for East Midlands Pharma Ltd.
Importing Pharmaceuticals into United Kingdom — Regulatory Framework
Regulatory authority, GMP requirements, import licensing for United Kingdom
1Regulatory Authority & Framework
In the United Kingdom, the Medicines and Healthcare products Regulatory Agency (MHRA) is the primary regulatory body overseeing the safety, efficacy, and quality of medicines and medical devices. The MHRA operates under the framework established by the Human Medicines Regulations 2012, which transposes European Union directives into UK law. This legislation outlines the requirements for marketing authorization, manufacturing, and distribution of pharmaceutical products within the UK. For Indian pharmaceutical companies seeking to export to the UK, obtaining a marketing authorization from the MHRA is mandatory. This process involves submitting comprehensive data on the product's quality, safety, and efficacy, as well as compliance with Good Manufacturing Practice (GMP) standards.
2Import Licensing & GMP
Import licensing in the UK is governed by the Human Medicines Regulations 2012, which require that all medicines imported into the country have a valid marketing authorization issued by the MHRA. For Indian pharmaceutical companies, this necessitates compliance with UK-specific GMP standards, which may differ from those in India. The MHRA recognizes GMP certificates from countries with which it has mutual recognition agreements, such as the European Union and the United States. However, Indian manufacturers must ensure that their facilities meet the MHRA's GMP requirements, which may involve additional inspections or certifications. Additionally, Indian exporters must obtain wholesale distribution authorization to legally supply medicines to UK wholesalers like East Midlands Pharma Ltd.
3Quality & Labeling
Pharmaceutical products imported into the UK must undergo batch testing to verify their quality, safety, and efficacy. The MHRA conducts these tests to ensure that products meet the required standards before they reach the market. Stability studies are also mandatory to confirm that products maintain their quality throughout their shelf life. Labeling requirements stipulate that all information be provided in English, including dosage instructions, storage conditions, and expiry dates. Serialization mandates are in place to prevent counterfeit medicines; each product unit must have a unique identifier that can be tracked through the supply chain. These measures are designed to protect consumers and maintain the integrity of the pharmaceutical supply chain.
4Recent Regulatory Changes
Between 2024 and 2026, the UK government implemented several policy changes affecting pharmaceutical imports, particularly from non-EU countries like India. These changes include stricter GMP compliance requirements, enhanced batch testing protocols, and more rigorous labeling standards to align with international best practices. Additionally, the MHRA introduced new serialization mandates to combat counterfeit medicines, requiring all imported pharmaceutical products to have unique identifiers. These regulatory updates aim to strengthen the safety and quality of medicines available in the UK market and may necessitate adjustments from Indian exporters to maintain compliance.
East Midlands Pharma Ltd — Procurement Pattern Analysis
Product strategy, sourcing profile, and market positioning
1Product Strategy
East Midlands Pharma Ltd's focus on Ayurvedic and herbal products aligns with the growing consumer interest in natural and alternative medicines in the UK. The market demand for such products is driven by consumers seeking holistic health solutions and a preference for remedies with fewer side effects. By importing these products from India, East Midlands Pharma Ltd leverages the country's rich tradition in herbal medicine and its established manufacturing capabilities. This strategic focus allows the company to cater to a niche market segment, differentiating itself from competitors and meeting the specific needs of health-conscious consumers.
2Sourcing Profile
East Midlands Pharma Ltd's sourcing strategy is centered on importing finished pharmaceutical formulations from India, particularly Ayurvedic and herbal products. This approach allows the company to offer a diverse range of products that appeal to consumers interested in natural health solutions. India's well-established pharmaceutical industry, with its expertise in herbal medicine and compliance with international GMP standards, makes it an ideal sourcing partner. By focusing on finished formulations, East Midlands Pharma Ltd ensures that products are ready for distribution, reducing lead times and streamlining the supply chain.
3Market Positioning
Based on its product mix, East Midlands Pharma Ltd serves the retail pharmacy segment of the UK market. The company's emphasis on Ayurvedic and herbal products positions it to meet the needs of consumers seeking alternative and complementary medicines. By importing these products from India, East Midlands Pharma Ltd offers unique and diverse options that are not widely available from other UK wholesalers. This strategic positioning allows the company to cater to a growing niche market, differentiating itself from competitors and establishing a strong presence in the sector.
Seller's Guide — How to Become a Supplier to East Midlands Pharma Ltd
Opportunity assessment, qualifications, and practical approach strategy
1Opportunity Assessment
There is a realistic opportunity for new Indian suppliers to partner with East Midlands Pharma Ltd, especially those specializing in Ayurvedic and herbal products. The company's current sourcing strategy, which relies exclusively on STERICON PHARMA PRIVATE LIMITED, indicates a potential openness to diversifying its supplier base. Indian exporters that can meet the MHRA's GMP requirements and provide high-quality, stable products with appropriate labeling and serialization are well-positioned to enter this market. Additionally, offering unique formulations or innovative products could further enhance the appeal to East Midlands Pharma Ltd, aligning with its focus on diversifying the UK's pharmaceutical offerings.
Frequently Asked Questions — East Midlands Pharma Ltd
What products does East Midlands Pharma Ltd import from India?
East Midlands Pharma Ltd imports 2 pharmaceutical products across 1 categories. Top imports: Vati ($500.0K), Natural ($366.4K).
Who supplies pharmaceuticals to East Midlands Pharma Ltd from India?
East Midlands Pharma Ltd sources from 1 verified Indian suppliers. The primary supplier is Stericon Pharma Private Limited (100.0% of imports, $9.3M).
What is East Midlands Pharma Ltd's total pharmaceutical import value?
East Midlands Pharma Ltd's total pharmaceutical import value from India is $866.4K, based on 23 verified shipments in Indian Customs (DGFT) data.
What therapeutic categories does East Midlands Pharma Ltd focus on?
East Midlands Pharma Ltd imports across 1 categories. The largest: Ayurvedic & Herbal Products (100.0%).
Get Full East Midlands Pharma Ltd Import Intelligence
Access shipment-level details, supplier connections, pricing data, and competitive analysis. TransData Nexus provides verified Indian Customs (DGFT) data trusted by pharmaceutical trade professionals worldwide.
Official References & Regulatory Resources
Verify import regulations and drug registration requirements with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Buyer Matching: East Midlands Pharma Ltd identified across shipments using consignee name normalization.
- 2.Statistical Normalization: Shipment values are statistically normalized to ensure accurate market share representation.
- 3.Market Share: Calculated per product as East Midlands Pharma Ltd's capped value divided by total Indian exports for that product.
- 4.Shipment Count: Based on 23 individual customs records matching East Midlands Pharma Ltd.
- 5.Supplier Verification: East Midlands Pharma Ltd sources from 1 verified Indian suppliers across 133 formulations, confirmed from customs records.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
2 Products Tracked
1 therapeutic categories
Expert-Reviewed
By pharmaceutical trade specialists
Data Source & Methodology
Trade data sourced from Indian Customs (DGFT) export shipment records. Values represent FOB export value in USD. For current shipment-level data, contact TransData Nexus.