Central Medical Stores
Pharmaceutical Importer · Botswana · Cardiovascular Focus · $300.0K Total Trade · DGFT Verified
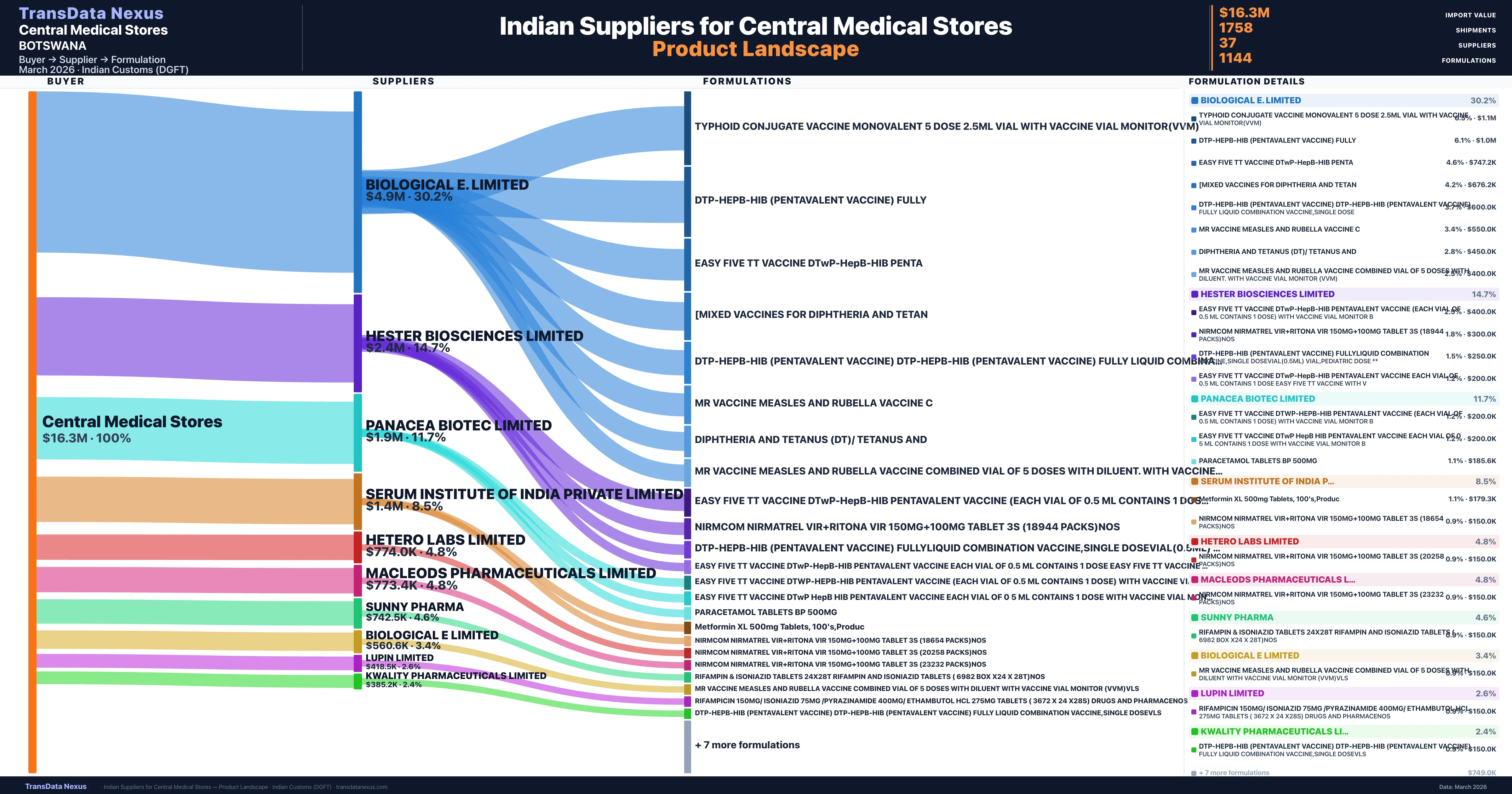
Central Medical Stores is a pharmaceutical importer based in Botswana with a total trade value of $300.0K across 2 products in 1 therapeutic categories. Based on 6 verified import shipments from Indian Customs (DGFT) records, the company actively imports across multiple product segments. Central Medical Stores sources from 37 verified Indian suppliers, with Biological E. Limited accounting for 30.2% of imports.
Central Medical Stores — Import Portfolio & Supplier Network
Who Are the Verified Indian Suppliers to Central Medical Stores?
Customs-verified supplier relationships from Indian DGFT records
| Supplier | Value | Shipments | Share |
|---|---|---|---|
| Biological E. Limited | $4.9M | 101 | 30.2% |
| Hester Biosciences Limited | $2.4M | 82 | 14.7% |
| Panacea Biotec Limited | $1.9M | 38 | 11.7% |
| Serum Institute Of India Private Limited | $1.4M | 35 | 8.5% |
| Hetero Labs Limited | $774.0K | 20 | 4.8% |
| Macleods Pharmaceuticals Limited | $773.4K | 65 | 4.8% |
| Sunny Pharma | $742.5K | 796 | 4.6% |
| Biological E Limited | $560.6K | 13 | 3.4% |
| Lupin Limited | $418.5K | 15 | 2.6% |
| Kwality Pharmaceuticals Limited | $385.2K | 216 | 2.4% |
| Mylan Laboratories Limited | $336.0K | 22 | 2.1% |
| Medopharm | $334.0K | 9 | 2.1% |
| Micro Labs Limited | $272.5K | 14 | 1.7% |
| Greensignal Bio Pharma Private Limited | $200.0K | 4 | 1.2% |
| Salvavita Pharmaceuticals Private Limited | $158.9K | 6 | 1.0% |
| Exsell Laboratories Private Limited | $137.3K | 59 | 0.8% |
| Fdc Limited | $100.1K | 3 | 0.6% |
| Cipla Limited | $98.9K | 37 | 0.6% |
| Milan Laboratories (india) Private Limited | $75.0K | 5 | 0.5% |
| Aurobindo Pharma Limited | $53.3K | 7 | 0.3% |
| Bharat Biotech International Limited | $50.0K | 1 | 0.3% |
| Spr Oncocare Private Limited | $48.4K | 130 | 0.3% |
| Ipca Laboratories Limited | $36.3K | 9 | 0.2% |
| Strides Pharma Science Limited | $29.8K | 5 | 0.2% |
| Gospel Pharmaceuticals | $21.9K | 10 | 0.1% |
| Emcure Pharmaceuticals Limited | $21.4K | 6 | 0.1% |
| Venus Remedies Limited | $18.4K | 2 | 0.1% |
| Phar Vide | $13.3K | 5 | 0.1% |
| Nova Impex | $10.1K | 14 | 0.1% |
| Devlife Corporation Private Limited | $7.1K | 8 | 0.0% |
Central Medical Stores sources from 37 verified Indian suppliers across 1,144 distinct formulations. The supply base is diversified across 37 suppliers, reducing single-source dependency risk.
What Formulations Does Central Medical Stores Import?
| Formulation | Value | Ships |
|---|---|---|
| Typhoid conjugate vaccine monovalent 5 dose 2.5ML vial with vaccine vial monitor(vvm) | $1.1M | 21 |
| Dtp-hepb-hib (pentavalent vaccine) | $1.0M | 20 |
| Easy five tt vaccine dtwp-hepb-hib | $747.2K | 15 |
| [mixed vaccines for diphtheria and | $676.2K | 15 |
| Dtp-hepb-hib (pentavalent vaccine) dtp-hepb-hib (pentavalent vaccine) fully liquid combination vaccine,single | $600.0K | 12 |
| Mr vaccine measles and rubella vaccine | $550.0K | 11 |
| Diphtheria and tetanus (dt)/ tetanus | $450.0K | 9 |
| Mr vaccine measles and rubella vaccine combined vial of 5 doses with diluent. with vaccine vial monitor (vvm) | $400.0K | 8 |
| Easy five tt vaccine dtwp-hepb-hib pentavalent vaccine (Each vial of 0.5 ML contains 1 dose) with vaccine vial monitor | $400.0K | 8 |
| Nirmcom nirmatrel vir+ritona vir 150MG+100MG tablet 3s (18944 packs | $300.0K | 6 |
| Dtp-hepb-hib (pentavalent vaccine) fullyliquid combination vaccine,single dosevial(0.5ML) vial,pediatric dose | $250.0K | 5 |
| Easy five tt vaccine dtwp-hepb-hib pentavalent vaccine Each vial of 0.5 ML contains 1 dose easy five tt vaccine with | $200.0K | 4 |
| Easy five tt vaccine dtwp-hepb-hib pentavalent vaccine (Each vial of 0.5 ML contains 1 dose) with vaccine vial monitor | $200.0K | 4 |
| Easy five tt vaccine dtwp hepb hib pentavalent vaccine Each vial of 0 5 ML contains 1 dose with vaccine vial monitor | $200.0K | 4 |
| Paracetamol tablets BP 500MG | $185.6K | 4 |
Central Medical Stores imports 1,144 distinct pharmaceutical formulations. Showing top 15 by value. For full formulation-level data, contact TransData Nexus.
What Products Does Central Medical Stores Import?
Top Products by Import Value
Central Medical Stores Therapeutic Categories — 1 Specializations
Central Medical Stores imports across 1 therapeutic categories, with Cardiovascular (100.0%), representing the largest segments. The portfolio is concentrated — top 5 products = 100% of total imports.
Cardiovascular
2 products · 100.0% · $300.0K
Import Portfolio — Top 2 by Import Value
| # | Product | Category | Value | Ships | Share | Rk |
|---|---|---|---|---|---|---|
| 1 | Enalapril | Cardiovascular | $150.0K | 3 | 0.1% | 13 |
| 2 | Nifedipine | Cardiovascular | $150.0K | 3 | 0.5% | 9 |
Central Medical Stores imports 2 pharmaceutical products across 1 categories into Botswana totaling $300.0K.
Key Metrics
Top Categories
Indian Suppliers
Related Trade Data
Need Detailed Data?
Shipment-level records, supplier connections & pricing for Central Medical Stores.
Request DemoCentral Medical Stores — Corporate Profile & Information
Company type, headquarters, distribution network, and industry role
1Company Overview
Central Medical Stores (CMS) is a pharmaceutical importer and buyer based in Gaborone, Botswana. Operating as a government procurement entity, CMS plays a pivotal role in the country's healthcare system by sourcing and distributing essential medicines and medical supplies. This ensures that both public and private healthcare facilities have access to necessary pharmaceuticals. The organization is headquartered at Gaborone West Industrial Site Kgomoka 22/02/3/456, P.O.Box 639, Gaborone.
CMS's primary function is to procure and supply a wide range of pharmaceutical products to meet the healthcare needs of Botswana's population. By centralizing procurement, CMS aims to achieve cost efficiencies and maintain consistent quality standards across the nation's medical facilities. This centralized approach also facilitates better inventory management and distribution logistics, ensuring timely delivery of medicines to various healthcare providers.
2Distribution Network
While specific details about CMS's warehouse locations and logistics capabilities are not publicly disclosed, it is known that the organization operates within Botswana's established distribution infrastructure. This includes utilizing the country's main ports of entry and collaborating with local transportation networks to ensure efficient delivery of pharmaceutical products. Given Botswana's landlocked geography, CMS likely relies on regional trade routes, particularly through neighboring South Africa, to import medicines. This necessitates coordination with customs authorities and adherence to import regulations to facilitate smooth cross-border trade.
CMS's distribution network is designed to cover the entire country, ensuring that both urban and rural healthcare facilities receive the necessary medical supplies. The organization likely employs a combination of direct deliveries and partnerships with local distributors to reach various regions effectively. This comprehensive coverage is essential for maintaining the health and well-being of Botswana's population, especially in remote areas where access to medical supplies can be limited.
3Industry Role
In Botswana's pharmaceutical supply chain, Central Medical Stores serves as a primary wholesaler and government procurement entity. By centralizing the procurement and distribution of pharmaceutical products, CMS ensures that healthcare facilities across the country have access to essential medicines. This centralized approach allows for standardized quality control, cost efficiencies, and streamlined logistics, which are crucial for maintaining a consistent supply of pharmaceuticals.
CMS's role is particularly significant in a landlocked country like Botswana, where importing medicines involves navigating complex logistics and regulatory requirements. By managing these processes centrally, CMS can negotiate better terms with suppliers, ensure compliance with import regulations, and address any supply chain challenges more effectively. This strategic position enables CMS to play a critical role in the nation's healthcare system, ensuring that both public and private healthcare providers have access to the necessary pharmaceutical products.
Supplier Relationship Intelligence — Central Medical Stores
Sourcing concentration, supply chain resilience, and strategic implications
1Sourcing Concentration Analysis
Central Medical Stores's sourcing strategy exhibits a high degree of concentration, with a total import value of $300,000 USD from India across six shipments. The top two products imported are Enalapril and Nifedipine, each accounting for $150,000 USD, representing 0.1% and 0.5% of the total import value, respectively. Both products fall under the cardiovascular therapeutic category, indicating a focused approach to sourcing specific pharmaceutical formulations.
This concentrated sourcing strategy suggests that CMS may be leveraging strategic partnerships with Indian suppliers to secure favorable pricing and ensure a consistent supply of these critical medications. However, such dependency on a limited number of suppliers and products could pose risks, including potential supply disruptions or price fluctuations. To mitigate these risks, CMS may consider diversifying its supplier base and product portfolio, ensuring a more resilient and adaptable supply chain.
2Supply Chain Resilience
The resilience of Central Medical Stores's Indian supply chain is influenced by several factors, including the stability of supplier relationships, the diversity of imported formulations, and exposure to shipping route disruptions. With a total of 37 unique suppliers and 1,144 unique formulations imported, CMS demonstrates a relatively diverse sourcing strategy. This diversity can enhance supply chain resilience by reducing dependency on a single supplier or product.
However, the concentration of imports in the cardiovascular therapeutic category, with Enalapril and Nifedipine accounting for a significant portion of the import value, indicates a potential vulnerability. To strengthen supply chain resilience, CMS could explore alternative suppliers for these key products and consider expanding its product range to include other therapeutic areas. Additionally, evaluating and diversifying shipping routes can help mitigate risks associated with logistical disruptions.
3Strategic Implications
Central Medical Stores's concentrated sourcing pattern, particularly from India, positions the organization to benefit from cost efficiencies and established supplier relationships. This strategy allows CMS to negotiate favorable terms and maintain a consistent supply of essential cardiovascular medications. However, the heavy reliance on a limited number of suppliers and products may expose CMS to risks such as supply chain disruptions or price volatility.
For Indian exporters, this sourcing pattern presents an opportunity to strengthen existing partnerships with CMS by offering a broader range of products or more competitive pricing. Additionally, demonstrating compliance with international quality standards and understanding the regulatory requirements of the Botswana market can enhance the attractiveness of Indian pharmaceutical products to CMS.
Importing Pharmaceuticals into Botswana — Regulatory Framework
Regulatory authority, GMP requirements, import licensing for Botswana
1Regulatory Authority & Framework
The Botswana Medicines Regulatory Authority (BoMRA) is the primary regulatory body responsible for overseeing the safety, efficacy, and quality of medicines and related substances in Botswana. Established under the Medicines and Related Substances Act (MRSA), BoMRA's key functions include product evaluation and registration, pharmacovigilance, clinical trials oversight, and import/export control. (bomra.co.bw)
The MRSA provides the legal framework for the importation, exportation, distribution, and sale of medicines in Botswana. According to the Act, no drug shall be imported into or exported from Botswana, or manufactured, distributed, or sold, unless such drug has been registered by the Director of Health Services.
2Import Licensing & GMP
Importers of pharmaceutical products into Botswana must obtain import licenses from BoMRA. The application process involves submitting the prescribed form, paying the required fee, and providing necessary information as specified by the authority. Importers must be residents of Botswana and operate under the continuous supervisory control of a pharmacist.
BoMRA recognizes Good Manufacturing Practice (GMP) certificates from reputable international regulatory bodies, including the European Union (EU) GMP, World Health Organization (WHO) GMP, and Pharmaceutical Inspection Co-operation Scheme (PIC/S). Importers must ensure that their suppliers hold valid GMP certifications to comply with BoMRA's standards.
3Quality & Labeling
Imported pharmaceutical products must undergo batch testing to ensure they meet the approved specifications and regulations. Stability studies are required to confirm the product's shelf life and efficacy over time. Labeling must be in English and include essential information such as the product name, active ingredients, dosage form, batch number, manufacturing date, expiry date, storage conditions, and the manufacturer's details. Serialization mandates may apply to facilitate traceability and prevent counterfeit products.
4Recent Regulatory Changes
Between 2024 and 2026, BoMRA has implemented several policy changes affecting pharmaceutical imports into Botswana. These include stricter enforcement of GMP certification requirements, enhanced scrutiny of import licenses, and updated guidelines for labeling and packaging to align with international standards. Importers must stay informed about these changes to ensure compliance and maintain uninterrupted access to the Botswana market.
Central Medical Stores — Procurement Pattern Analysis
Product strategy, sourcing profile, and market positioning
1Product Strategy
Central Medical Stores's focus on importing cardiovascular medications, specifically Enalapril and Nifedipine, aligns with the high prevalence of cardiovascular diseases in Botswana. This strategic choice is driven by the significant demand for these medications to manage conditions such as hypertension and heart failure. By prioritizing these products, CMS aims to address a critical health need and improve patient outcomes.
The decision to concentrate on cardiovascular drugs may also be influenced by cost considerations, as these medications are often available in generic forms, offering more affordable options. Additionally, the established efficacy and safety profiles of these drugs make them a reliable choice for treatment protocols.
2Sourcing Profile
CMS's sourcing strategy is centered on importing generic formulations from India, a country known for its robust pharmaceutical manufacturing sector. This approach allows CMS to access a wide range of cost-effective and quality-assured medications. By focusing on finished pharmaceutical formulations, CMS ensures that the products are ready for distribution and use, minimizing the need for additional processing or repackaging.
India's compliance with international quality standards, such as WHO GMP, further enhances the reliability of the imported products. This sourcing strategy supports CMS's objective of providing affordable and effective medications to the Botswana market.
3Market Positioning
Based on its product mix, Central Medical Stores primarily serves the public healthcare sector in Botswana, including government hospitals and clinics. By focusing on essential medications like Enalapril and Nifedipine, CMS ensures that these facilities have access to critical treatments for cardiovascular conditions. This positioning underscores CMS's role in supporting the government's efforts to provide comprehensive
Frequently Asked Questions — Central Medical Stores
What products does Central Medical Stores import from India?
Central Medical Stores imports 2 pharmaceutical products across 1 categories. Top imports: Enalapril ($150.0K), Nifedipine ($150.0K).
Who supplies pharmaceuticals to Central Medical Stores from India?
Central Medical Stores sources from 37 verified Indian suppliers. The primary supplier is Biological E. Limited (30.2% of imports, $4.9M).
What is Central Medical Stores's total pharmaceutical import value?
Central Medical Stores's total pharmaceutical import value from India is $300.0K, based on 6 verified shipments in Indian Customs (DGFT) data.
What therapeutic categories does Central Medical Stores focus on?
Central Medical Stores imports across 1 categories. The largest: Cardiovascular (100.0%).
Get Full Central Medical Stores Import Intelligence
Access shipment-level details, supplier connections, pricing data, and competitive analysis. TransData Nexus provides verified Indian Customs (DGFT) data trusted by pharmaceutical trade professionals worldwide.
Official References & Regulatory Resources
Verify import regulations and drug registration requirements with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Buyer Matching: Central Medical Stores identified across shipments using consignee name normalization.
- 2.Statistical Normalization: Shipment values are statistically normalized to ensure accurate market share representation.
- 3.Market Share: Calculated per product as Central Medical Stores's capped value divided by total Indian exports for that product.
- 4.Shipment Count: Based on 6 individual customs records matching Central Medical Stores.
- 5.Supplier Verification: Central Medical Stores sources from 37 verified Indian suppliers across 1,144 formulations, confirmed from customs records.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
2 Products Tracked
1 therapeutic categories
Expert-Reviewed
By pharmaceutical trade specialists
Data Source & Methodology
Trade data sourced from Indian Customs (DGFT) export shipment records. Values represent FOB export value in USD. For current shipment-level data, contact TransData Nexus.