Cameg
Pharmaceutical Importer · Cote D Ivoire · Analgesics & Antipyretics Focus · $5.3M Total Trade · DGFT Verified
Cameg is a pharmaceutical importer based in Cote D Ivoire with a total trade value of $5.3M across 8 products in 5 therapeutic categories. Based on 115 verified import shipments from Indian Customs (DGFT) records, the company actively imports across multiple product segments. Cameg sources from 37 verified Indian suppliers, with Sidhaant Life Sciences Private Limited accounting for 34.5% of imports.
Cameg — Import Portfolio & Supplier Network
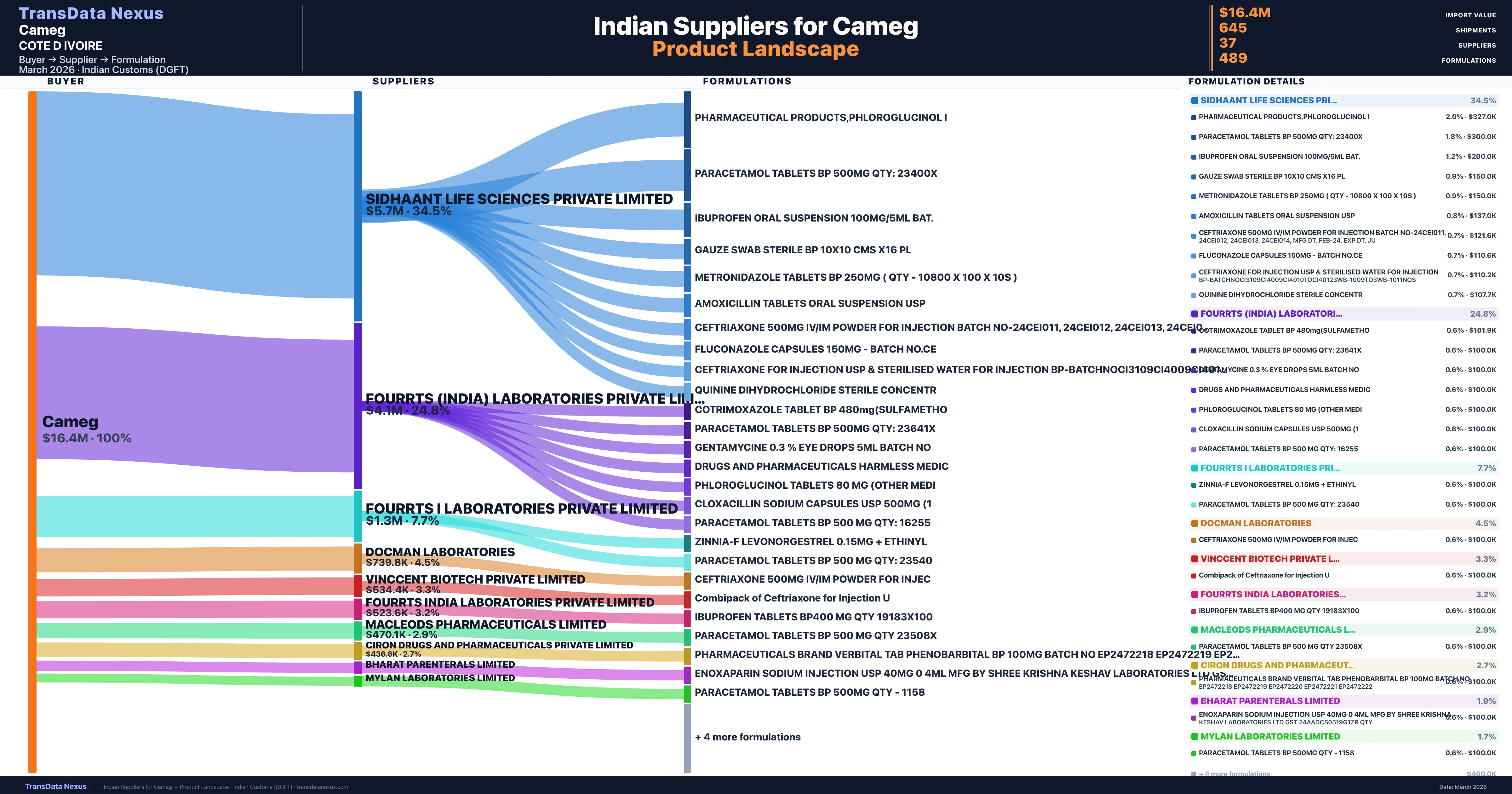
Who Are the Verified Indian Suppliers to Cameg?
Customs-verified supplier relationships from Indian DGFT records
| Supplier | Value | Shipments | Share |
|---|---|---|---|
| Sidhaant Life Sciences Private Limited | $5.7M | 188 | 34.5% |
| Fourrts (india) Laboratories Private Limited | $4.1M | 131 | 24.8% |
| Fourrts I Laboratories Private Limited | $1.3M | 34 | 7.7% |
| Docman Laboratories | $739.8K | 19 | 4.5% |
| Vinccent Biotech Private Limited | $534.4K | 13 | 3.3% |
| Fourrts India Laboratories Private Limited | $523.6K | 16 | 3.2% |
| Macleods Pharmaceuticals Limited | $470.1K | 23 | 2.9% |
| Ciron Drugs And Pharmaceuticals Private Limited | $436.6K | 20 | 2.7% |
| Bharat Parenterals Limited | $310.7K | 11 | 1.9% |
| Mylan Laboratories Limited | $273.0K | 6 | 1.7% |
| Venor Pharma Limited | $265.4K | 24 | 1.6% |
| Kilitch Drugs India Limited | $241.7K | 9 | 1.5% |
| Medopharm Private Limited | $237.0K | 5 | 1.4% |
| Medicamen Biotech Limited | $173.9K | 4 | 1.1% |
| Kwality Pharmaceuticals Limited | $157.4K | 20 | 1.0% |
| Mednext Biotech Limited | $120.5K | 5 | 0.7% |
| Vins Bioproducts Limited | $112.8K | 3 | 0.7% |
| Kilitch Drugs (india) Limited | $106.4K | 5 | 0.6% |
| Africure Pharmaceuticals (india) Privatelimited | $100.0K | 4 | 0.6% |
| Verve Human Care Laboratories | $89.1K | 9 | 0.5% |
| Rena Exports Private Limited | $84.3K | 7 | 0.5% |
| Aishwarya Healthcare | $67.9K | 8 | 0.4% |
| Sparsh Bio-tech Private Limited | $62.5K | 2 | 0.4% |
| Gtonic Lifesciences Limited Liability Partnership | $51.0K | 2 | 0.3% |
| Suave Med Sciences Private Limited | $50.0K | 1 | 0.3% |
| Solitaire Trade Link Private Limited | $46.3K | 1 | 0.3% |
| Aishwarya Health Care | $37.9K | 4 | 0.2% |
| Globela Pharma Private Limited | $34.4K | 2 | 0.2% |
| Otsuka Pharmaceutical India Private Limited | $33.5K | 13 | 0.2% |
| Troikaa Pharmaceuticals Limited | $18.9K | 2 | 0.1% |
Cameg sources from 37 verified Indian suppliers across 489 distinct formulations. The supply base is diversified across 37 suppliers, reducing single-source dependency risk.
What Formulations Does Cameg Import?
| Formulation | Value | Ships |
|---|---|---|
| Pharmaceutical products,phloroglucinol | $327.0K | 7 |
| Paracetamol tablets BP 500MG qty | $300.0K | 6 |
| Ibuprofen oral suspension 100MG/5ML bat. | $200.0K | 4 |
| Gauze swab sterile BP 10x10 cms x16 | $150.0K | 3 |
| Metronidazole tablets BP 250MG ( qty - 10800 x 100 x 10s ) | $150.0K | 3 |
| Amoxicillin tablets oral suspension | $137.0K | 3 |
| Ceftriaxone 500MG iv/im powder for injection batch no-24cei011, 24cei012, 24cei013, 24cei014, mfg dt. feb-24, exp dt. | $121.6K | 3 |
| Fluconazole capsules 150MG - batch | $110.6K | 8 |
| Ceftriaxone for injection USP & sterilised water for injection | $110.2K | 3 |
| Quinine dihydrochloride sterile | $107.7K | 4 |
| Cotrimoxazole tablet BP | $101.9K | 3 |
| Paracetamol tablets BP 500MG qty | $100.0K | 2 |
| Gentamycine 0.3 % eye drops 5ML batch | $100.0K | 2 |
| Drugs and pharmaceuticals harmless | $100.0K | 2 |
| Phloroglucinol tablets 80 MG (other | $100.0K | 2 |
Cameg imports 489 distinct pharmaceutical formulations. Showing top 15 by value. For full formulation-level data, contact TransData Nexus.
What Products Does Cameg Import?
Cameg Therapeutic Categories — 5 Specializations
Cameg imports across 5 therapeutic categories, with Analgesics & Antipyretics (47.1%), Advanced Antibiotics (31.1%), Antimalarial & Antiparasitic (10.2%) representing the largest segments. The portfolio is concentrated — top 5 products = 84% of total imports.
Analgesics & Antipyretics
1 products · 47.1% · $2.5M
Advanced Antibiotics
3 products · 31.1% · $1.6M
Antimalarial & Antiparasitic
2 products · 10.2% · $540.8K
Advanced Antifungals
1 products · 6.0% · $317.1K
Cardiovascular
1 products · 5.7% · $300.0K
Import Portfolio — Top 8 by Import Value
| # | Product | Category | Value | Ships | Share | Rk |
|---|---|---|---|---|---|---|
| 1 | Paracetamol | Analgesics & Antipyretics | $2.5M | 50 | 0.7% | 4 |
| 2 | Ceftriaxone | Advanced Antibiotics | $850.0K | 17 | 0.3% | 17 |
| 3 | Sulfamethoxazole | Advanced Antibiotics | $400.0K | 8 | 2.0% | 3 |
| 4 | Trimethoprim | Advanced Antibiotics | $400.0K | 8 | 1.5% | 3 |
| 5 | Griseofulvin | Advanced Antifungals | $317.1K | 7 | 0.9% | 8 |
| 6 | Mebendazole | Antimalarial & Antiparasitic | $301.3K | 13 | 2.2% | 6 |
| 7 | Enoxaparin | Cardiovascular | $300.0K | 6 | 0.1% | 19 |
| 8 | Quinine | Antimalarial & Antiparasitic | $239.5K | 6 | 1.2% | 12 |
Cameg imports 8 pharmaceutical products across 5 categories into Cote D Ivoire totaling $5.3M.
Key Metrics
Top Categories
Indian Suppliers
Related Trade Data
Cameg — Corporate Profile & Information
Company type, headquarters, distribution network, and industry role
1Company Overview
CAMEG, the Centrale d'Achat des Médicaments Essentiels Génériques et des Consommables Médicaux, is a non-profit organization established to ensure the availability and accessibility of essential generic medicines and medical consumables in Côte d'Ivoire. Operating under the Ministry of Health and Public Hygiene, CAMEG plays a pivotal role in the national healthcare supply chain by procuring, storing, and distributing pharmaceutical products to both public and private health facilities. This mission aligns with the government's objective to provide quality healthcare services to the population.
Headquartered in Abidjan, Côte d'Ivoire, CAMEG's operations are guided by the Direction de la Pharmacie, du Médicament et des Laboratoires (DPML), the country's pharmaceutical regulatory authority. The organization is committed to adhering to the regulatory frameworks established by the DPML, ensuring that all imported pharmaceutical products meet the required standards for safety, efficacy, and quality. CAMEG's strategic focus on essential medicines underscores its dedication to public health and its integral role in the nation's healthcare infrastructure.
2Distribution Network
CAMEG's distribution network is designed to ensure the efficient delivery of pharmaceutical products across Côte d'Ivoire. The organization operates a centralized warehouse located in Abidjan, which serves as the main hub for receiving, storing, and dispatching medical supplies. From this central point, products are distributed to various regional centers and health facilities throughout the country, ensuring that essential medicines are accessible to both urban and rural populations. This centralized approach allows for streamlined operations and effective inventory management, facilitating timely and reliable supply chain processes.
In addition to its domestic distribution capabilities, CAMEG engages in international procurement to source pharmaceutical products that are not locally available. This global sourcing strategy enables the organization to diversify its product offerings and meet the diverse healthcare needs of the Ivorian population. By leveraging both domestic and international supply chains, CAMEG enhances its capacity to provide a comprehensive range of medical supplies, contributing to the overall resilience and effectiveness of the country's healthcare system.
3Industry Role
Within Côte d'Ivoire's pharmaceutical supply chain, CAMEG functions primarily as a wholesaler and distributor. Its core responsibility is to procure and supply essential medicines and medical consumables to public and private health facilities, ensuring that these institutions have access to the necessary pharmaceutical products to deliver quality healthcare services. By centralizing the procurement and distribution processes, CAMEG aims to achieve economies of scale, reduce costs, and improve the efficiency of the healthcare supply chain. This centralized model also facilitates better inventory management and ensures a consistent supply of essential medicines across the country.
CAMEG's role extends beyond mere distribution; it also involves strategic planning and coordination with various stakeholders, including the Ministry of Health, healthcare providers, and international suppliers. This collaborative approach enables CAMEG to anticipate and respond to the evolving healthcare needs of the population, adapt to emerging health challenges, and implement initiatives that enhance the overall effectiveness of the healthcare system. Through its comprehensive and integrated approach, CAMEG plays a crucial role in maintaining the stability and reliability of Côte d'Ivoire's pharmaceutical supply chain.
Supplier Relationship Intelligence — Cameg
Sourcing concentration, supply chain resilience, and strategic implications
1Sourcing Concentration Analysis
CAMEG's sourcing strategy reveals a significant concentration in its supplier base, with a substantial portion of its pharmaceutical imports originating from a select group of Indian suppliers. The data indicates that the top five suppliers account for approximately 84.2% of the total import value, highlighting a dependency on these suppliers for the majority of its pharmaceutical products. This concentration suggests that CAMEG's procurement strategy is heavily reliant on a few key partners, which could pose risks related to supply chain disruptions, price fluctuations, or changes in supplier reliability.
The shipment data further illustrates the stability of these supplier relationships, with consistent and frequent shipments over the years. For instance, SIDHAANT LIFE SCIENCES PRIVATE LIMITED has been a dominant supplier, contributing 34.5% of the total import value through 188 shipments. Similarly, FOURRTS (INDIA) LABORATORIES PRIVATE LIMITED has supplied 24.8% of the total import value across 131 shipments. This consistent engagement indicates a stable and ongoing partnership between CAMEG and these suppliers. However, the high concentration also underscores the potential vulnerability of CAMEG's supply chain to any disruptions affecting these key suppliers.
2Supply Chain Resilience
CAMEG's supply chain resilience is closely tied to its sourcing strategy, which is characterized by a high degree of concentration among a few key Indian suppliers. While this approach offers benefits such as streamlined procurement processes and established relationships, it also exposes CAMEG to potential risks. For example, any disruptions in the operations of these primary suppliers—such as production delays, quality issues, or geopolitical challenges—could significantly impact CAMEG's ability to maintain a consistent supply of essential medicines.
To enhance supply chain resilience, it would be prudent for CAMEG to diversify its supplier base by engaging with additional manufacturers, both within India and internationally. This diversification strategy would mitigate the risks associated with over-reliance on a limited number of suppliers and provide alternative sources in case of disruptions. Additionally, establishing contingency plans, maintaining safety stock levels, and fostering relationships with multiple suppliers can further strengthen the resilience of CAMEG's supply chain, ensuring the continuous availability of essential medicines to meet the healthcare needs of the Ivorian population.
3Strategic Implications
CAMEG's current sourcing pattern, characterized by a high concentration of imports from a select group of Indian suppliers, has several strategic implications. For CAMEG, this approach offers the advantages of established relationships, potential cost efficiencies, and streamlined procurement processes. However, the dependency on a limited number of suppliers also introduces risks related to supply chain disruptions, price volatility, and potential challenges in negotiating favorable terms.
For Indian exporters, the existing sourcing pattern presents both opportunities and challenges. The established relationships between CAMEG and its primary suppliers may make it challenging for new entrants to penetrate the market. However, by offering competitive pricing, high-quality products, and reliable delivery schedules, new Indian suppliers can position themselves as viable alternatives to the existing suppliers. Additionally, diversifying the supplier base can be a strategic move for CAMEG to mitigate risks associated with over-reliance on a few suppliers, creating opportunities for new entrants to establish partnerships and expand their market presence in Côte d'Ivoire.
Importing Pharmaceuticals into Cote D Ivoire — Regulatory Framework
Regulatory authority, GMP requirements, import licensing for Cote D Ivoire
1Regulatory Authority & Framework
In Côte d'Ivoire, the regulation of pharmaceutical products is overseen by the Direction de la Pharmacie, du Médicament et des Laboratoires (DPML), which operates under the Ministry of Health and Public Hygiene. The DPML is responsible for ensuring that all pharmaceutical products imported into the country meet the required standards for safety, efficacy, and quality. This includes the registration and approval of pharmaceutical products, as well as the issuance of import authorizations. The regulatory framework established by the DPML is designed to protect public health by ensuring that only safe and effective pharmaceutical products are available in the Ivorian market.
The legal basis for the regulation of the pharmaceutical sector in Côte d'Ivoire is provided by Law No. 2017-541 of 3 August 2017. This law establishes the institutional and functional framework for the pharmaceutical regulatory system, aiming to guarantee the use of accessible, safe, effective, and high-quality pharmaceutical products. The law outlines the responsibilities of various stakeholders, including importers, exporters, and regulatory authorities, and sets forth the procedures for the import and export of pharmaceutical products. By adhering to this legal framework, CAMEG ensures compliance with national regulations and contributes to the overall integrity of the pharmaceutical supply chain in Côte d'Ivoire.
2Import Licensing & GMP
Importing pharmaceutical products into Côte d'Ivoire requires obtaining an import authorization from the DPML. This process involves submitting an application that includes necessary documentation, such as a commercial invoice, certificate of origin, and a packing list. The application must be submitted to the DPML for review and approval before the shipment of pharmaceutical products. This regulatory requirement ensures that all imported pharmaceutical products meet the safety and quality standards set by the Ivorian authorities.
In addition to import authorization, pharmaceutical products must comply with Good Manufacturing Practice (GMP) standards. Côte d'Ivoire recognizes GMP certifications from reputable organizations, including the European Union (EU) GMP, World Health Organization (WHO) GMP, and Pharmaceutical Inspection Co-operation Scheme (PIC/S). Importers are required to ensure that their products are manufactured in facilities that adhere to these recognized GMP standards. This requirement underscores the commitment to maintaining high-quality pharmaceutical products in the Ivorian market.
3Quality & Labeling
Imported pharmaceutical products are subject to quality control measures to ensure their safety and efficacy. This includes batch testing to verify the identity, purity, and potency of the products. Stability studies are also conducted to determine the shelf life and storage conditions of the products. These quality control processes are essential to ensure that pharmaceutical products remain effective and safe for use throughout their intended shelf life.
Labeling requirements for pharmaceutical products in Côte d'Ivoire include the use of the French language for all labeling and packaging materials. This ensures
Frequently Asked Questions — Cameg
What products does Cameg import from India?
Cameg imports 8 pharmaceutical products across 5 categories. Top imports: Paracetamol ($2.5M), Ceftriaxone ($850.0K), Sulfamethoxazole ($400.0K), Trimethoprim ($400.0K), Griseofulvin ($317.1K).
Who supplies pharmaceuticals to Cameg from India?
Cameg sources from 37 verified Indian suppliers. The primary supplier is Sidhaant Life Sciences Private Limited (34.5% of imports, $5.7M).
What is Cameg's total pharmaceutical import value?
Cameg's total pharmaceutical import value from India is $5.3M, based on 115 verified shipments in Indian Customs (DGFT) data.
What therapeutic categories does Cameg focus on?
Cameg imports across 5 categories. The largest: Analgesics & Antipyretics (47.1%), Advanced Antibiotics (31.1%), Antimalarial & Antiparasitic (10.2%).
Get Full Cameg Import Intelligence
Access shipment-level details, supplier connections, pricing data, and competitive analysis. TransData Nexus provides verified Indian Customs (DGFT) data trusted by pharmaceutical trade professionals worldwide.
Official References & Regulatory Resources
Verify import regulations and drug registration requirements with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Buyer Matching: Cameg identified across shipments using consignee name normalization.
- 2.Statistical Normalization: Shipment values are statistically normalized to ensure accurate market share representation.
- 3.Market Share: Calculated per product as Cameg's capped value divided by total Indian exports for that product.
- 4.Shipment Count: Based on 115 individual customs records matching Cameg.
- 5.Supplier Verification: Cameg sources from 37 verified Indian suppliers across 489 formulations, confirmed from customs records.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
8 Products Tracked
5 therapeutic categories
Expert-Reviewed
By pharmaceutical trade specialists
Data Source & Methodology
Trade data sourced from Indian Customs (DGFT) export shipment records. Values represent FOB export value in USD. For current shipment-level data, contact TransData Nexus.