Brown AND Burk Philippines Inc
Pharmaceutical Importer · Philippines · Diabetes & Endocrine Focus · $887.9K Total Trade · DGFT Verified
Brown AND Burk Philippines Inc is a pharmaceutical importer based in Philippines with a total trade value of $887.9K across 2 products in 1 therapeutic categories. Based on 25 verified import shipments from Indian Customs (DGFT) records, the company actively imports across multiple product segments. Brown AND Burk Philippines Inc sources from 1 verified Indian supplier, with Micro Labs Limited accounting for 100.0% of imports.
Brown AND Burk Philippines Inc — Import Portfolio & Supplier Network
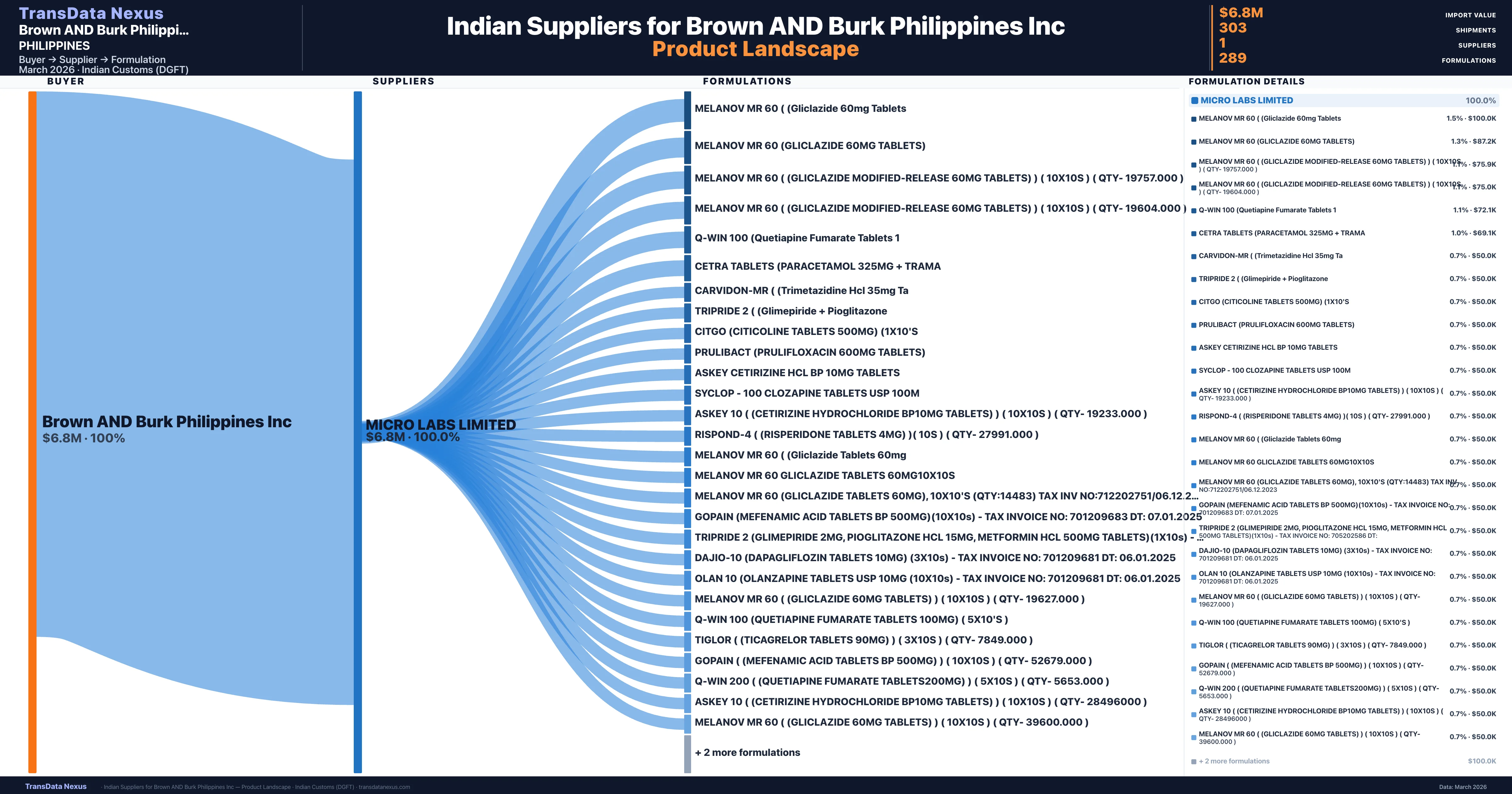
Who Are the Verified Indian Suppliers to Brown AND Burk Philippines Inc?
Customs-verified supplier relationships from Indian DGFT records
| Supplier | Value | Shipments | Share |
|---|---|---|---|
| Micro Labs Limited | $6.8M | 303 | 100.0% |
Brown AND Burk Philippines Inc sources from 1 verified Indian supplier across 289 distinct formulations. The sourcing is highly concentrated — Micro Labs Limited accounts for 100.0% of total imports, indicating a strategic single-source relationship.
What Formulations Does Brown AND Burk Philippines Inc Import?
| Formulation | Value | Ships |
|---|---|---|
| Melanov mr 60 ( (gliclazide 60MG | $100.0K | 2 |
| Melanov mr 60 (gliclazide 60MG tablets) | $87.2K | 2 |
| Melanov mr 60 ( (gliclazide modified-release 60MG tablets) ) ( 10x10s ) ( qty- 19757.000 ) | $75.9K | 3 |
| Melanov mr 60 ( (gliclazide modified-release 60MG tablets) ) ( 10x10s ) ( qty- 19604.000 ) | $75.0K | 3 |
| Q-win 100 (quetiapine fumarate tablets | $72.1K | 2 |
| Cetra tablets (paracetamol 325MG + | $69.1K | 2 |
| Carvidon-mr ( (trimetazidine hcl 35MG | $50.0K | 1 |
| Tripride 2 ( (glimepiride + | $50.0K | 1 |
| Citgo (citicoline tablets 500MG) | $50.0K | 1 |
| Prulibact (prulifloxacin 600MG tablets) | $50.0K | 1 |
| Askey cetirizine hcl BP 10MG | $50.0K | 1 |
| Syclop - 100 clozapine tablets USP | $50.0K | 1 |
| Askey 10 ( (cetirizine hydrochloride bp10mg tablets) ) ( 10x10s ) ( qty- 19233.000 ) | $50.0K | 1 |
| Rispond-4 ( (risperidone tablets 4MG) )( 10s ) ( qty- 27991.000 ) | $50.0K | 1 |
| Melanov mr 60 ( (gliclazide tablets 60MG | $50.0K | 1 |
Brown AND Burk Philippines Inc imports 289 distinct pharmaceutical formulations. Showing top 15 by value. For full formulation-level data, contact TransData Nexus.
What Products Does Brown AND Burk Philippines Inc Import?
Top Products by Import Value
Brown AND Burk Philippines Inc Therapeutic Categories — 1 Specializations
Brown AND Burk Philippines Inc imports across 1 therapeutic categories, with Diabetes & Endocrine (100.0%), representing the largest segments. The portfolio is concentrated — top 5 products = 100% of total imports.
Diabetes & Endocrine
2 products · 100.0% · $887.9K
Import Portfolio — Top 2 by Import Value
| # | Product | Category | Value | Ships | Share | Rk |
|---|---|---|---|---|---|---|
| 1 | Glimepiride | Diabetes & Endocrine | $460.0K | 13 | 0.6% | 12 |
| 2 | Pioglitazone | Diabetes & Endocrine | $427.9K | 12 | 1.1% | 19 |
Brown AND Burk Philippines Inc imports 2 pharmaceutical products across 1 categories into Philippines totaling $887.9K.
Key Metrics
Top Categories
Indian Suppliers
Related Trade Data
Need Detailed Data?
Shipment-level records, supplier connections & pricing for Brown AND Burk Philippines Inc.
Request DemoBrown AND Burk Philippines Inc — Corporate Profile & Information
Company type, headquarters, distribution network, and industry role
1Company Overview
Brown & Burk Philippines Inc. is a privately held pharmaceutical company headquartered in Makati City, Metro Manila, Philippines. Established on May 23, 2005, the company is dedicated to the professional and profitable distribution of pharmaceutical products within the country. Its mission is to provide Filipinos with international-quality medicines manufactured at US-FDA facilities, ensuring affordability and accessibility. The company emphasizes values such as integrity, excellence, discipline, fairness, and respect in its operations.
Brown & Burk Philippines Inc. operates as a pharmaceutical importer and distributor, focusing on branded generic pharmaceuticals. It collaborates with manufacturers like Micro Labs Limited, an Indian multinational pharmaceutical company, to source its product portfolio. The company markets its products to various healthcare professionals, including internists, family practitioners, general practitioners, psychiatrists, neurologists, gastroenterologists, endocrinologists, cardiologists, and specialists in otolaryngology, gynecology, and dermatology across the Philippines. (philjobnet.gov.ph)
2Distribution Network
Brown & Burk Philippines Inc. operates from its headquarters located at Unit 501, SEDCCO I Building, 120 Rada Street, Legaspi Village, Makati City, Metro Manila. (philjobnet.gov.ph) The company collaborates with distribution partners, such as RBC-MDC Corporation, to ensure its products reach a wide network of healthcare facilities, including leading chain pharmacies, independent pharmacies, private and government hospitals, and medical centers across the Philippines. (philjobnet.gov.ph) While specific details about its logistics capabilities and geographic coverage beyond the Philippines are not publicly disclosed, the company's extensive distribution network within the country indicates a robust logistics framework.
3Industry Role
In the Philippines' pharmaceutical supply chain, Brown & Burk Philippines Inc. functions primarily as a pharmaceutical importer and distributor. By sourcing branded generic pharmaceuticals from manufacturers like Micro Labs Limited, the company plays a crucial role in making these products accessible to a broad spectrum of healthcare providers and patients across the nation. Its focus on branded generics allows it to offer cost-effective alternatives to branded drugs, thereby enhancing the affordability and availability of essential medications in the Philippine market.
Supplier Relationship Intelligence — Brown AND Burk Philippines Inc
Sourcing concentration, supply chain resilience, and strategic implications
1Sourcing Concentration Analysis
Brown & Burk Philippines Inc. demonstrates a high degree of supplier concentration, sourcing its pharmaceutical products exclusively from Micro Labs Limited. This single-source dependency could pose risks related to supply chain disruptions, such as production delays or quality control issues. However, the company's consistent importation of 25 shipments valued at $888,000 USD over the period from 2022 to 2026 suggests a stable and ongoing relationship with Micro Labs Limited. The substantial import value and frequency of shipments indicate a strong and reliable partnership, which may mitigate potential risks associated with supplier concentration.
2Supply Chain Resilience
The resilience of Brown & Burk Philippines Inc.'s supply chain is closely tied to its exclusive sourcing from Micro Labs Limited. While this concentrated sourcing strategy may expose the company to risks associated with supply chain disruptions, such as production delays or quality control issues, the consistent importation of 25 shipments valued at $888,000 USD over the period from 2022 to 2026 suggests a stable and ongoing relationship with Micro Labs Limited. This stability may help mitigate potential risks associated with supplier concentration. However, the lack of publicly available information regarding backup suppliers, formulation diversity, shipping route exposure, and the regulatory compliance status of Micro Labs Limited's facilities limits a comprehensive assessment of the supply chain's overall resilience.
3Strategic Implications
The sourcing pattern of Brown & Burk Philippines Inc., characterized by exclusive procurement from Micro Labs Limited, positions the company as a significant player in the branded generic pharmaceutical market within the Philippines. This strategic choice allows the company to offer a focused product portfolio, potentially enhancing brand recognition and operational efficiency. For Indian exporters seeking to become alternative suppliers, this presents an opportunity to diversify Brown & Burk's product offerings and reduce dependency on a single supplier. Establishing a relationship with Brown & Burk could involve demonstrating compliance with the Philippines' regulatory standards and offering competitive pricing to meet the company's quality and affordability criteria.
Importing Pharmaceuticals into Philippines — Regulatory Framework
Regulatory authority, GMP requirements, import licensing for Philippines
1Regulatory Authority & Framework
In the Philippines, the Food and Drug Administration (FDA) is the primary regulatory body overseeing the importation, registration, and distribution of pharmaceutical products. The FDA operates under the Department of Health (DOH) and is responsible for ensuring the safety, efficacy, and quality of health products entering the country. Key legislation governing pharmaceutical imports includes the FDA Act of 2009 (Republic Act No. 9711) and the Food, Drugs, and Devices Act (Republic Act No. 3720). These laws establish the FDA's authority to regulate all health products, issue permits, licenses, and product registrations, and enforce standards for pharmaceutical imports. (respicio.ph)
2Import Licensing & GMP
Import licensing requirements in the Philippines stipulate that all pharmaceutical products must be registered with the FDA before they can be imported, sold, or distributed. The FDA requires a valid License to Operate (LTO) for importers and a Certificate of Good Manufacturing Practice (GMP) Compliance for manufacturers. GMP certificates from recognized authorities, such as the European Union (EU), World Health Organization (WHO), and Pharmaceutical Inspection Co-operation Scheme (PIC/S), are generally accepted by the FDA. Additionally, importers must secure the necessary import permits and clearances from the Bureau of Customs (BOC) prior to importation. (respicio.ph)
3Quality & Labeling
Pharmaceutical products imported into the Philippines must comply with the FDA's quality standards, which include batch testing and stability requirements to ensure product safety and efficacy. Labeling must adhere to FDA guidelines, which typically require information such as the product's generic and brand names, dosage strength, dosage form, manufacturer's details, and importer's information. Labels must be in English and Filipino, and include the phrase "Imported by [importer's name]" and the suggested retail price. Serialization mandates may apply to facilitate traceability and prevent counterfeit products, though specific requirements can vary. (respicio.ph)
4Recent Regulatory Changes
Between 2024 and 2026, the Philippines FDA has implemented several regulatory changes affecting pharmaceutical imports. Notably, in August 2024, the FDA issued Administrative Order No. 2024-0012, outlining regulations for registering pharmaceutical products and Active Pharmaceutical Ingredients (APIs) intended exclusively for export. This order sets forth guidelines, requirements, and procedures for the registration process of export-only pharmaceutical products and APIs. Additionally, in November 2025, the Philippines published two new pharmaceutical regulations: the Implementing Guidelines of the WHO Collaborative Procedure on the Registration of Pharmaceutical Products Approved by Stringent Regulatory Authorities (SRAs) and the Classification of Pharmaceutical Products Intended for Human Use. These regulations aim to streamline the registration process and enhance the availability of essential medicines in the local market.
Brown AND Burk Philippines Inc — Procurement Pattern Analysis
Product strategy, sourcing profile, and market positioning
1Product Strategy
Brown & Burk Philippines Inc. focuses on therapeutic areas such as psychiatry, neurology, cardiology, endocrinology, antibiotics and analgesics, gastroenterology, and ophthalmology. This strategic focus aligns with the company's mission to provide Filipinos with international-quality medicines manufactured at US-FDA facilities at affordable prices. The emphasis on these therapeutic areas is driven by the market demand for effective treatments in these specialties, addressing prevalent health conditions in the Philippines. By concentrating on these segments, the company aims to meet the needs of healthcare professionals and patients, thereby enhancing its market presence and impact.
2Sourcing Profile
Brown & Burk Philippines Inc. employs a sourcing strategy that focuses on branded generic pharmaceuticals, primarily procuring products from Micro Labs Limited, an Indian multinational pharmaceutical company. This approach allows the company to offer cost-effective alternatives to branded drugs, enhancing the affordability and accessibility of essential medications in the Philippine market. The preference for branded generics aligns with the company's mission to provide international-quality medicines at affordable prices, ensuring that patients receive effective treatments without the high costs associated with branded pharmaceuticals.
3Market Positioning
Based on its product mix, Brown & Burk Philippines Inc. serves multiple segments of the Philippine pharmaceutical market, including retail pharmacies, hospitals, government tenders, and wholesale distribution. By offering a range of branded generic pharmaceuticals across various therapeutic areas, the company caters to the diverse needs of healthcare providers and patients. Its strategic focus on affordability and quality positions it as a key player in enhancing the accessibility of essential medications across the country.
Seller's Guide — How to Become a Supplier to Brown AND Burk Philippines Inc
Opportunity assessment, qualifications, and practical approach strategy
1Opportunity Assessment
There is a realistic opportunity for new Indian suppliers to collaborate with Brown & Burk Philippines Inc. to diversify its product offerings and reduce dependency on a single supplier. Establishing a relationship with Brown & Burk could involve demonstrating compliance with the Philippines' regulatory standards and offering competitive pricing to meet the company's quality and affordability criteria
Frequently Asked Questions — Brown AND Burk Philippines Inc
What products does Brown AND Burk Philippines Inc import from India?
Brown AND Burk Philippines Inc imports 2 pharmaceutical products across 1 categories. Top imports: Glimepiride ($460.0K), Pioglitazone ($427.9K).
Who supplies pharmaceuticals to Brown AND Burk Philippines Inc from India?
Brown AND Burk Philippines Inc sources from 1 verified Indian suppliers. The primary supplier is Micro Labs Limited (100.0% of imports, $6.8M).
What is Brown AND Burk Philippines Inc's total pharmaceutical import value?
Brown AND Burk Philippines Inc's total pharmaceutical import value from India is $887.9K, based on 25 verified shipments in Indian Customs (DGFT) data.
What therapeutic categories does Brown AND Burk Philippines Inc focus on?
Brown AND Burk Philippines Inc imports across 1 categories. The largest: Diabetes & Endocrine (100.0%).
Get Full Brown AND Burk Philippines Inc Import Intelligence
Access shipment-level details, supplier connections, pricing data, and competitive analysis. TransData Nexus provides verified Indian Customs (DGFT) data trusted by pharmaceutical trade professionals worldwide.
Official References & Regulatory Resources
Verify import regulations and drug registration requirements with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Buyer Matching: Brown AND Burk Philippines Inc identified across shipments using consignee name normalization.
- 2.Statistical Normalization: Shipment values are statistically normalized to ensure accurate market share representation.
- 3.Market Share: Calculated per product as Brown AND Burk Philippines Inc's capped value divided by total Indian exports for that product.
- 4.Shipment Count: Based on 25 individual customs records matching Brown AND Burk Philippines Inc.
- 5.Supplier Verification: Brown AND Burk Philippines Inc sources from 1 verified Indian suppliers across 289 formulations, confirmed from customs records.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
2 Products Tracked
1 therapeutic categories
Expert-Reviewed
By pharmaceutical trade specialists
Data Source & Methodology
Trade data sourced from Indian Customs (DGFT) export shipment records. Values represent FOB export value in USD. For current shipment-level data, contact TransData Nexus.