Brocacef Nl:eu HUB
Pharmaceutical Importer · Netherlands · Diuretics Focus · $1.7M Total Trade · DGFT Verified
Brocacef Nl:eu HUB is a pharmaceutical importer based in Netherlands with a total trade value of $1.7M across 2 products in 2 therapeutic categories. Based on 65 verified import shipments from Indian Customs (DGFT) records, the company actively imports across multiple product segments. Brocacef Nl:eu HUB sources from 1 verified Indian supplier, with Intas Pharmaceuticals Limited accounting for 100.0% of imports.
Brocacef Nl:eu HUB — Import Portfolio & Supplier Network
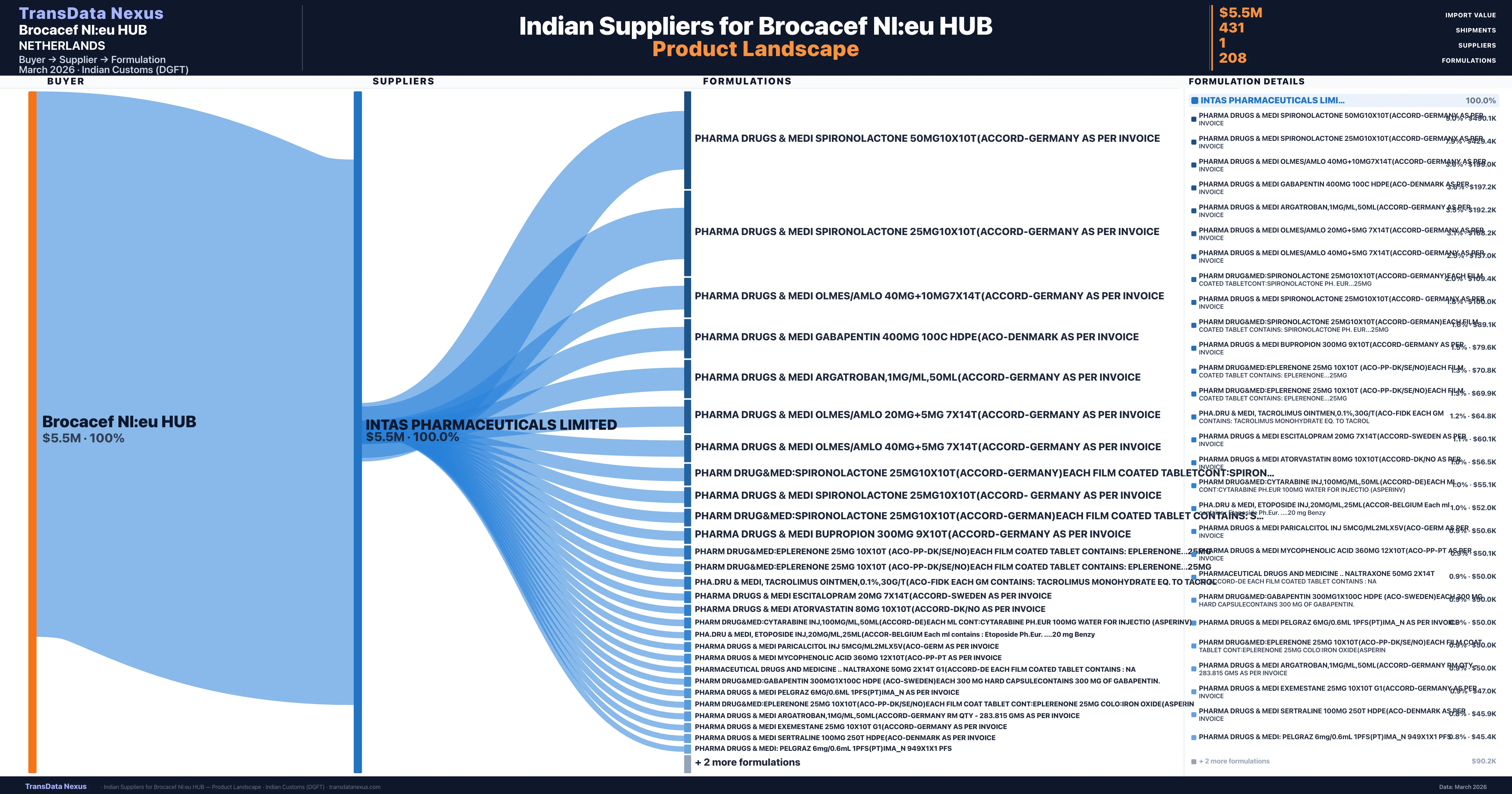
Who Are the Verified Indian Suppliers to Brocacef Nl:eu HUB?
Customs-verified supplier relationships from Indian DGFT records
| Supplier | Value | Shipments | Share |
|---|---|---|---|
| Intas Pharmaceuticals Limited | $5.5M | 431 | 100.0% |
Brocacef Nl:eu HUB sources from 1 verified Indian supplier across 208 distinct formulations. The sourcing is highly concentrated — Intas Pharmaceuticals Limited accounts for 100.0% of total imports, indicating a strategic single-source relationship.
What Formulations Does Brocacef Nl:eu HUB Import?
| Formulation | Value | Ships |
|---|---|---|
| Pharma drugs & medi spironolactone 50mg10x10t(accord-germany as per | $490.1K | 19 |
| Pharma drugs & medi spironolactone 25mg10x10t(accord-germany as per | $429.4K | 16 |
| Pharma drugs & medi olmes/amlo 40MG+10mg7x14t(accord-germany as per | $199.0K | 10 |
| Pharma drugs & medi gabapentin 400MG 100c hdpe(aco-denmark as per | $197.2K | 7 |
| Pharma drugs & medi argatroban,1MG/ML,50ML(accord-germany as per | $192.2K | 8 |
| Pharma drugs & medi olmes/amlo 20MG+5MG 7x14t(accord-germany as per | $168.2K | 14 |
| Pharma drugs & medi olmes/amlo 40MG+5MG 7x14t(accord-germany as per | $137.0K | 6 |
| Pharm drug&med:spironolactone 25mg10x10t(accord-germany)Each film coated tabletcont:spironolactone ph. eur...25MG | $109.4K | 3 |
| Pharma drugs & medi spironolactone 25mg10x10t(accord- germany as per | $100.0K | 4 |
| Pharm drug&med:spironolactone 25mg10x10t(accord-german)Each film coated tablet contains: spironolactone ph. eur...25MG | $89.1K | 2 |
| Pharma drugs & medi bupropion 300MG 9x10t(accord-germany as per | $79.6K | 4 |
| Pharm drug&med:eplerenone 25MG 10x10t (aco-pp-dk/se/no)Each film coated tablet contains: eplerenone...25MG | $70.8K | 2 |
| Pharm drug&med:eplerenone 25MG 10x10t (aco-pp-dk/se/no)Each film coated tablet contains: eplerenone...25MG | $69.9K | 2 |
| Pha.dru & medi, tacrolimus ointmen,0.1%,30g/t(aco-fidk Each GM contains: tacrolimus monohydrate eq. to | $64.8K | 5 |
| Pharma drugs & medi escitalopram 20MG 7x14t(accord-sweden as per | $60.1K | 6 |
Brocacef Nl:eu HUB imports 208 distinct pharmaceutical formulations. Showing top 15 by value. For full formulation-level data, contact TransData Nexus.
What Products Does Brocacef Nl:eu HUB Import?
Top Products by Import Value
Brocacef Nl:eu HUB Therapeutic Categories — 2 Specializations
Brocacef Nl:eu HUB imports across 2 therapeutic categories, with Diuretics (95.9%), Advanced Oncology (4.1%), representing the largest segments. The portfolio is concentrated — top 5 products = 100% of total imports.
Diuretics
1 products · 95.9% · $1.6M
Advanced Oncology
1 products · 4.1% · $70.0K
Import Portfolio — Top 2 by Import Value
| # | Product | Category | Value | Ships | Share | Rk |
|---|---|---|---|---|---|---|
| 1 | Spironolactone | Diuretics | $1.6M | 57 | 0.8% | 11 |
| 2 | Topotecan | Advanced Oncology | $70.0K | 8 | 1.9% | 11 |
Brocacef Nl:eu HUB imports 2 pharmaceutical products across 2 categories into Netherlands totaling $1.7M.
Key Metrics
Top Categories
Indian Suppliers
Related Trade Data
Need Detailed Data?
Shipment-level records, supplier connections & pricing for Brocacef Nl:eu HUB.
Request DemoBrocacef Nl:eu HUB — Corporate Profile & Information
Company type, headquarters, distribution network, and industry role
1Company Overview
Brocacef Nl:eu HUB is a pharmaceutical importer and buyer based in the Netherlands, operating under the Brocacef Group, which has a rich history dating back to 1798 when founded by Willem Brocades in Meppel. The company has evolved into a significant player in the Dutch pharmaceutical sector, providing comprehensive healthcare logistics and services to a diverse clientele, including pharmacies, hospitals, general practitioners, nursing homes, suppliers, healthcare insurers, and private individuals. (brocacef.nl)
As part of the Brocacef Group, Brocacef Nl:eu HUB contributes to the organization's extensive network, which includes approximately 5,300 employees and a nationwide presence with around 350 BENU pharmacies. The group's annual revenue is approximately €2.8 billion, reflecting its substantial impact on the healthcare industry. (bitscale.ai)
2Distribution Network
Brocacef Nl:eu HUB operates a robust distribution network within the Netherlands, with a significant warehouse located in Ede. This facility enables efficient storage and distribution of pharmaceutical products across the country. The company's logistics capabilities are designed to ensure timely and reliable delivery to various healthcare providers, including pharmacies, hospitals, and nursing homes. While the primary focus is on the Dutch market, Brocacef's affiliation with the German PHOENIX Group suggests potential for broader European distribution, leveraging the group's extensive logistics infrastructure. (brocacef.nl)
3Industry Role
Within the Netherlands' pharmaceutical supply chain, Brocacef Nl:eu HUB functions as a primary wholesaler and distributor. The company imports finished pharmaceutical formulations from international suppliers, including India, and supplies these products to various healthcare providers. This role is crucial in ensuring the availability of a wide range of pharmaceutical products to meet the diverse needs of the Dutch healthcare system. Additionally, Brocacef's operations encompass the management of its own pharmacy chain, BENU, serving the consumer market and further solidifying its position in the pharmaceutical sector. (brocacef.nl)
Supplier Relationship Intelligence — Brocacef Nl:eu HUB
Sourcing concentration, supply chain resilience, and strategic implications
1Sourcing Concentration Analysis
Brocacef Nl:eu HUB's sourcing strategy exhibits a high degree of concentration, with a total import value from India amounting to $1.7 million USD across 65 shipments. The primary supplier is INTAS Pharmaceuticals Limited, accounting for 100% of these imports. This single-source dependency may pose risks related to supply chain disruptions, such as production delays or geopolitical issues affecting trade routes. However, the consistent volume of shipments suggests a stable and well-established relationship between Brocacef and INTAS Pharmaceuticals, potentially mitigating some of these risks. The portfolio concentration, with the top five products comprising 100% of the imports, indicates a focused product strategy, which could be advantageous in terms of inventory management and supplier negotiations.
2Supply Chain Resilience
The resilience of Brocacef Nl:eu HUB's supply chain is closely tied to its exclusive reliance on INTAS Pharmaceuticals Limited for imports from India. The absence of backup suppliers for these products introduces vulnerability to potential supply chain disruptions. The limited diversity in imported formulations, with only two products across two therapeutic categories, further constrains flexibility in responding to market changes or supply issues. Additionally, the concentration of shipments through a single supplier and potentially limited shipping routes may expose the supply chain to risks associated with transportation disruptions or regulatory changes. Ensuring that INTAS Pharmaceuticals complies with international quality standards and maintains robust manufacturing practices is essential to uphold the integrity and reliability of the supply chain.
3Strategic Implications
Brocacef Nl:eu HUB's concentrated sourcing strategy positions the company to leverage strong supplier relationships, potentially securing favorable terms and ensuring consistent product quality. However, the lack of supplier diversification may limit the company's ability to adapt to market fluctuations or supply chain disruptions. For Indian exporters, this presents an opportunity to become alternative suppliers by offering competitive pricing, reliable delivery schedules, and adherence to international quality standards. Diversifying Brocacef's supplier base could enhance supply chain resilience and provide a more flexible response to market demands.
Importing Pharmaceuticals into Netherlands — Regulatory Framework
Regulatory authority, GMP requirements, import licensing for Netherlands
1Regulatory Authority & Framework
In the Netherlands, the regulatory authority overseeing pharmaceutical imports is the Dutch Medicines Evaluation Board (CBG-MEB), operating under the Ministry of Health, Welfare, and Sport (VWS). The primary legislation governing pharmaceutical imports is the Medicines Act (Geneesmiddelenwet), which aligns with European Union directives to ensure the safety, efficacy, and quality of medicinal products. The marketing authorization pathway for Indian generics involves obtaining a marketing authorization from the CBG-MEB, demonstrating compliance with EU standards. This process includes the submission of comprehensive data on the product's quality, safety, and efficacy, as well as compliance with Good Manufacturing Practice (GMP) standards. (business.gov.nl)
2Import Licensing & GMP
Importers of pharmaceutical products into the Netherlands must obtain a wholesale distribution authorization from the CBG-MEB, ensuring compliance with the European guidelines on Good Distribution Practice (GDP) of medicinal products for human use. Additionally, importers must ensure that their suppliers hold valid GMP certificates recognized by the EU, WHO, or PIC/S. These certifications confirm that the manufacturing processes meet the required quality standards. For Indian exporters, obtaining GMP certification from recognized authorities is essential to facilitate the importation of their products into the Netherlands. (business.gov.nl)
3Quality & Labeling
Imported pharmaceutical products must undergo batch testing to verify their quality and compliance with safety standards. Stability requirements ensure that products maintain their efficacy and safety throughout their shelf life. Labeling must be in Dutch and include all necessary information, such as dosage instructions, side effects, and storage conditions, to ensure proper use by consumers and healthcare professionals. Serialization mandates may apply to facilitate traceability and prevent counterfeit products from entering the market. Indian exporters should ensure that their products meet these requirements to comply with Dutch regulations and facilitate smooth market entry. (business.gov.nl)
4Recent Regulatory Changes
Effective from 1 July 2024, significant changes have been implemented regarding the declaration of pharmaceutical products and active substances for import into the Netherlands. Importers are now required to include specific document codes in their declarations, such as 1400 for marketing authorization and 1450 for API registration. Failure to include the appropriate codes will result in declaration rejections. This change emphasizes the need for importers and exporters to stay updated with regulatory requirements to ensure compliance and avoid disruptions in the supply chain.
Brocacef Nl:eu HUB — Procurement Pattern Analysis
Product strategy, sourcing profile, and market positioning
1Product Strategy
Brocacef Nl:eu HUB's product strategy focuses on importing finished pharmaceutical formulations in the therapeutic areas of diuretics and advanced oncology. The substantial import value of spironolactone, a diuretic, indicates a strong market demand for such products in the Netherlands. The inclusion of advanced oncology products suggests an alignment with the growing need for specialized cancer treatments. This strategic focus allows Brocacef to cater to critical healthcare needs, positioning itself as a key supplier in these therapeutic categories.
2Sourcing Profile
Brocacef Nl:eu HUB's sourcing strategy is centered on importing generic pharmaceutical formulations from India, particularly in the diuretics and advanced oncology categories. The exclusive reliance on INTAS Pharmaceuticals Limited for these imports indicates a focused approach to procurement. This strategy may be driven by considerations such as cost-effectiveness, product quality, and supplier reliability. For Indian exporters, understanding Brocacef's sourcing preferences and aligning with their quality standards and regulatory requirements is crucial to establish a successful partnership.
3Market Positioning
Based on its product mix, Brocacef Nl:eu HUB serves the wholesale distribution segment of the Netherlands market. By importing and supplying pharmaceutical products to various healthcare providers, including pharmacies, hospitals, and nursing homes, the company plays a pivotal role in ensuring the availability of essential medications across the country. Additionally, through its BENU pharmacy chain, Brocacef extends its reach to the retail pharmacy segment, offering consumers direct access to pharmaceutical care.
Seller's Guide — How to Become a Supplier to Brocacef Nl:eu HUB
Opportunity assessment, qualifications, and practical approach strategy
1Opportunity Assessment
There is a realistic opportunity for new Indian suppliers to collaborate with Brocacef Nl:eu HUB, especially if they can offer products that complement the existing portfolio or provide competitive advantages in terms of pricing, quality, or innovation. Identifying gaps in Brocacef's current sourcing, such as unmet therapeutic needs or emerging market trends, can present avenues for new suppliers to enter the market. Establishing a diversified supplier base can enhance Brocacef's supply chain resilience and responsiveness to market demands.
2Requirements & Qualifications
Indian exporters aiming to supply Brocacef Nl:eu HUB and the broader Netherlands market must ensure that their products meet the regulatory requirements set by the Dutch Medicines Evaluation Board (CBG-MEB). This includes obtaining marketing authorization, adhering to Good Manufacturing Practice (GMP) standards recognized by the EU, WHO, or PIC/S, and ensuring compliance with labeling
Frequently Asked Questions — Brocacef Nl:eu HUB
What products does Brocacef Nl:eu HUB import from India?
Brocacef Nl:eu HUB imports 2 pharmaceutical products across 2 categories. Top imports: Spironolactone ($1.6M), Topotecan ($70.0K).
Who supplies pharmaceuticals to Brocacef Nl:eu HUB from India?
Brocacef Nl:eu HUB sources from 1 verified Indian suppliers. The primary supplier is Intas Pharmaceuticals Limited (100.0% of imports, $5.5M).
What is Brocacef Nl:eu HUB's total pharmaceutical import value?
Brocacef Nl:eu HUB's total pharmaceutical import value from India is $1.7M, based on 65 verified shipments in Indian Customs (DGFT) data.
What therapeutic categories does Brocacef Nl:eu HUB focus on?
Brocacef Nl:eu HUB imports across 2 categories. The largest: Diuretics (95.9%), Advanced Oncology (4.1%).
Get Full Brocacef Nl:eu HUB Import Intelligence
Access shipment-level details, supplier connections, pricing data, and competitive analysis. TransData Nexus provides verified Indian Customs (DGFT) data trusted by pharmaceutical trade professionals worldwide.
Official References & Regulatory Resources
Verify import regulations and drug registration requirements with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Buyer Matching: Brocacef Nl:eu HUB identified across shipments using consignee name normalization.
- 2.Statistical Normalization: Shipment values are statistically normalized to ensure accurate market share representation.
- 3.Market Share: Calculated per product as Brocacef Nl:eu HUB's capped value divided by total Indian exports for that product.
- 4.Shipment Count: Based on 65 individual customs records matching Brocacef Nl:eu HUB.
- 5.Supplier Verification: Brocacef Nl:eu HUB sources from 1 verified Indian suppliers across 208 formulations, confirmed from customs records.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
2 Products Tracked
2 therapeutic categories
Expert-Reviewed
By pharmaceutical trade specialists
Data Source & Methodology
Trade data sourced from Indian Customs (DGFT) export shipment records. Values represent FOB export value in USD. For current shipment-level data, contact TransData Nexus.