Boston Oncology Arabia
Pharmaceutical Importer · Saudi Arabia · Cardiovascular Focus · $29.2M Total Trade · DGFT Verified
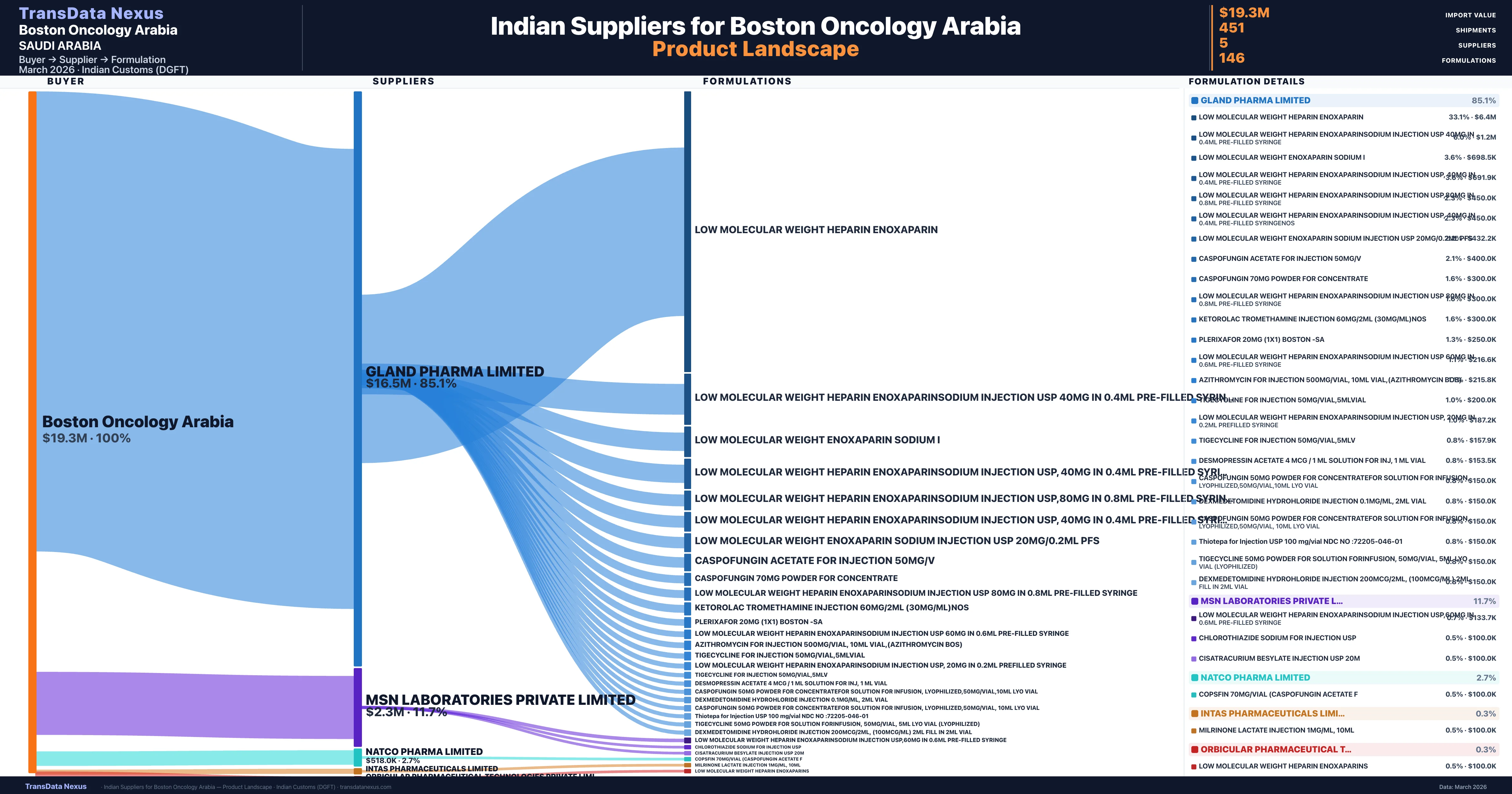
Boston Oncology Arabia is a pharmaceutical importer based in Saudi Arabia with a total trade value of $29.2M across 5 products in 4 therapeutic categories. Based on 588 verified import shipments from Indian Customs (DGFT) records, the company actively imports across multiple product segments. Boston Oncology Arabia sources from 5 verified Indian suppliers, with Gland Pharma Limited accounting for 85.1% of imports.
Boston Oncology Arabia — Import Portfolio & Supplier Network
Who Are the Verified Indian Suppliers to Boston Oncology Arabia?
Customs-verified supplier relationships from Indian DGFT records
| Supplier | Value | Shipments | Share |
|---|---|---|---|
| Gland Pharma Limited | $16.5M | 349 | 85.1% |
| Msn Laboratories Private Limited | $2.3M | 81 | 11.7% |
| Natco Pharma Limited | $518.0K | 19 | 2.7% |
| Intas Pharmaceuticals Limited | $50.0K | 1 | 0.3% |
| Orbicular Pharmaceutical Technologies Private Limi | $50.0K | 1 | 0.3% |
Boston Oncology Arabia sources from 5 verified Indian suppliers across 146 distinct formulations. The sourcing is highly concentrated — Gland Pharma Limited accounts for 85.1% of total imports, indicating a strategic single-source relationship.
What Formulations Does Boston Oncology Arabia Import?
| Formulation | Value | Ships |
|---|---|---|
| Low molecular weight heparin | $6.4M | 130 |
| Low molecular weight heparin enoxaparinsodium injection USP 40MG in 0.4ML pre-filled | $1.2M | 24 |
| Low molecular weight enoxaparin sodium | $698.5K | 16 |
| Low molecular weight heparin enoxaparinsodium injection USP, 40MG in 0.4ML pre-filled | $691.9K | 14 |
| Low molecular weight heparin enoxaparinsodium injection USP,80MG in 0.8ML pre-filled | $450.0K | 9 |
| Low molecular weight heparin enoxaparinsodium injection USP, 40MG in 0.4ML pre-filled | $450.0K | 9 |
| Low molecular weight enoxaparin sodium injection USP 20MG/0.2ML | $432.2K | 11 |
| Caspofungin acetate for injection | $400.0K | 8 |
| Caspofungin 70MG powder for | $300.0K | 6 |
| Low molecular weight heparin enoxaparinsodium injection USP 80MG in 0.8ML pre-filled | $300.0K | 6 |
| Ketorolac tromethamine injection 60MG/2ML (30MG/ML | $300.0K | 6 |
| Plerixafor 20MG (1x1) boston | $250.0K | 5 |
| Low molecular weight heparin enoxaparinsodium injection USP 60MG in 0.6ML pre-filled | $216.6K | 5 |
| Azithromycin for injection 500MG/vial, 10ML vial,(azithromycin bos) | $215.8K | 5 |
| Tigecycline for injection 50MG/vial | $200.0K | 4 |
Boston Oncology Arabia imports 146 distinct pharmaceutical formulations. Showing top 15 by value. For full formulation-level data, contact TransData Nexus.
What Products Does Boston Oncology Arabia Import?
Top Products by Import Value
Boston Oncology Arabia Therapeutic Categories — 4 Specializations
Boston Oncology Arabia imports across 4 therapeutic categories, with Cardiovascular (81.9%), Medical Devices & Diagnostics (16.3%), Immunosuppressants (1.0%) representing the largest segments. The portfolio is concentrated — top 5 products = 100% of total imports.
Cardiovascular
2 products · 81.9% · $23.9M
Medical Devices & Diagnostics
1 products · 16.3% · $4.8M
Immunosuppressants
1 products · 1.0% · $300.0K
Advanced Oncology
1 products · 0.8% · $222.3K
Import Portfolio — Top 5 by Import Value
| # | Product | Category | Value | Ships | Share | Rk |
|---|---|---|---|---|---|---|
| 1 | Enoxaparin | Cardiovascular | $12.8M | 255 | 6.1% | 2 |
| 2 | Heparin | Cardiovascular | $11.2M | 223 | 3.2% | 2 |
| 3 | Syringe | Medical Devices & Diagnostics | $4.8M | 95 | 2.6% | 3 |
| 4 | Everolimus | Immunosuppressants | $300.0K | 6 | 0.8% | 6 |
| 5 | Dasatinib | Advanced Oncology | $222.3K | 9 | 1.4% | 17 |
Boston Oncology Arabia imports 5 pharmaceutical products across 4 categories into Saudi Arabia totaling $29.2M.
Key Metrics
Top Categories
Indian Suppliers
Related Trade Data
Need Detailed Data?
Shipment-level records, supplier connections & pricing for Boston Oncology Arabia.
Request DemoBoston Oncology Arabia — Corporate Profile & Information
Company type, headquarters, distribution network, and industry role
1Company Overview
Boston Oncology Arabia is a Saudi Arabian pharmaceutical company specializing in the importation and distribution of finished pharmaceutical formulations. Operating as a subsidiary of Boston Oncology, headquartered in Cambridge, Massachusetts, the company focuses on providing high-quality medicines to the Saudi market. Boston Oncology Arabia has established itself as a significant player in the Kingdom's pharmaceutical sector, particularly in the oncology and critical care segments. The company's operations are aligned with Saudi Arabia's Vision 2030, aiming to enhance local manufacturing capabilities and reduce dependency on imports.
2Distribution Network
Boston Oncology Arabia's distribution network is centered in Riyadh, with its main office located at Roshn Front Gate 8, Building N8, 1st floor, Airport Street, Sedra District, Riyadh, 13413, Saudi Arabia. The company operates a state-of-the-art GMP-certified manufacturing facility in Sudair Industrial City, which has an annual vial capacity of over 30 million units. This facility enables Boston Oncology Arabia to efficiently supply the Saudi market and supports its commitment to local manufacturing and distribution.
3Industry Role
In Saudi Arabia's pharmaceutical supply chain, Boston Oncology Arabia functions primarily as a pharmaceutical importer and distributor. The company imports finished pharmaceutical formulations from international suppliers, particularly from India, and distributes these products to various healthcare providers across the Kingdom. By focusing on the importation of finished products, Boston Oncology Arabia plays a crucial role in ensuring the availability of essential medicines, especially in the oncology and critical care sectors. This approach aligns with the company's strategy to enhance local access to high-quality pharmaceuticals.
Supplier Relationship Intelligence — Boston Oncology Arabia
Sourcing concentration, supply chain resilience, and strategic implications
1Sourcing Concentration Analysis
Boston Oncology Arabia's sourcing strategy exhibits a high degree of concentration, with a significant portion of its imports originating from a single supplier, GLAND PHARMA LIMITED. Between 2022 and 2026, GLAND PHARMA LIMITED accounted for approximately 85.1% of the company's total imports, amounting to $16.5 million across 349 shipments. This heavy reliance on a single supplier could pose risks related to supply chain disruptions, such as production delays or quality control issues. However, the consistent volume of shipments and the long-standing relationship between Boston Oncology Arabia and GLAND PHARMA LIMITED suggest a stable and reliable partnership. The company's strategy to diversify its supplier base, as evidenced by engagements with other suppliers like MSN LABORATORIES PRIVATE LIMITED and NATCO PHARMA LIMITED, indicates a proactive approach to mitigating potential risks associated with supplier concentration.
2Supply Chain Resilience
Boston Oncology Arabia's supply chain resilience is bolstered by its diversified supplier base and the establishment of a local manufacturing facility in Sudair Industrial City. While the company imports a significant portion of its products from India, the local manufacturing capability allows for greater control over production timelines and quality standards. The partnership with GLAND PHARMA LIMITED, along with collaborations with other suppliers, provides a buffer against potential supply chain disruptions. Additionally, Boston Oncology Arabia's adherence to international quality standards, including GMP certifications, ensures that its products meet the highest safety and efficacy benchmarks. This combination of strategic supplier relationships and local manufacturing enhances the company's ability to maintain a resilient and responsive supply chain.
3Strategic Implications
Boston Oncology Arabia's sourcing pattern, characterized by a heavy reliance on GLAND PHARMA LIMITED, positions the company to benefit from economies of scale and potentially favorable pricing due to the volume of imports. This strategic choice may also foster a strong, collaborative relationship with the supplier, facilitating smoother logistics and consistent product quality. For Indian exporters seeking to become alternative suppliers, this presents an opportunity to diversify Boston Oncology Arabia's supplier base by offering competitive pricing, high-quality products, and reliable delivery schedules. Engaging in partnerships that align with Boston Oncology Arabia's commitment to local manufacturing and Vision 2030 objectives could further strengthen these relationships.
Importing Pharmaceuticals into Saudi Arabia — Regulatory Framework
Regulatory authority, GMP requirements, import licensing for Saudi Arabia
1Regulatory Authority & Framework
The Saudi Food and Drug Authority (SFDA) is the primary regulatory body overseeing the safety, efficacy, and quality of pharmaceutical products in Saudi Arabia. The SFDA is responsible for the registration of pharmaceutical products, ensuring compliance with international standards, and monitoring the post-marketing surveillance of medicines. The Law of Pharmaceutical Establishments and Products (2023), issued under Royal Decree No. M/108, provides the legal framework governing the importation, registration, and distribution of pharmaceutical products in the Kingdom. This legislation aligns with international guidelines and is enforced by the SFDA to maintain high standards in the pharmaceutical sector. (sfda.gov.sa)
2Import Licensing & GMP
Import licensing in Saudi Arabia requires pharmaceutical companies to obtain approval from the SFDA before importing medicines. This process involves submitting detailed product information, including safety and efficacy data, to ensure compliance with Saudi regulations. Good Manufacturing Practice (GMP) certification is a critical requirement for both local and international manufacturers. The SFDA recognizes GMP certifications from reputable international bodies, such as the European Union (EU) GMP, World Health Organization (WHO) GMP, and Pharmaceutical Inspection Co-operation Scheme (PIC/S). Pharmaceutical companies must ensure that their manufacturing facilities meet these standards to gain approval for product registration and importation. (sfda.gov.sa)
3Quality & Labeling
Pharmaceutical products imported into Saudi Arabia must undergo batch testing to verify their quality, safety, and efficacy. The SFDA conducts routine checks and cooperates with central laboratories to monitor the quality of pharmaceutical products in the local market. Stability studies are required to ensure that products maintain their quality throughout their shelf life under various storage conditions. Labeling requirements include providing information in Arabic, detailing the product's name, composition, dosage instructions, and storage conditions. Serialization mandates are implemented to enhance traceability and prevent counterfeit products from entering the market. (sfda.gov.sa)
4Recent Regulatory Changes
Between 2024 and 2026, Saudi Arabia has implemented several regulatory changes affecting pharmaceutical imports. The Law of Pharmaceutical Establishments and Products (2023) introduced comprehensive regulations governing the importation, registration, and distribution of pharmaceutical products, aligning with international standards. The SFDA has also enhanced its post-marketing surveillance and pharmacovigilance activities to ensure ongoing product safety and efficacy. These regulatory updates aim to strengthen the Kingdom's pharmaceutical sector, improve drug security, and support the goals of Vision 2030.
Boston Oncology Arabia — Procurement Pattern Analysis
Product strategy, sourcing profile, and market positioning
1Product Strategy
Boston Oncology Arabia's product strategy focuses on importing finished pharmaceutical formulations across various therapeutic categories, with a significant emphasis on cardiovascular medications. The company's top five imported products include Enoxaparin and Heparin, both essential in the cardiovascular segment, accounting for a combined total of $24 million in imports. The inclusion of medical devices like syringes and oncology treatments such as Everolimus and Dasatinib reflects a comprehensive approach to addressing critical healthcare needs in Saudi Arabia. This strategic focus aligns with the Kingdom's healthcare priorities and supports the objectives of Vision 2030 by enhancing access to essential and specialty medicines.
2Sourcing Profile
Boston Oncology Arabia's sourcing strategy is centered on importing finished pharmaceutical formulations from India, with a notable concentration on products from GLAND PHARMA LIMITED. This approach leverages India's robust pharmaceutical manufacturing capabilities and cost advantages. The company's preference for finished products over active pharmaceutical ingredients (APIs) indicates a focus on ready-to-use medicines that meet international quality standards. By sourcing from reputable Indian manufacturers, Boston Oncology Arabia ensures a reliable supply of high-quality pharmaceuticals to the Saudi market.
3Market Positioning
Based on its product mix, Boston Oncology Arabia serves multiple segments of the Saudi Arabian pharmaceutical market, including hospital systems, government health institutions, and potentially retail pharmacies. The company's focus on critical care and oncology products positions it as a key supplier to hospitals and specialized medical centers. Its participation in government tenders, such as those with the National Unified Procurement Company for Medical Supplies (NUPCO), indicates a strong presence in the public sector. Additionally, the importation of medical devices suggests potential engagement with retail pharmacies and clinics. This diversified approach enables Boston Oncology Arabia to address a broad spectrum of healthcare needs across the Kingdom.
Seller's Guide — How to Become a Supplier to Boston Oncology Arabia
Opportunity assessment, qualifications, and practical approach strategy
1Opportunity Assessment
There is a realistic opportunity for new Indian suppliers to engage with Boston Oncology Arabia, particularly in therapeutic areas where the company seeks to diversify its product offerings. The company's existing relationships with suppliers like MSN LABORATORIES PRIVATE LIMITED and NATCO PHARMA LIMITED demonstrate an openness to expanding its supplier base. Indian exporters can explore opportunities by offering high-quality products, competitive pricing, and reliable delivery schedules. Aligning with Boston Oncology Arabia's commitment to
Frequently Asked Questions — Boston Oncology Arabia
What products does Boston Oncology Arabia import from India?
Boston Oncology Arabia imports 5 pharmaceutical products across 4 categories. Top imports: Enoxaparin ($12.8M), Heparin ($11.2M), Syringe ($4.8M), Everolimus ($300.0K), Dasatinib ($222.3K).
Who supplies pharmaceuticals to Boston Oncology Arabia from India?
Boston Oncology Arabia sources from 5 verified Indian suppliers. The primary supplier is Gland Pharma Limited (85.1% of imports, $16.5M).
What is Boston Oncology Arabia's total pharmaceutical import value?
Boston Oncology Arabia's total pharmaceutical import value from India is $29.2M, based on 588 verified shipments in Indian Customs (DGFT) data.
What therapeutic categories does Boston Oncology Arabia focus on?
Boston Oncology Arabia imports across 4 categories. The largest: Cardiovascular (81.9%), Medical Devices & Diagnostics (16.3%), Immunosuppressants (1.0%).
Get Full Boston Oncology Arabia Import Intelligence
Access shipment-level details, supplier connections, pricing data, and competitive analysis. TransData Nexus provides verified Indian Customs (DGFT) data trusted by pharmaceutical trade professionals worldwide.
Official References & Regulatory Resources
Verify import regulations and drug registration requirements with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Buyer Matching: Boston Oncology Arabia identified across shipments using consignee name normalization.
- 2.Statistical Normalization: Shipment values are statistically normalized to ensure accurate market share representation.
- 3.Market Share: Calculated per product as Boston Oncology Arabia's capped value divided by total Indian exports for that product.
- 4.Shipment Count: Based on 588 individual customs records matching Boston Oncology Arabia.
- 5.Supplier Verification: Boston Oncology Arabia sources from 5 verified Indian suppliers across 146 formulations, confirmed from customs records.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
5 Products Tracked
4 therapeutic categories
Expert-Reviewed
By pharmaceutical trade specialists
Data Source & Methodology
Trade data sourced from Indian Customs (DGFT) export shipment records. Values represent FOB export value in USD. For current shipment-level data, contact TransData Nexus.