Borneo Pacific Pharmaceuticals Ltd
Pharmaceutical Importer · Papua New Guinea · Antimalarial & Antiparasitic Focus · $465.0K Total Trade · DGFT Verified
Borneo Pacific Pharmaceuticals Ltd is a pharmaceutical importer based in Papua New Guinea with a total trade value of $465.0K across 3 products in 1 therapeutic categories. Based on 11 verified import shipments from Indian Customs (DGFT) records, Borneo Pacific Pharmaceuticals Ltd is the #1 buyer in 1 product including Primaquine. Borneo Pacific Pharmaceuticals Ltd sources from 6 verified Indian suppliers, with Bdh Industries Limited accounting for 70.3% of imports.
Borneo Pacific Pharmaceuticals Ltd — Import Portfolio & Supplier Network
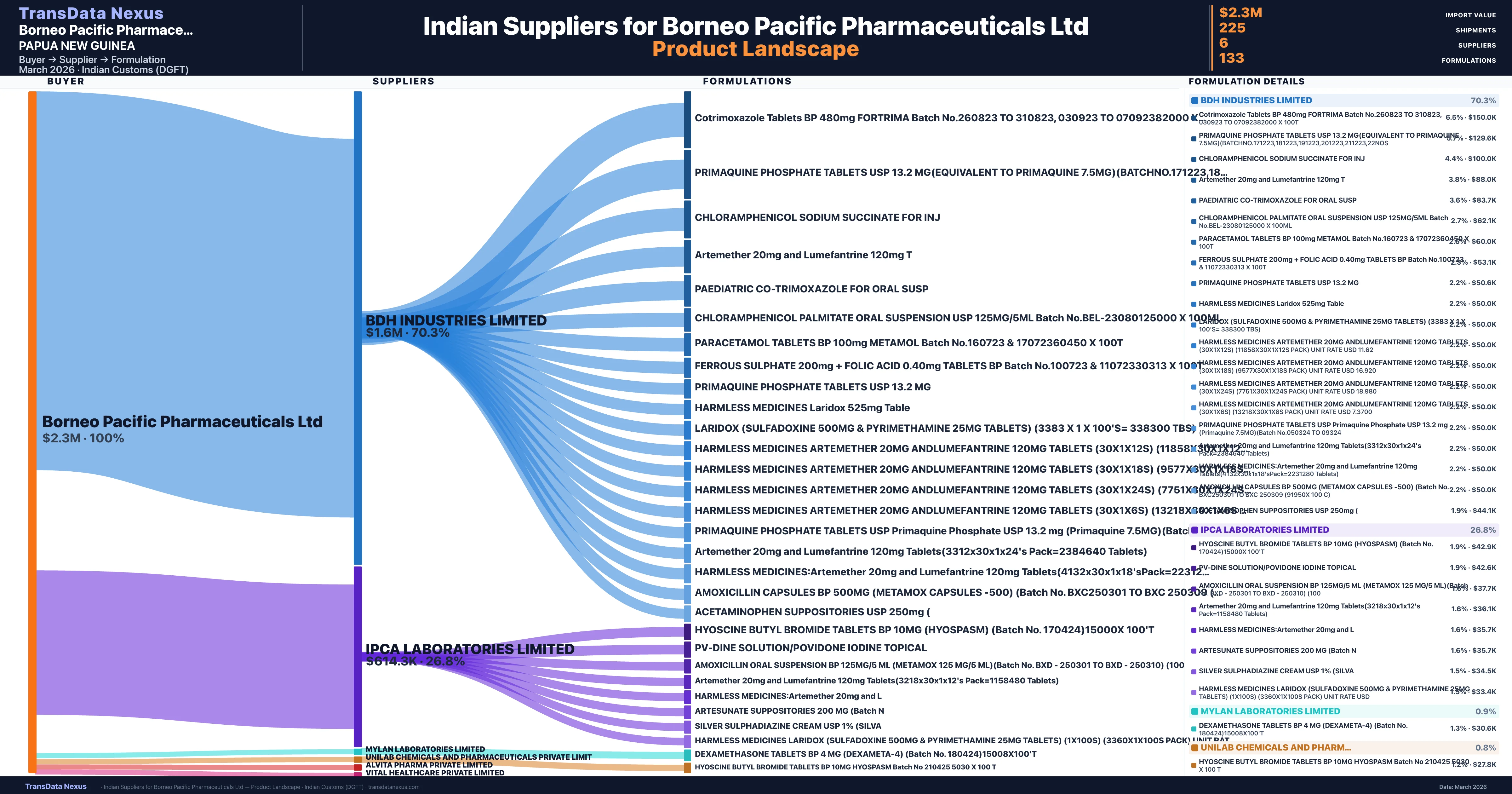
Who Are the Verified Indian Suppliers to Borneo Pacific Pharmaceuticals Ltd?
Customs-verified supplier relationships from Indian DGFT records
| Supplier | Value | Shipments | Share |
|---|---|---|---|
| Bdh Industries Limited | $1.6M | 193 | 70.3% |
| Ipca Laboratories Limited | $614.3K | 15 | 26.8% |
| Mylan Laboratories Limited | $20.4K | 1 | 0.9% |
| Unilab Chemicals And Pharmaceuticals Private Limit | $17.9K | 1 | 0.8% |
| Alvita Pharma Private Limited | $16.9K | 1 | 0.7% |
| Vital Healthcare Private Limited | $12.4K | 14 | 0.5% |
Borneo Pacific Pharmaceuticals Ltd sources from 6 verified Indian suppliers across 133 distinct formulations. The supply base is diversified across 6 suppliers, reducing single-source dependency risk.
What Formulations Does Borneo Pacific Pharmaceuticals Ltd Import?
| Formulation | Value | Ships |
|---|---|---|
| Cotrimoxazole tablets BP 480MG fortrima batch no.260823 to 310823, 030923 to 07092382000 x | $150.0K | 3 |
| Primaquine phosphate tablets USP 13.2 MG(equivalent to primaquine 7.5MG)(batchno.171223,181223,191223,201223,211223 | $129.6K | 3 |
| Chloramphenicol sodium succinate for INJ | $100.0K | 2 |
| Artemether 20MG and lumefantrine 120MG | $88.0K | 3 |
| Paediatric co-trimoxazole for oral | $83.7K | 3 |
| Chloramphenicol palmitate oral suspension USP 125MG/5ML batch no.bel-23080125000 x 100ML | $62.1K | 3 |
| Paracetamol tablets BP 100MG metamol batch no.160723 & 17072360450 x | $60.0K | 3 |
| Ferrous sulphate 200MG + folic acid 0.40MG tablets BP batch no.100723 & 11072330313 x | $53.1K | 3 |
| Primaquine phosphate tablets USP 13.2 MG | $50.6K | 3 |
| Harmless medicines laridox 525MG | $50.0K | 1 |
| Laridox (sulfadoxine 500MG & pyrimethamine 25MG tablets) (3383 x 1 x 100's= 338300 tbs) | $50.0K | 1 |
| Harmless medicines artemether 20MG andlumefantrine 120MG tablets (30x1x12s) (11858x30x1x12s Pack) unit rate usd | $50.0K | 1 |
| Harmless medicines artemether 20MG andlumefantrine 120MG tablets (30x1x18s) (9577x30x1x18s Pack) unit rate usd | $50.0K | 1 |
| Harmless medicines artemether 20MG andlumefantrine 120MG tablets (30x1x24s) (7751x30x1x24s Pack) unit rate usd | $50.0K | 1 |
| Harmless medicines artemether 20MG andlumefantrine 120MG tablets (30x1x6s) (13218x30x1x6s Pack) unit rate usd | $50.0K | 1 |
Borneo Pacific Pharmaceuticals Ltd imports 133 distinct pharmaceutical formulations. Showing top 15 by value. For full formulation-level data, contact TransData Nexus.
What Products Does Borneo Pacific Pharmaceuticals Ltd Import?
Top Products by Import Value
Borneo Pacific Pharmaceuticals Ltd Therapeutic Categories — 1 Specializations
Borneo Pacific Pharmaceuticals Ltd imports across 1 therapeutic categories, with Antimalarial & Antiparasitic (100.0%), representing the largest segments. The portfolio is concentrated — top 5 products = 100% of total imports.
Antimalarial & Antiparasitic
3 products · 100.0% · $465.0K
Import Portfolio — Top 3 by Import Value
| # | Product | Category | Value | Ships | Share | Rk |
|---|---|---|---|---|---|---|
| 1 | Primaquine | Antimalarial & Antiparasitic | $265.0K | 7 | 11.9% | 1 |
| 2 | Pyrimethamine | Antimalarial & Antiparasitic | $100.0K | 2 | 0.2% | 2 |
| 3 | Sulfadoxine | Antimalarial & Antiparasitic | $100.0K | 2 | 0.2% | 2 |
Borneo Pacific Pharmaceuticals Ltd imports 3 pharmaceutical products across 1 categories into Papua New Guinea totaling $465.0K. The company is the #1 buyer for 1 product: Primaquine.
Key Metrics
Top Categories
Indian Suppliers
Related Trade Data
Need Detailed Data?
Shipment-level records, supplier connections & pricing for Borneo Pacific Pharmaceuticals Ltd.
Request DemoBorneo Pacific Pharmaceuticals Ltd — Corporate Profile & Information
Company type, headquarters, distribution network, and industry role
1Company Overview
Borneo Pacific Pharmaceuticals Ltd, established in 1996 and operational since 1997, is a leading pharmaceutical importer and distributor based in Port Moresby, Papua New Guinea. The company is headquartered at Lot 22, Section 496, Hohola Ward, Gordons, Port Moresby, with a postal address of P.O. Box 1614, Port Moresby. As of 2026, Borneo Pacific Pharmaceuticals Ltd employs approximately 50 staff members and generates sales of around $5.33 million USD. The company operates as a primary wholesaler, sourcing a diverse range of pharmaceutical products to meet the healthcare needs of Papua New Guinea.
2Distribution Network
Borneo Pacific Pharmaceuticals Ltd maintains a centralized distribution hub in Port Moresby, which serves as the focal point for its logistics operations. While specific details about additional warehouse locations are not publicly disclosed, the company's strategic position in the capital city enables efficient distribution throughout Papua New Guinea. The firm has demonstrated its logistics capabilities by handling a total of 11 shipments from India between 2022 and 2026, indicating a well-established supply chain.
3Industry Role
In Papua New Guinea's pharmaceutical supply chain, Borneo Pacific Pharmaceuticals Ltd functions as a primary wholesaler. The company imports finished pharmaceutical formulations, including tablets, capsules, syrups, and injections, primarily from India. Between 2022 and 2026, Borneo Pacific Pharmaceuticals Ltd imported a total of 11 shipments from India, encompassing 133 unique formulations. This extensive portfolio underscores its significant role in supplying a wide array of pharmaceutical products to meet the diverse healthcare needs of the nation.
Supplier Relationship Intelligence — Borneo Pacific Pharmaceuticals Ltd
Sourcing concentration, supply chain resilience, and strategic implications
1Sourcing Concentration Analysis
Borneo Pacific Pharmaceuticals Ltd exhibits a high degree of sourcing concentration, with 100% of its pharmaceutical imports from India. Between 2022 and 2026, the company imported a total of 11 shipments from India, encompassing 133 unique formulations. This concentrated sourcing strategy may offer benefits such as streamlined logistics and potentially favorable pricing agreements. However, it also introduces risks associated with over-reliance on a single supplier base, including exposure to geopolitical tensions, supply chain disruptions, and currency fluctuations. The stability of this relationship is crucial; any significant disruptions in the Indian pharmaceutical sector could impact Borneo Pacific Pharmaceuticals Ltd's ability to meet domestic demand.
2Supply Chain Resilience
The resilience of Borneo Pacific Pharmaceuticals Ltd's Indian supply chain is a critical factor in its operational stability. While the company has established relationships with multiple Indian suppliers, including BDH Industries Limited, IPCA Laboratories Limited, and Mylan Laboratories Limited, the exact number of unique suppliers is not specified in the available data. The company's import data indicates a diversified sourcing strategy within India, which can mitigate risks associated with single-source dependency. However, the overall resilience is contingent upon the reliability and compliance of these suppliers, as well as the robustness of the shipping routes and logistics infrastructure. Ensuring that all suppliers adhere to international Good Manufacturing Practices (GMP) and maintaining alternative sourcing options are essential strategies to enhance supply chain resilience.
3Strategic Implications
Borneo Pacific Pharmaceuticals Ltd's concentrated sourcing pattern from India positions it to leverage cost efficiencies and strong supplier relationships. However, this strategy also exposes the company to risks associated with over-reliance on a single supplier base. For Indian exporters, this presents an opportunity to strengthen existing partnerships and explore avenues for expanding their product offerings to Borneo Pacific Pharmaceuticals Ltd. Diversifying the product range and ensuring compliance with international quality standards can enhance the attractiveness of Indian exports to this key importer.
Importing Pharmaceuticals into Papua New Guinea — Regulatory Framework
Regulatory authority, GMP requirements, import licensing for Papua New Guinea
1Regulatory Authority & Framework
In Papua New Guinea, the regulation of pharmaceutical imports is overseen by the Pharmaceutical Services Standards Branch (PSSB) of the National Department of Health. The PSSB is responsible for administering and implementing the Medicines and Cosmetics Act 1999 and its regulations, ensuring that medicinal products and medical devices are safe, effective, and of good quality for use by the population. (health.gov.pg) The Medicines and Cosmetics Act 1999 provides the legal framework for the registration, importation, and distribution of pharmaceutical products in the country. Additionally, the Drugs Regulations 1958, made under the Drugs Act 1952, outline specific requirements and standards for drugs, including importation procedures and compliance with established standards. (wipo.int)
2Import Licensing & GMP
Import licensing in Papua New Guinea is governed by the Medicines and Cosmetics Act 1999 and the Drugs Regulations 1958. To import restricted drugs, an importer must obtain a drug import license from the Commissioner General, which is subject to specific conditions and requirements. (customs.gov.pg) Importers are also required to ensure that their products comply with international Good Manufacturing Practices (GMP). The Pharmaceutical Society of Papua New Guinea provides guidelines on GMP clearance, emphasizing the importance of adhering to recognized standards to ensure the quality and safety of imported medicines.
3Quality & Labeling
Imported pharmaceutical products must undergo batch testing and meet stability requirements as stipulated by the PSSB to ensure their safety and efficacy. Labeling requirements include the use of the English language, as it is the official language of Papua New Guinea. Additionally, serialization mandates may be enforced to enhance traceability and prevent counterfeit products from entering the market. These measures are part of the regulatory framework designed to uphold the integrity of the pharmaceutical supply chain and protect public health.
4Recent Regulatory Changes
Between 2024 and 2026, there have been no publicly disclosed policy changes affecting Indian pharmaceutical imports into Papua New Guinea. The regulatory environment remains stable, with existing laws and regulations continuing to govern the importation and distribution of pharmaceutical products. Importers are advised to stay informed about any potential changes and ensure ongoing compliance with all applicable regulations.
Borneo Pacific Pharmaceuticals Ltd — Procurement Pattern Analysis
Product strategy, sourcing profile, and market positioning
1Product Strategy
Borneo Pacific Pharmaceuticals Ltd's focus on antimalarial and antiparasitic products, particularly Primaquine, Pyrimethamine, and Sulfadoxine, aligns with the significant public health challenges posed by malaria and other parasitic diseases in Papua New Guinea. The company's import data indicates that these products constitute 100% of its pharmaceutical imports from India between 2022 and 2026, with Primaquine alone accounting for 11.9% of the total import value. This strategic focus underscores the company's commitment to addressing critical health needs and its responsiveness to market demand for effective treatments.
2Sourcing Profile
Borneo Pacific Pharmaceuticals Ltd's sourcing strategy is centered on importing generic drug formulations from India, a country renowned for its robust pharmaceutical manufacturing sector. The company's preference for finished pharmaceutical formulations, including tablets, capsules, syrups, and injections, reflects a strategic decision to provide ready-to-use products that meet the diverse needs of the Papua New Guinea market. India's adherence to international GMP standards and its competitive pricing further enhance the attractiveness of Indian products in Borneo Pacific Pharmaceuticals Ltd's procurement strategy.
3Market Positioning
Based on its product mix, Borneo Pacific Pharmaceuticals Ltd primarily serves the wholesale distribution segment of the Papua New Guinea market. By importing and distributing a range of pharmaceutical products, the company supplies hospitals, clinics, pharmacies, and other healthcare providers across the nation. Its strategic focus on antimalarial and antiparasitic medications positions it as a key player in addressing the country's public health priorities.
Seller's Guide — How to Become a Supplier to Borneo Pacific Pharmaceuticals Ltd
Opportunity assessment, qualifications, and practical approach strategy
1Opportunity Assessment
There is a realistic opportunity for new Indian suppliers to engage with Borneo Pacific Pharmaceuticals Ltd, particularly in the areas of antimalarial and antiparasitic medications. The company's concentrated sourcing pattern from India indicates a preference for established relationships, but also suggests potential for diversification within the Indian supplier base. Indian exporters can explore this opportunity by ensuring compliance with international quality standards, obtaining necessary certifications, and demonstrating the efficacy and safety of their products.
2Requirements & Qualifications
Indian exporters seeking to supply Borneo Pacific Pharmaceuticals Ltd and the Papua New Guinea market must obtain the necessary import licenses and ensure their products comply with international GMP standards. Adherence to the Medicines and Cosmetics Act 1999 and the Drugs Regulations 1958 is mandatory. Additionally, products must meet batch testing and stability requirements, and labeling must be in English. Serialization may also be required to enhance traceability. Maintaining these standards is essential for successful market entry and ongoing compliance.
3How to Approach
Indian exporters should initiate engagement with Borneo Pacific Pharmaceuticals Ltd by establishing direct communication with key decision-makers, such as the President & Chief Executive Officer, Michael Robinson. Participating in relevant tenders and responding to Requests for Proposals (RFPs) can provide avenues for collaboration. Develop
Frequently Asked Questions — Borneo Pacific Pharmaceuticals Ltd
What products does Borneo Pacific Pharmaceuticals Ltd import from India?
Borneo Pacific Pharmaceuticals Ltd imports 3 pharmaceutical products across 1 categories. Top imports: Primaquine ($265.0K), Pyrimethamine ($100.0K), Sulfadoxine ($100.0K).
Who supplies pharmaceuticals to Borneo Pacific Pharmaceuticals Ltd from India?
Borneo Pacific Pharmaceuticals Ltd sources from 6 verified Indian suppliers. The primary supplier is Bdh Industries Limited (70.3% of imports, $1.6M).
What is Borneo Pacific Pharmaceuticals Ltd's total pharmaceutical import value?
Borneo Pacific Pharmaceuticals Ltd's total pharmaceutical import value from India is $465.0K, based on 11 verified shipments in Indian Customs (DGFT) data.
What therapeutic categories does Borneo Pacific Pharmaceuticals Ltd focus on?
Borneo Pacific Pharmaceuticals Ltd imports across 1 categories. The largest: Antimalarial & Antiparasitic (100.0%).
Get Full Borneo Pacific Pharmaceuticals Ltd Import Intelligence
Access shipment-level details, supplier connections, pricing data, and competitive analysis. TransData Nexus provides verified Indian Customs (DGFT) data trusted by pharmaceutical trade professionals worldwide.
Official References & Regulatory Resources
Verify import regulations and drug registration requirements with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Buyer Matching: Borneo Pacific Pharmaceuticals Ltd identified across shipments using consignee name normalization, aggregating 2 name variants.
- 2.Statistical Normalization: Shipment values are statistically normalized to ensure accurate market share representation.
- 3.Market Share: Calculated per product as Borneo Pacific Pharmaceuticals Ltd's capped value divided by total Indian exports for that product.
- 4.Shipment Count: Based on 11 individual customs records matching Borneo Pacific Pharmaceuticals Ltd.
- 5.Supplier Verification: Borneo Pacific Pharmaceuticals Ltd sources from 6 verified Indian suppliers across 133 formulations, confirmed from customs records.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
3 Products Tracked
1 therapeutic categories
Expert-Reviewed
By pharmaceutical trade specialists
Data Source & Methodology
Trade data sourced from Indian Customs (DGFT) export shipment records. Values represent FOB export value in USD. Profile aggregates 2 company name variants from customs records. For current shipment-level data, contact TransData Nexus.