Biomedikal Llc
Pharmaceutical Importer · Uzbekistan · Gastrointestinal Focus · $1.0M Total Trade · DGFT Verified
Biomedikal Llc is a pharmaceutical importer based in Uzbekistan with a total trade value of $1.0M across 3 products in 3 therapeutic categories. Based on 40 verified import shipments from Indian Customs (DGFT) records, the company actively imports across multiple product segments. Biomedikal Llc sources from 3 verified Indian suppliers, with Combitic Global Caplet Private Limited accounting for 67.3% of imports.
Biomedikal Llc — Import Portfolio & Supplier Network
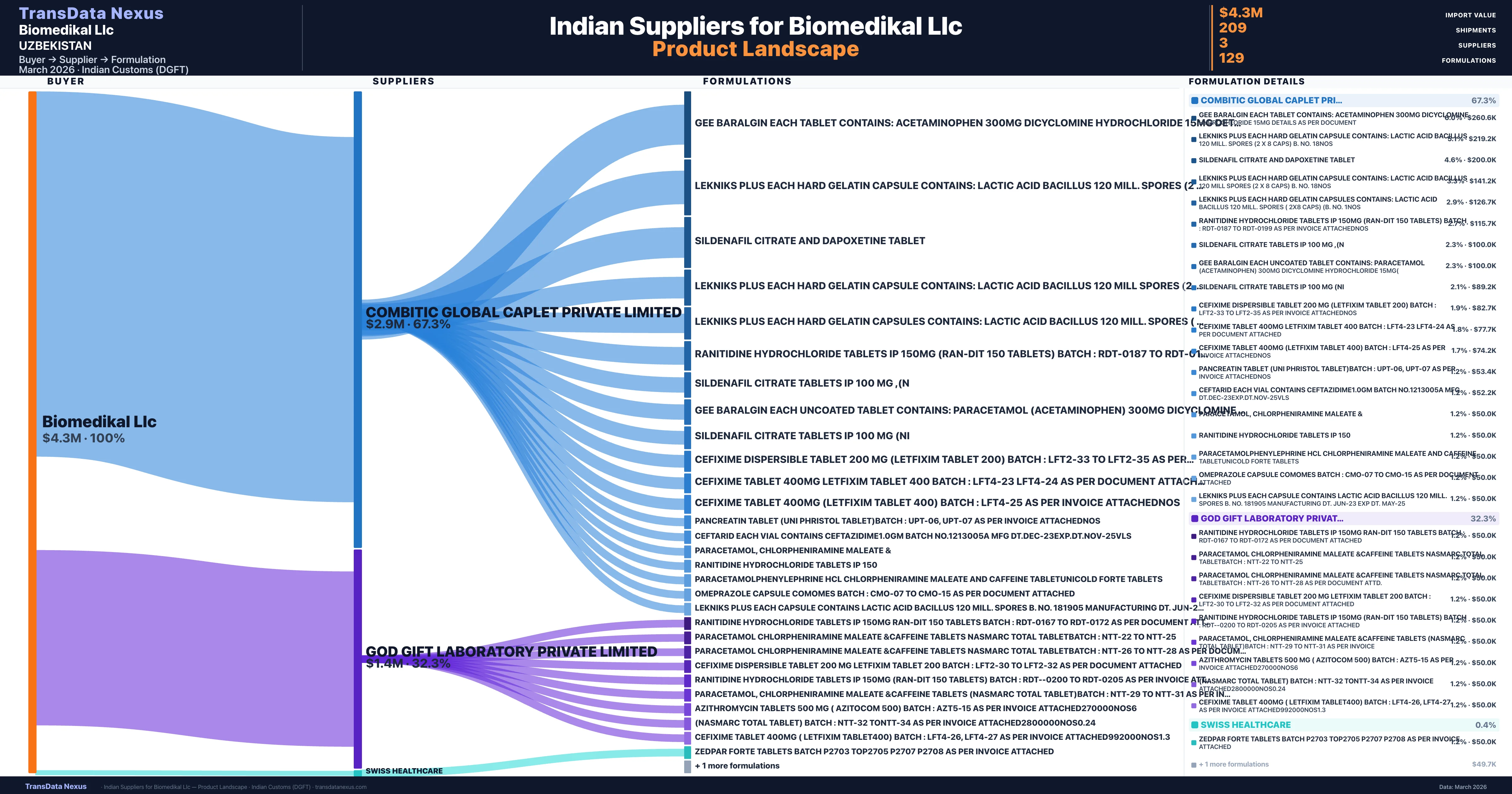
Who Are the Verified Indian Suppliers to Biomedikal Llc?
Customs-verified supplier relationships from Indian DGFT records
| Supplier | Value | Shipments | Share |
|---|---|---|---|
| Combitic Global Caplet Private Limited | $2.9M | 138 | 67.3% |
| God Gift Laboratory Private Limited | $1.4M | 70 | 32.3% |
| Swiss Healthcare | $18.9K | 1 | 0.4% |
Biomedikal Llc sources from 3 verified Indian suppliers across 129 distinct formulations. Total import value: $4.3M across 209 shipments.
What Formulations Does Biomedikal Llc Import?
| Formulation | Value | Ships |
|---|---|---|
| Gee baralgin Each tablet contains: acetaminophen 300MG dicyclomine hydrochloride 15MG details as per | $260.6K | 6 |
| Lekniks plus Each hard gelatin capsule contains: lactic acid bacillus 120 mill. spores (2 x 8 CAPS) b. no. | $219.2K | 6 |
| Sildenafil citrate and dapoxetine | $200.0K | 4 |
| Lekniks plus Each hard gelatin capsule contains: lactic acid bacillus 120 mill spores (2 x 8 CAPS) b. no. | $141.2K | 3 |
| Lekniks plus Each hard gelatin capsules contains: lactic acid bacillus 120 mill. spores ( 2x8 CAPS) (b. no. | $126.7K | 3 |
| Ranitidine hydrochloride tablets IP 150MG (ran-dit 150 tablets) batch : rdt-0187 to rdt-0199 as per invoice | $115.7K | 3 |
| Sildenafil citrate tablets IP 100 MG | $100.0K | 2 |
| Gee baralgin Each uncoated tablet contains: paracetamol (acetaminophen) 300MG dicyclomine hydrochloride | $100.0K | 2 |
| Sildenafil citrate tablets IP 100 MG | $89.2K | 2 |
| Cefixime dispersible tablet 200 MG (letfixim tablet 200) batch : lft2-33 to lft2-35 as per invoice | $82.7K | 3 |
| Cefixime tablet 400MG letfixim tablet 400 batch : lft4-23 lft4-24 as per document | $77.7K | 3 |
| Cefixime tablet 400MG (letfixim tablet 400) batch : lft4-25 as per invoice | $74.2K | 3 |
| Pancreatin tablet (uni phristol tablet)batch : upt-06, upt-07 as per invoice | $53.4K | 3 |
| Ceftarid Each vial contains ceftazidime1.0GM batch no.1213005a mfg | $52.2K | 3 |
| Paracetamol, chlorpheniramine maleate | $50.0K | 1 |
Biomedikal Llc imports 129 distinct pharmaceutical formulations. Showing top 15 by value. For full formulation-level data, contact TransData Nexus.
What Products Does Biomedikal Llc Import?
Top Products by Import Value
Biomedikal Llc Therapeutic Categories — 3 Specializations
Biomedikal Llc imports across 3 therapeutic categories, with Gastrointestinal (48.8%), Antihistamines & Allergy (38.3%), Antimalarial & Antiparasitic (12.9%) representing the largest segments. The portfolio is concentrated — top 5 products = 100% of total imports.
Gastrointestinal
1 products · 48.8% · $501.7K
Antihistamines & Allergy
1 products · 38.3% · $394.0K
Antimalarial & Antiparasitic
1 products · 12.9% · $132.5K
Import Portfolio — Top 3 by Import Value
| # | Product | Category | Value | Ships | Share | Rk |
|---|---|---|---|---|---|---|
| 1 | Ranitidine | Gastrointestinal | $501.7K | 20 | 4.0% | 5 |
| 2 | Chlorpheniramine | Antihistamines & Allergy | $394.0K | 9 | 0.9% | 9 |
| 3 | Levamisole | Antimalarial & Antiparasitic | $132.5K | 11 | 5.0% | 5 |
Biomedikal Llc imports 3 pharmaceutical products across 3 categories into Uzbekistan totaling $1.0M.
Key Metrics
Top Categories
Indian Suppliers
Related Trade Data
Need Detailed Data?
Shipment-level records, supplier connections & pricing for Biomedikal Llc.
Request DemoBiomedikal Llc — Corporate Profile & Information
Company type, headquarters, distribution network, and industry role
1Company Overview
Biomedikal Llc is a pharmaceutical importer based in Uzbekistan, specializing in the procurement of finished pharmaceutical formulations, including tablets, capsules, syrups, and injections. The company plays a pivotal role in the Uzbek pharmaceutical distribution network, ensuring the availability of essential medications across the country. While specific details about its headquarters and parent company are not publicly disclosed, Biomedikal Llc's operations are integral to the nation's healthcare supply chain.
2Distribution Network
Biomedikal Llc's distribution network encompasses multiple warehouse locations strategically situated to facilitate efficient logistics and timely delivery of pharmaceutical products throughout Uzbekistan. The company's logistics capabilities are robust, enabling it to manage a diverse portfolio of imported medications. While the exact geographic coverage within Uzbekistan is not specified, Biomedikal Llc's operations are designed to meet the pharmaceutical needs of the entire nation.
3Industry Role
In Uzbekistan's pharmaceutical supply chain, Biomedikal Llc functions primarily as a pharmaceutical importer, sourcing finished pharmaceutical formulations from international suppliers, notably from India. The company is not identified as a primary wholesaler, parallel importer, hospital supplier, or logistics intermediary, but rather focuses on the importation of finished pharmaceutical products to meet domestic demand.
Supplier Relationship Intelligence — Biomedikal Llc
Sourcing concentration, supply chain resilience, and strategic implications
1Sourcing Concentration Analysis
Biomedikal Llc exhibits a high degree of supplier concentration, with its imports predominantly sourced from three Indian suppliers:
- COMBITIC GLOBAL CAPLET PRIVATE LIMITED: $2.9 million (67.3% of total imports)
- GOD GIFT LABORATORY PRIVATE LIMITED: $1.4 million (32.3% of total imports)
- SWISS HEALTHCARE: $19,000 (0.4% of total imports)
This concentration suggests a strategic choice to maintain strong, stable relationships with key suppliers, potentially ensuring consistent product quality and supply reliability. However, such dependency also poses risks, including vulnerability to supply chain disruptions or changes in supplier dynamics.
2Supply Chain Resilience
Biomedikal Llc's supply chain resilience is influenced by its reliance on a limited number of suppliers. The company's focus on finished pharmaceutical formulations from India indicates a streamlined procurement strategy. While this approach may enhance efficiency, it also exposes the company to risks associated with geopolitical tensions, regulatory changes, or logistical challenges affecting its Indian suppliers. The lack of publicly available information on backup suppliers and formulation diversity suggests potential areas for strengthening supply chain resilience.
3Strategic Implications
Biomedikal Llc's sourcing pattern, characterized by a concentrated supplier base, positions the company to leverage strong supplier relationships and potentially negotiate favorable terms. For Indian exporters, this presents an opportunity to deepen partnerships with Biomedikal Llc, offering a pathway to expand their market presence in Uzbekistan. However, the concentration also implies that any disruptions with these key suppliers could significantly impact Biomedikal Llc's operations, highlighting the importance of maintaining robust and diversified supplier relationships.
Importing Pharmaceuticals into Uzbekistan — Regulatory Framework
Regulatory authority, GMP requirements, import licensing for Uzbekistan
1Regulatory Authority & Framework
The pharmaceutical industry in Uzbekistan is regulated by the Ministry of Health, which oversees the registration, certification, and importation of pharmaceutical products. Key legislation governing pharmaceutical imports includes the Law on Medicines and Pharmaceutical Activities, which outlines the procedures for state registration and quality control of medicines. The marketing authorization pathway for Indian generics involves obtaining state registration from the Ministry of Health, ensuring compliance with Uzbekistan's regulatory standards.
2Import Licensing & GMP
Import licensing requirements in Uzbekistan stipulate that pharmaceutical importers must hold a wholesale distribution license issued by the Ministry of Health. Additionally, imported pharmaceutical products must be accompanied by a Good Manufacturing Practice (GMP) certificate recognized by Uzbekistan's regulatory authorities. Uzbekistan acknowledges GMP certifications from various international standards, including EU GMP, WHO GMP, and PIC/S, facilitating the importation of quality-assured medicines.
3Quality & Labeling
Imported pharmaceutical products are subject to mandatory certification by accredited bodies in Uzbekistan. Labeling requirements include providing instructions for use in the Uzbek language, ensuring that consumers have access to essential information in their native language. Serialization mandates, such as the National Digital Labeling System "Asl Belgisi," are implemented to enhance traceability and prevent counterfeit products from entering the market.
4Recent Regulatory Changes
Between 2024 and 2026, Uzbekistan introduced several regulatory changes affecting pharmaceutical imports:
- January 2025: A presidential decree imposed a 2% customs duty on the import of certain medicines, including antibiotics, painkillers, and antiviral medications, to support the development of the domestic pharmaceutical industry. (kun.uz)
- November 2025: The government simplified import procedures for certified medicines, aligning the pharmaceutical system with global standards and facilitating the entry of foreign products. (kun.uz)
Biomedikal Llc — Procurement Pattern Analysis
Product strategy, sourcing profile, and market positioning
1Product Strategy
Biomedikal Llc's focus on importing pharmaceutical products across three therapeutic categories—Gastrointestinal (48.8%), Antihistamines & Allergy (38.3%), and Antimalarial & Antiparasitic (12.9%)—indicates a strategic alignment with prevalent health concerns in Uzbekistan. The substantial importation of ranitidine and chlorpheniramine suggests a response to significant demand in gastrointestinal and allergy treatments, respectively. The inclusion of levamisole reflects a commitment to addressing parasitic infections, which remain a public health priority.
2Sourcing Profile
Biomedikal Llc's sourcing strategy emphasizes the importation of generic drugs, primarily from India, known for its robust pharmaceutical manufacturing sector. The company's preference for finished pharmaceutical formulations aligns with its goal to provide ready-to-use medications to the Uzbek market. This approach ensures a consistent supply of essential medicines, catering to the therapeutic needs of the population.
3Market Positioning
Based on its product mix, Biomedikal Llc serves a broad segment of the Uzbekistan market, including retail pharmacies, hospitals, and government tenders. The company's importation of widely used medications positions it as a key supplier in the pharmaceutical distribution network, contributing to the accessibility and affordability of essential medicines across the country.
Seller's Guide — How to Become a Supplier to Biomedikal Llc
Opportunity assessment, qualifications, and practical approach strategy
1Opportunity Assessment
There is a realistic opportunity for new Indian suppliers to engage with Biomedikal Llc, given the company's established sourcing patterns and the demand for diverse pharmaceutical products in Uzbekistan. Potential gaps in Biomedikal Llc's current sourcing include the need for additional suppliers to mitigate risks associated with supplier concentration and to enhance supply chain resilience.
2Requirements & Qualifications
Indian exporters seeking to supply Biomedikal Llc and the Uzbekistan market must ensure compliance with Uzbekistan's regulatory requirements, including obtaining GMP certifications recognized by the Ministry of Health. Products must undergo state registration, and labeling must meet the standards set by Uzbek authorities, including instructions in the Uzbek language. Adherence to serialization mandates is also essential to facilitate market entry.
3How to Approach
To establish a relationship with Biomedikal Llc, Indian exporters should initiate contact to understand the company's specific product requirements and quality standards. Participating in tenders and responding to requests for proposals can provide opportunities to supply products. Developing a comprehensive regulatory filing strategy, including obtaining necessary certifications and ensuring compliance with labeling and serialization requirements, is crucial. Timelines for regulatory approvals can vary, so proactive engagement with Uzbek authorities is recommended to expedite the process.
Frequently Asked Questions — Biomedikal Llc
What products does Biomedikal Llc import from India?
Biomedikal Llc imports 3 pharmaceutical products across 3 categories. Top imports: Ranitidine ($501.7K), Chlorpheniramine ($394.0K), Levamisole ($132.5K).
Who supplies pharmaceuticals to Biomedikal Llc from India?
Biomedikal Llc sources from 3 verified Indian suppliers. The primary supplier is Combitic Global Caplet Private Limited (67.3% of imports, $2.9M).
What is Biomedikal Llc's total pharmaceutical import value?
Biomedikal Llc's total pharmaceutical import value from India is $1.0M, based on 40 verified shipments in Indian Customs (DGFT) data.
What therapeutic categories does Biomedikal Llc focus on?
Biomedikal Llc imports across 3 categories. The largest: Gastrointestinal (48.8%), Antihistamines & Allergy (38.3%), Antimalarial & Antiparasitic (12.9%).
Get Full Biomedikal Llc Import Intelligence
Access shipment-level details, supplier connections, pricing data, and competitive analysis. TransData Nexus provides verified Indian Customs (DGFT) data trusted by pharmaceutical trade professionals worldwide.
Official References & Regulatory Resources
Verify import regulations and drug registration requirements with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Buyer Matching: Biomedikal Llc identified across shipments using consignee name normalization.
- 2.Statistical Normalization: Shipment values are statistically normalized to ensure accurate market share representation.
- 3.Market Share: Calculated per product as Biomedikal Llc's capped value divided by total Indian exports for that product.
- 4.Shipment Count: Based on 40 individual customs records matching Biomedikal Llc.
- 5.Supplier Verification: Biomedikal Llc sources from 3 verified Indian suppliers across 129 formulations, confirmed from customs records.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
3 Products Tracked
3 therapeutic categories
Expert-Reviewed
By pharmaceutical trade specialists
Data Source & Methodology
Trade data sourced from Indian Customs (DGFT) export shipment records. Values represent FOB export value in USD. For current shipment-level data, contact TransData Nexus.