BGP Products Gmbh
Pharmaceutical Importer · Netherlands · Cardiovascular Focus · $35.4M Total Trade · DGFT Verified
BGP Products Gmbh is a pharmaceutical importer based in Netherlands with a total trade value of $35.4M across 3 products in 3 therapeutic categories. Based on 1,216 verified import shipments from Indian Customs (DGFT) records, BGP Products Gmbh is the #1 buyer in 1 product including Brufen. BGP Products Gmbh sources from 7 verified Indian suppliers, with Medreich Limited accounting for 39.1% of imports.
BGP Products Gmbh — Import Portfolio & Supplier Network
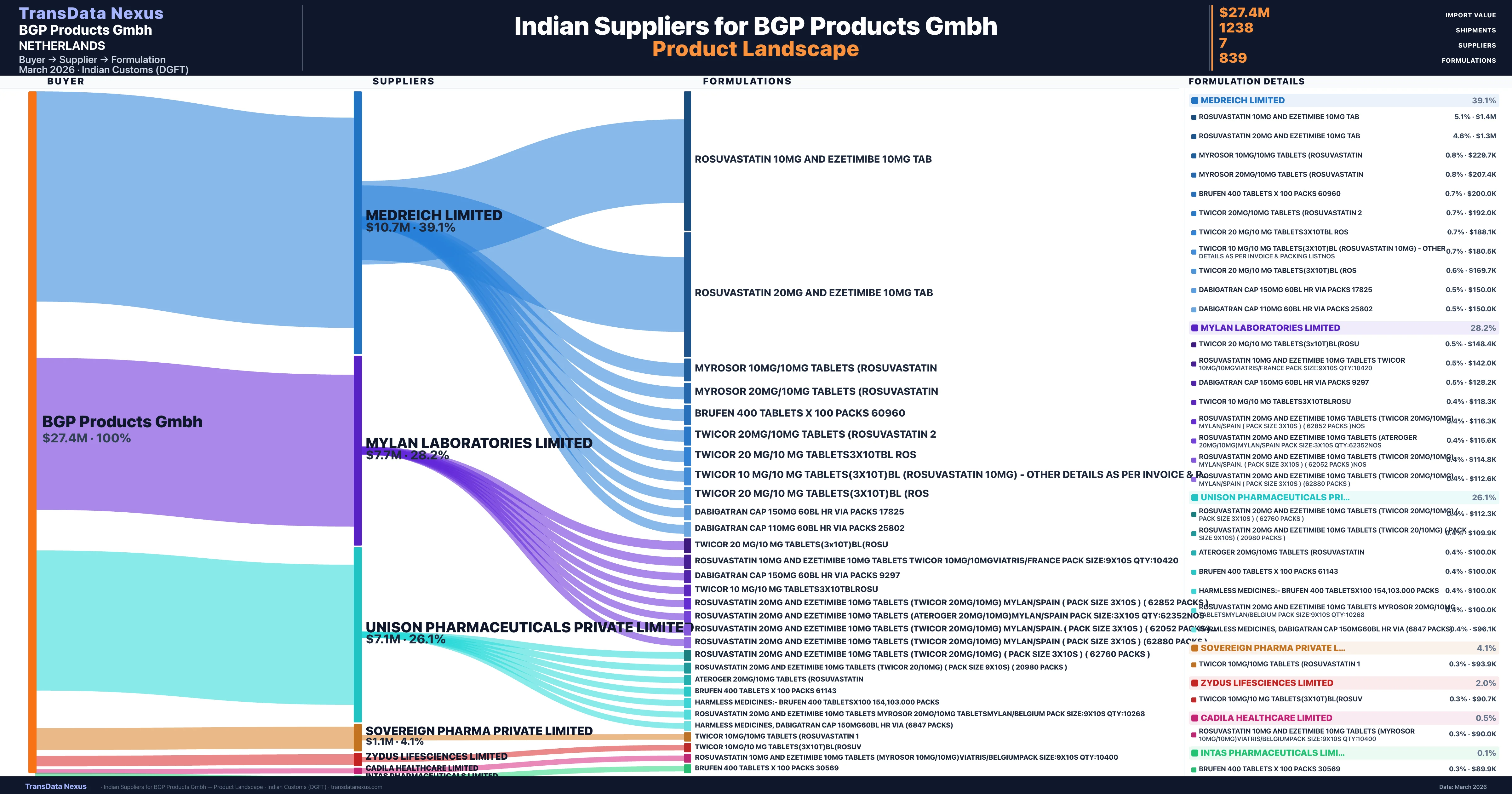
Who Are the Verified Indian Suppliers to BGP Products Gmbh?
Customs-verified supplier relationships from Indian DGFT records
| Supplier | Value | Shipments | Share |
|---|---|---|---|
| Medreich Limited | $10.7M | 371 | 39.1% |
| Mylan Laboratories Limited | $7.7M | 444 | 28.2% |
| Unison Pharmaceuticals Private Limited | $7.1M | 323 | 26.1% |
| Sovereign Pharma Private Limited | $1.1M | 60 | 4.1% |
| Zydus Lifesciences Limited | $535.8K | 31 | 2.0% |
| Cadila Healthcare Limited | $141.7K | 7 | 0.5% |
| Intas Pharmaceuticals Limited | $19.5K | 2 | 0.1% |
BGP Products Gmbh sources from 7 verified Indian suppliers across 839 distinct formulations. The supply base is diversified across 7 suppliers, reducing single-source dependency risk.
What Formulations Does BGP Products Gmbh Import?
| Formulation | Value | Ships |
|---|---|---|
| Rosuvastatin 10MG and ezetimibe 10MG TAB | $1.4M | 47 |
| Rosuvastatin 20MG and ezetimibe 10MG TAB | $1.3M | 36 |
| Myrosor 10MG/10MG tablets | $229.7K | 11 |
| Myrosor 20MG/10MG tablets | $207.4K | 8 |
| Brufen 400 tablets x 100 packs | $200.0K | 4 |
| Twicor 20MG/10MG tablets (rosuvastatin | $192.0K | 9 |
| Twicor 20 MG/10 MG tablets3x10tbl | $188.1K | 7 |
| Twicor 10 MG/10 MG tablets(3x10t)bl (rosuvastatin 10MG) - other details as per invoice & packing | $180.5K | 8 |
| Twicor 20 MG/10 MG tablets(3x10t)bl | $169.7K | 5 |
| Dabigatran cap 150MG 60bl hr via packs | $150.0K | 3 |
| Dabigatran cap 110MG 60bl hr via packs | $150.0K | 3 |
| Twicor 20 MG/10 MG tablets(3x10t | $148.4K | 6 |
| Rosuvastatin 10MG and ezetimibe 10MG tablets twicor 10MG/10mgviatris/france Pack size:9x10s | $142.0K | 3 |
| Dabigatran cap 150MG 60bl hr via packs | $128.2K | 3 |
| Twicor 10 MG/10 MG tablets3x10tblrosu | $118.3K | 6 |
BGP Products Gmbh imports 839 distinct pharmaceutical formulations. Showing top 15 by value. For full formulation-level data, contact TransData Nexus.
What Products Does BGP Products Gmbh Import?
Top Products by Import Value
BGP Products Gmbh Therapeutic Categories — 3 Specializations
BGP Products Gmbh imports across 3 therapeutic categories, with Cardiovascular (49.8%), Lipid & Metabolism (46.2%), Brand Names & OTC Products (4.0%) representing the largest segments. The portfolio is concentrated — top 5 products = 100% of total imports.
Cardiovascular
1 products · 49.8% · $17.6M
Lipid & Metabolism
1 products · 46.2% · $16.4M
Brand Names & OTC Products
1 products · 4.0% · $1.4M
Import Portfolio — Top 3 by Import Value
| # | Product | Category | Value | Ships | Share | Rk |
|---|---|---|---|---|---|---|
| 1 | Rosuvastatin | Cardiovascular | $17.6M | 621 | 3.8% | 5 |
| 2 | Ezetimibe | Lipid & Metabolism | $16.4M | 566 | 3.2% | 4 |
| 3 | Brufen | Brand Names & OTC Products | $1.4M | 29 | 15.6% | 1 |
BGP Products Gmbh imports 3 pharmaceutical products across 3 categories into Netherlands totaling $35.4M. The company is the #1 buyer for 1 product: Brufen.
Key Metrics
Top Categories
Indian Suppliers
Related Trade Data
Need Detailed Data?
Shipment-level records, supplier connections & pricing for BGP Products Gmbh.
Request DemoBGP Products Gmbh — Corporate Profile & Information
Company type, headquarters, distribution network, and industry role
1Company Overview
BGP Products GmbH is a pharmaceutical importer and buyer based in the Netherlands, specializing in the acquisition of finished pharmaceutical formulations from India. The company operates as a non-trading entity, focusing on the procurement and distribution of pharmaceutical products within the Dutch market. Established in 2014, BGP Products GmbH is headquartered at Krijgsman 20, 1186 DM, Amstelveen, Netherlands.
As a key player in the Netherlands' pharmaceutical distribution network, BGP Products GmbH plays a pivotal role in ensuring the availability of essential medications sourced from India. The company's operations are integral to the supply chain, facilitating the importation and distribution of a diverse range of pharmaceutical products to meet the healthcare needs of the Dutch population.
2Distribution Network
While specific details regarding BGP Products GmbH's warehouse locations and logistics capabilities are not publicly disclosed, the company is known to import pharmaceutical products from various ports in India, including Nhava Sheva Sea, Bombay Air Cargo, Bangalore Air Cargo, Bangalore ICD, and Mundra Sea. This indicates a well-established logistics network capable of handling a significant volume of shipments. (eximpedia.app)
The company's geographic coverage primarily focuses on the Netherlands, ensuring a steady supply of pharmaceutical products to meet domestic demand. Given the strategic importance of the Netherlands as a central hub in European logistics, BGP Products GmbH's distribution network is likely designed to efficiently serve both national and broader European markets.
3Industry Role
BGP Products GmbH functions as a pharmaceutical importer and buyer, sourcing finished pharmaceutical formulations from India for distribution within the Netherlands. The company's role is crucial in bridging the supply of pharmaceutical products from India to the Dutch market, ensuring the availability of essential medications. By importing a diverse range of products, BGP Products GmbH contributes to the competitiveness and diversity of the pharmaceutical supply chain in the Netherlands.
Supplier Relationship Intelligence — BGP Products Gmbh
Sourcing concentration, supply chain resilience, and strategic implications
1Sourcing Concentration Analysis
BGP Products GmbH's sourcing strategy exhibits a high degree of concentration, with the top five products accounting for 100% of the company's imports from India. This indicates a focused procurement approach, potentially leveraging economies of scale and establishing strong supplier relationships. The company's significant importation of Rosuvastatin and Ezetimibe suggests a strategic emphasis on cardiovascular and lipid metabolism therapies, aligning with prevalent health concerns in the Netherlands.
The concentration of imports from a limited number of suppliers—seven verified Indian suppliers—may present both opportunities and risks. On one hand, it allows for streamlined logistics and potentially favorable terms. On the other hand, dependency on a small group of suppliers could expose BGP Products GmbH to supply chain disruptions. However, the company's substantial volume of shipments (1,216 total shipments) and the diversity of suppliers suggest a level of stability and risk mitigation in its sourcing strategy.
2Supply Chain Resilience
BGP Products GmbH's supply chain resilience is supported by its diversified supplier base, comprising seven verified Indian suppliers. This diversification helps mitigate risks associated with over-reliance on a single supplier. The company's substantial import volume and the variety of suppliers indicate a robust supply chain capable of adapting to potential disruptions.
The importation of 839 unique pharmaceutical formulations further demonstrates the company's commitment to offering a comprehensive product portfolio. This diversity not only caters to a wide range of therapeutic needs but also enhances the company's ability to respond to changing market demands and regulatory requirements.
3Strategic Implications
BGP Products GmbH's focused sourcing strategy positions it as a significant player in the Netherlands' pharmaceutical market, particularly in the cardiovascular and lipid metabolism segments. The company's substantial import volumes and supplier relationships provide a competitive edge in negotiating favorable terms and ensuring a steady supply of high-demand medications.
For Indian pharmaceutical exporters, BGP Products GmbH represents a valuable partner, especially for products like Rosuvastatin and Ezetimibe. Establishing a relationship with BGP Products GmbH could facilitate entry into the Dutch market and potentially open avenues for broader European distribution.
Importing Pharmaceuticals into Netherlands — Regulatory Framework
Regulatory authority, GMP requirements, import licensing for Netherlands
1Regulatory Authority & Framework
In the Netherlands, the Medicines Evaluation Board (CBG-MEB) is the primary regulatory authority overseeing the authorization and supervision of medicinal products. The CBG-MEB ensures that all pharmaceutical products meet the required safety, efficacy, and quality standards before they are marketed. (english.cbg-meb.nl)
The importation of pharmaceutical products into the Netherlands is governed by the Medicines Act (Geneesmiddelenwet), which outlines the requirements for marketing authorization, import licenses, and compliance with Good Manufacturing Practice (GMP) standards. This legislation ensures that imported medicines are safe for public use and meet the necessary quality standards.
2Import Licensing & GMP
Companies involved in the importation of medicinal products into the Netherlands must obtain a manufacturing authorization or a wholesale distribution authorization. A manufacturing authorization is required for the preparation, trading in, import, and export of medicinal products and active substances. A wholesale distribution authorization is necessary for activities such as procuring, holding, supplying, delivering, or exporting medicinal products or active substances prepared or imported by a third party.
Compliance with GMP standards is mandatory for all pharmaceutical products imported into the Netherlands. This ensures that the products are consistently produced and controlled to the quality standards appropriate to their intended use. GMP compliance is verified through inspections and certifications by recognized authorities.
3Quality & Labeling
Imported pharmaceutical products must undergo batch testing to verify their quality, safety, and efficacy. This testing ensures that the products meet the standards set by the CBG-MEB and are safe for public use.
Stability requirements are also enforced to ensure that pharmaceutical products maintain their intended quality throughout their shelf life. This includes testing under various environmental conditions to confirm the product's stability.
Labeling requirements stipulate that product information must be clear, accurate, and in Dutch. This includes details such as dosage instructions, active ingredients, and any potential side effects. Serialization mandates are in place to track and trace pharmaceutical products, enhancing safety and preventing counterfeit products from entering the market.
4Recent Regulatory Changes
Effective from 1 July 2024, significant changes have been implemented regarding the declaration of pharmaceutical products and active substances for import into the Netherlands. Importers are now required to include specific document codes in their declarations, such as 1400 for marketing authorization holders and 1450 for API registration holders. Failure to include the appropriate codes will result in the rejection of the import declaration.
BGP Products Gmbh — Procurement Pattern Analysis
Product strategy, sourcing profile, and market positioning
1Product Strategy
BGP Products GmbH's product strategy focuses on importing pharmaceutical formulations that address prevalent health concerns in the Netherlands, particularly in the cardiovascular and lipid metabolism therapeutic areas. The company's significant importation of Rosuvastatin and Ezetimibe aligns with the high demand for cholesterol-lowering medications in the Dutch market.
The emphasis on these therapeutic areas is driven by the increasing incidence of cardiovascular diseases and metabolic disorders in the Netherlands. By focusing on these segments, BGP Products GmbH aims to meet the critical healthcare needs of the population and establish a strong presence in these high-demand markets.
2Sourcing Profile
BGP Products GmbH's sourcing strategy is centered on importing generic pharmaceutical formulations from India, leveraging the country's robust pharmaceutical manufacturing capabilities. The company's focus on finished formulations, rather than raw active pharmaceutical ingredients (APIs), indicates a preference for ready-to-market products that comply with Dutch and EU regulatory standards.
India's well-established pharmaceutical industry, known for its adherence to international quality standards, makes it a strategic sourcing partner for BGP Products GmbH. The company's diversified supplier base from India further enhances its ability to procure a wide range of pharmaceutical products to meet market demands.
3Market Positioning
Based on its product mix, BGP Products GmbH primarily serves the wholesale distribution segment of the Netherlands' pharmaceutical market. The company's focus on importing and distributing generic pharmaceutical formulations positions it to supply a broad spectrum of medications to various stakeholders, including retail pharmacies, hospitals, and healthcare providers.
By offering a diverse portfolio of products, BGP Products GmbH contributes to the accessibility and affordability of essential medications, thereby supporting the overall healthcare infrastructure in the Netherlands.
Seller's Guide — How to Become a Supplier to BGP Products Gmbh
Opportunity assessment, qualifications, and practical approach strategy
1Opportunity Assessment
There is a realistic opportunity for new Indian suppliers to establish relationships with BGP Products GmbH, particularly in the therapeutic areas of cardiovascular and lipid metabolism. The company's significant import volumes and focus on these segments indicate a sustained demand for such products in the Dutch market.
Potential gaps in BGP Products GmbH's current sourcing include the need for additional suppliers to diversify its portfolio and mitigate supply chain risks. New Indian suppliers can explore these opportunities by offering high-quality products that meet the regulatory standards of the Netherlands and align with the company's strategic focus areas.
Frequently Asked Questions — BGP Products Gmbh
What products does BGP Products Gmbh import from India?
BGP Products Gmbh imports 3 pharmaceutical products across 3 categories. Top imports: Rosuvastatin ($17.6M), Ezetimibe ($16.4M), Brufen ($1.4M).
Who supplies pharmaceuticals to BGP Products Gmbh from India?
BGP Products Gmbh sources from 7 verified Indian suppliers. The primary supplier is Medreich Limited (39.1% of imports, $10.7M).
What is BGP Products Gmbh's total pharmaceutical import value?
BGP Products Gmbh's total pharmaceutical import value from India is $35.4M, based on 1,216 verified shipments in Indian Customs (DGFT) data.
What therapeutic categories does BGP Products Gmbh focus on?
BGP Products Gmbh imports across 3 categories. The largest: Cardiovascular (49.8%), Lipid & Metabolism (46.2%), Brand Names & OTC Products (4.0%).
Get Full BGP Products Gmbh Import Intelligence
Access shipment-level details, supplier connections, pricing data, and competitive analysis. TransData Nexus provides verified Indian Customs (DGFT) data trusted by pharmaceutical trade professionals worldwide.
Official References & Regulatory Resources
Verify import regulations and drug registration requirements with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Buyer Matching: BGP Products Gmbh identified across shipments using consignee name normalization, aggregating 4 name variants.
- 2.Statistical Normalization: Shipment values are statistically normalized to ensure accurate market share representation.
- 3.Market Share: Calculated per product as BGP Products Gmbh's capped value divided by total Indian exports for that product.
- 4.Shipment Count: Based on 1,216 individual customs records matching BGP Products Gmbh.
- 5.Supplier Verification: BGP Products Gmbh sources from 7 verified Indian suppliers across 839 formulations, confirmed from customs records.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
3 Products Tracked
3 therapeutic categories
Expert-Reviewed
By pharmaceutical trade specialists
Data Source & Methodology
Trade data sourced from Indian Customs (DGFT) export shipment records. Values represent FOB export value in USD. Profile aggregates 4 company name variants from customs records. For current shipment-level data, contact TransData Nexus.