B Logistik Gmbh
Pharmaceutical Importer · Germany · Cardiovascular Focus · $138.4M Total Trade · DGFT Verified
B Logistik Gmbh is a pharmaceutical importer based in Germany with a total trade value of $138.4M across 2 products in 1 therapeutic categories. Based on 2,768 verified import shipments from Indian Customs (DGFT) records, B Logistik Gmbh is the #1 buyer in 2 products including Telmisartan, Amlodipine. B Logistik Gmbh sources from 1 verified Indian supplier, with Cipla Limited accounting for 100.0% of imports.
B Logistik Gmbh — Import Portfolio & Supplier Network
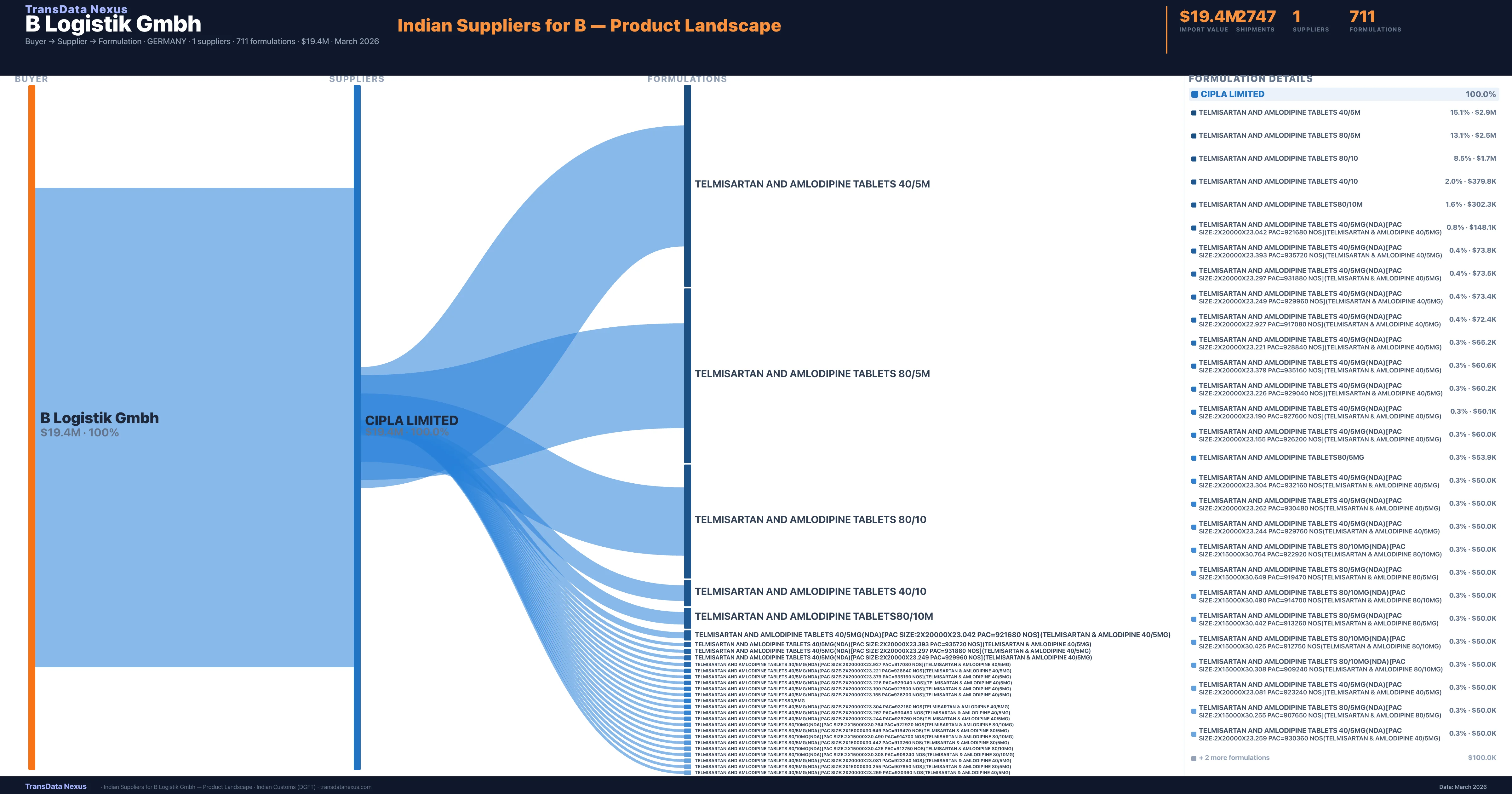
Who Are the Verified Indian Suppliers to B Logistik Gmbh?
Customs-verified supplier relationships from Indian DGFT records
| Supplier | Value | Shipments | Share |
|---|---|---|---|
| Cipla Limited | $19.4M | 2,747 | 100.0% |
B Logistik Gmbh sources from 1 verified Indian supplier across 711 distinct formulations. The sourcing is highly concentrated — Cipla Limited accounts for 100.0% of total imports, indicating a strategic single-source relationship.
What Formulations Does B Logistik Gmbh Import?
| Formulation | Value | Ships |
|---|---|---|
| Telmisartan and amlodipine tablets | $2.9M | 209 |
| Telmisartan and amlodipine tablets | $2.5M | 178 |
| Telmisartan and amlodipine tablets | $1.7M | 117 |
| Telmisartan and amlodipine tablets | $379.8K | 27 |
| Telmisartan and amlodipine | $302.3K | 22 |
| Telmisartan and amlodipine tablets 40/5MG(nda)[pac size:2x20000x23.042 pac=921680 nos](telmisartan & amlodipine 40/5MG) | $148.1K | 10 |
| Telmisartan and amlodipine tablets 40/5MG(nda)[pac size:2x20000x23.393 pac=935720 nos](telmisartan & amlodipine 40/5MG) | $73.8K | 5 |
| Telmisartan and amlodipine tablets 40/5MG(nda)[pac size:2x20000x23.297 pac=931880 nos](telmisartan & amlodipine 40/5MG) | $73.5K | 5 |
| Telmisartan and amlodipine tablets 40/5MG(nda)[pac size:2x20000x23.249 pac=929960 nos](telmisartan & amlodipine 40/5MG) | $73.4K | 5 |
| Telmisartan and amlodipine tablets 40/5MG(nda)[pac size:2x20000x22.927 pac=917080 nos](telmisartan & amlodipine 40/5MG) | $72.4K | 5 |
| Telmisartan and amlodipine tablets 40/5MG(nda)[pac size:2x20000x23.221 pac=928840 nos](telmisartan & amlodipine 40/5MG) | $65.2K | 2 |
| Telmisartan and amlodipine tablets 40/5MG(nda)[pac size:2x20000x23.379 pac=935160 nos](telmisartan & amlodipine 40/5MG) | $60.6K | 4 |
| Telmisartan and amlodipine tablets 40/5MG(nda)[pac size:2x20000x23.226 pac=929040 nos](telmisartan & amlodipine 40/5MG) | $60.2K | 4 |
| Telmisartan and amlodipine tablets 40/5MG(nda)[pac size:2x20000x23.190 pac=927600 nos](telmisartan & amlodipine 40/5MG) | $60.1K | 4 |
| Telmisartan and amlodipine tablets 40/5MG(nda)[pac size:2x20000x23.155 pac=926200 nos](telmisartan & amlodipine 40/5MG) | $60.0K | 4 |
B Logistik Gmbh imports 711 distinct pharmaceutical formulations. Showing top 15 by value. For full formulation-level data, contact TransData Nexus.
What Products Does B Logistik Gmbh Import?
Top Products by Import Value
B Logistik Gmbh Therapeutic Categories — 1 Specializations
B Logistik Gmbh imports across 1 therapeutic categories, with Cardiovascular (100.0%), representing the largest segments. The portfolio is concentrated — top 5 products = 100% of total imports.
Cardiovascular
2 products · 100.0% · $138.4M
Import Portfolio — Top 2 by Import Value
| # | Product | Category | Value | Ships | Share | Rk |
|---|---|---|---|---|---|---|
| 1 | Telmisartan | Cardiovascular | $77.0M | 1,540 | 0.7% | 1 |
| 2 | Amlodipine | Cardiovascular | $61.4M | 1,228 | 0.5% | 1 |
B Logistik Gmbh imports 2 pharmaceutical products across 1 categories into Germany totaling $138.4M. The company is the #1 buyer for 2 products: Telmisartan, Amlodipine.
Key Metrics
Top Categories
Indian Suppliers
Related Trade Data
Need Detailed Data?
Shipment-level records, supplier connections & pricing for B Logistik Gmbh.
Request DemoB Logistik Gmbh — Corporate Profile & Information
Company type, headquarters, distribution network, and industry role
1Company Overview
B Logistik GmbH is a logistics company headquartered in Ennigerloh, North Rhine-Westphalia, Germany. Established in the early 2000s, the company specializes in the transportation and storage of sensitive goods, including pharmaceuticals. With a workforce of approximately 200 employees, B Logistik GmbH has developed a reputation for reliability and efficiency in the logistics sector.
In the pharmaceutical distribution landscape, B Logistik GmbH operates as a key player, focusing on the import and distribution of finished pharmaceutical formulations. Their primary role involves sourcing pharmaceutical products from international markets, particularly India, and ensuring their delivery to various stakeholders within Germany's healthcare system. This includes supplying hospitals, pharmacies, and other healthcare providers with essential medications.
2Distribution Network
B Logistik GmbH's distribution network is strategically designed to ensure efficient and timely delivery of pharmaceutical products across Germany. The company operates multiple warehouses equipped with advanced temperature-controlled facilities to maintain the integrity of sensitive pharmaceutical products. These facilities are located in key regions, facilitating rapid distribution to major cities and healthcare centers nationwide.
Logistically, B Logistik GmbH employs a fleet of modern vehicles capable of handling temperature-sensitive shipments, ensuring compliance with Good Distribution Practice (GDP) standards. Their geographic coverage extends beyond Germany, with established routes to neighboring European countries, enabling the company to meet the diverse needs of its clientele.
3Industry Role
Within Germany's pharmaceutical supply chain, B Logistik GmbH functions primarily as a pharmaceutical importer and distributor. By sourcing finished pharmaceutical formulations from international markets, especially India, the company plays a crucial role in enhancing the availability of essential medications within Germany. Their operations support the healthcare system by ensuring a steady supply of quality-assured pharmaceutical products to various healthcare providers.
Supplier Relationship Intelligence — B Logistik Gmbh
Sourcing concentration, supply chain resilience, and strategic implications
1Sourcing Concentration Analysis
B Logistik GmbH's sourcing strategy exhibits a high degree of concentration, with a significant portion of their pharmaceutical imports originating from a single supplier, CIPLA LIMITED. This concentrated sourcing approach suggests a strategic partnership aimed at ensuring consistent product quality and supply reliability. The substantial import value of $19.4 million from CIPLA LIMITED, encompassing 2,747 shipments, indicates a stable and long-term relationship between the two entities.
While this single-source dependency may present risks related to supply chain disruptions, B Logistik GmbH's extensive experience and established relationship with CIPLA LIMITED likely mitigate such concerns. The company's focus on a limited number of therapeutic categories, particularly cardiovascular medications like Telmisartan and Amlodipine, further underscores the strategic nature of this sourcing concentration.
2Supply Chain Resilience
B Logistik GmbH's supply chain resilience is bolstered by its strategic partnership with CIPLA LIMITED, a reputable Indian pharmaceutical manufacturer. This collaboration ensures access to a consistent supply of high-quality finished pharmaceutical formulations. The company's focus on a limited number of therapeutic categories, such as cardiovascular medications, allows for streamlined logistics and inventory management, enhancing overall supply chain efficiency.
However, the heavy reliance on a single supplier may expose B Logistik GmbH to risks associated with supply chain disruptions. To mitigate these risks, the company may consider diversifying its supplier base and exploring alternative sourcing options. Additionally, maintaining robust relationships with multiple suppliers can provide a buffer against potential supply chain challenges.
3Strategic Implications
B Logistik GmbH's concentrated sourcing strategy positions the company to leverage strong partnerships with key suppliers, ensuring consistent product quality and supply reliability. This approach can lead to favorable pricing and preferential treatment from suppliers, enhancing the company's competitive edge in the German pharmaceutical market.
For Indian pharmaceutical exporters, the established relationship between B Logistik GmbH and CIPLA LIMITED may present challenges in penetrating this specific supply chain. However, opportunities exist for exporters offering complementary products or those that can meet the specific quality and regulatory standards required by B Logistik GmbH. Engaging in direct negotiations and demonstrating compliance with German pharmaceutical regulations can facilitate entry into this market segment.
Importing Pharmaceuticals into Germany — Regulatory Framework
Regulatory authority, GMP requirements, import licensing for Germany
1Regulatory Authority & Framework
In Germany, the Federal Institute for Drugs and Medical Devices (BfArM) serves as the primary regulatory authority overseeing the approval and monitoring of pharmaceutical products. The German Medicinal Products Act (Arzneimittelgesetz - AMG) provides the legal framework governing the import, distribution, and marketing of medicinal products within the country. (zoll.de)
For Indian pharmaceutical exporters, obtaining marketing authorization from the European Medicines Agency (EMA) is a critical step in entering the German market. The EMA evaluates and approves medicinal products for the entire European Union, ensuring they meet stringent safety, efficacy, and quality standards. Once approved by the EMA, products can be marketed in Germany, subject to compliance with national regulations.
2Import Licensing & GMP
Importing pharmaceutical products into Germany requires compliance with specific licensing and certification requirements. Importers must obtain an import license in accordance with Section 72 of the German Medicinal Products Act (AMG). Additionally, manufacturers outside the European Union must provide a Good Manufacturing Practice (GMP) certificate to demonstrate adherence to internationally recognized quality standards. (bosporus24.de)
Importers must also hold a wholesale distribution authorization, ensuring they meet the necessary regulatory standards for the storage and distribution of pharmaceutical products. This authorization is essential for maintaining the integrity and safety of medicinal products throughout the supply chain.
3Quality & Labeling
Pharmaceutical products imported into Germany must undergo batch testing to verify their quality, safety, and efficacy. This process ensures that all medicinal products meet the stringent standards set by German regulatory authorities. (zoll.de)
Stability requirements dictate that pharmaceutical products maintain their quality and efficacy throughout their shelf life. Proper storage conditions, including temperature control, are essential to preserve product integrity. Labeling must be in German, providing clear and accurate information to consumers and healthcare professionals. Serialization mandates, as outlined in the Falsified Medicines Directive (FMD), require unique identifiers on packaging to prevent counterfeit products from entering the supply chain.
4Recent Regulatory Changes
Between 2024 and 2026, Germany implemented several regulatory changes affecting pharmaceutical imports. These include stricter compliance requirements for Good Distribution Practice (GDP), enhanced serialization protocols to combat counterfeit medicines, and updated labeling standards to improve patient safety. Importers must stay informed about these changes to ensure ongoing compliance and maintain the integrity of their supply chains.
B Logistik Gmbh — Procurement Pattern Analysis
Product strategy, sourcing profile, and market positioning
1Product Strategy
B Logistik GmbH's focus on importing cardiovascular medications, specifically Telmisartan and Amlodipine, aligns with the significant demand for these drugs in Germany. Cardiovascular diseases are prevalent, necessitating a steady supply of effective treatments. By concentrating on these therapeutic areas, B Logistik GmbH addresses a critical segment of the healthcare market, ensuring that essential medications are readily available to patients and healthcare providers.
2Sourcing Profile
B Logistik GmbH's sourcing strategy emphasizes the procurement of generic drugs, particularly those manufactured by CIPLA LIMITED in India. The company's preference for finished pharmaceutical formulations allows for streamlined logistics and compliance with regulatory standards. India's robust pharmaceutical manufacturing sector, known for its adherence to international quality standards, makes it a strategic sourcing partner for B Logistik GmbH.
3Market Positioning
By importing and distributing essential cardiovascular medications, B Logistik GmbH serves a critical role in Germany's pharmaceutical supply chain. The company primarily caters to wholesale distribution, supplying hospitals, pharmacies, and other healthcare providers with necessary treatments. This positioning underscores B Logistik GmbH's commitment to enhancing patient care through the reliable delivery of quality-assured pharmaceutical products.
Seller's Guide — How to Become a Supplier to B Logistik Gmbh
Opportunity assessment, qualifications, and practical approach strategy
1Opportunity Assessment
For new Indian suppliers, there is a realistic opportunity to engage with B Logistik GmbH by offering complementary products or those that meet specific quality and regulatory standards required by the German market. Demonstrating compliance with German pharmaceutical regulations and establishing direct communication can facilitate entry into this market segment. However, the existing strong partnership between B Logistik GmbH and CIPLA LIMITED may present challenges for new entrants.
2Requirements & Qualifications
Indian exporters aiming to supply B Logistik GmbH and the German market must obtain marketing authorization from the European Medicines Agency (EMA). Additionally, they must provide a Good Manufacturing Practice (GMP) certificate to demonstrate adherence to internationally recognized quality standards. Compliance with German labeling requirements, including the use of the German language and adherence to serialization mandates, is also essential.
3How to Approach
Indian exporters should initiate contact with B Logistik GmbH by presenting their product portfolio and demonstrating compliance with German pharmaceutical regulations. Participating in relevant industry trade fairs and establishing a local presence can enhance visibility and facilitate relationship-building. Understanding the regulatory filing process and preparing for potential audits or inspections will be crucial for successful market entry. Setting realistic timelines and maintaining open communication will aid in navigating
Frequently Asked Questions — B Logistik Gmbh
What products does B Logistik Gmbh import from India?
B Logistik Gmbh imports 2 pharmaceutical products across 1 categories. Top imports: Telmisartan ($77.0M), Amlodipine ($61.4M).
Who supplies pharmaceuticals to B Logistik Gmbh from India?
B Logistik Gmbh sources from 1 verified Indian suppliers. The primary supplier is Cipla Limited (100.0% of imports, $19.4M).
What is B Logistik Gmbh's total pharmaceutical import value?
B Logistik Gmbh's total pharmaceutical import value from India is $138.4M, based on 2,768 verified shipments in Indian Customs (DGFT) data.
What therapeutic categories does B Logistik Gmbh focus on?
B Logistik Gmbh imports across 1 categories. The largest: Cardiovascular (100.0%).
Get Full B Logistik Gmbh Import Intelligence
Access shipment-level details, supplier connections, pricing data, and competitive analysis. TransData Nexus provides verified Indian Customs (DGFT) data trusted by pharmaceutical trade professionals worldwide.
Official References & Regulatory Resources
Verify import regulations and drug registration requirements with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Buyer Matching: B Logistik Gmbh identified across shipments using consignee name normalization.
- 2.Statistical Normalization: Shipment values are statistically normalized to ensure accurate market share representation.
- 3.Market Share: Calculated per product as B Logistik Gmbh's capped value divided by total Indian exports for that product.
- 4.Shipment Count: Based on 2,768 individual customs records matching B Logistik Gmbh.
- 5.Supplier Verification: B Logistik Gmbh sources from 1 verified Indian suppliers across 711 formulations, confirmed from customs records.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
2 Products Tracked
1 therapeutic categories
Expert-Reviewed
By pharmaceutical trade specialists
Data Source & Methodology
Trade data sourced from Indian Customs (DGFT) export shipment records. Values represent FOB export value in USD. For current shipment-level data, contact TransData Nexus.