Astra Pharma (T) Ltd
Pharmaceutical Importer · Tanzania · Antibiotics Focus · $931.5K Total Trade · DGFT Verified
Astra Pharma (T) Ltd is a pharmaceutical importer based in Tanzania with a total trade value of $931.5K across 2 products in 2 therapeutic categories. Based on 26 verified import shipments from Indian Customs (DGFT) records, the company actively imports across multiple product segments. Astra Pharma (T) Ltd sources from 5 verified Indian suppliers, with Ajanta Pharma Limited accounting for 55.7% of imports.
Astra Pharma (T) Ltd — Import Portfolio & Supplier Network
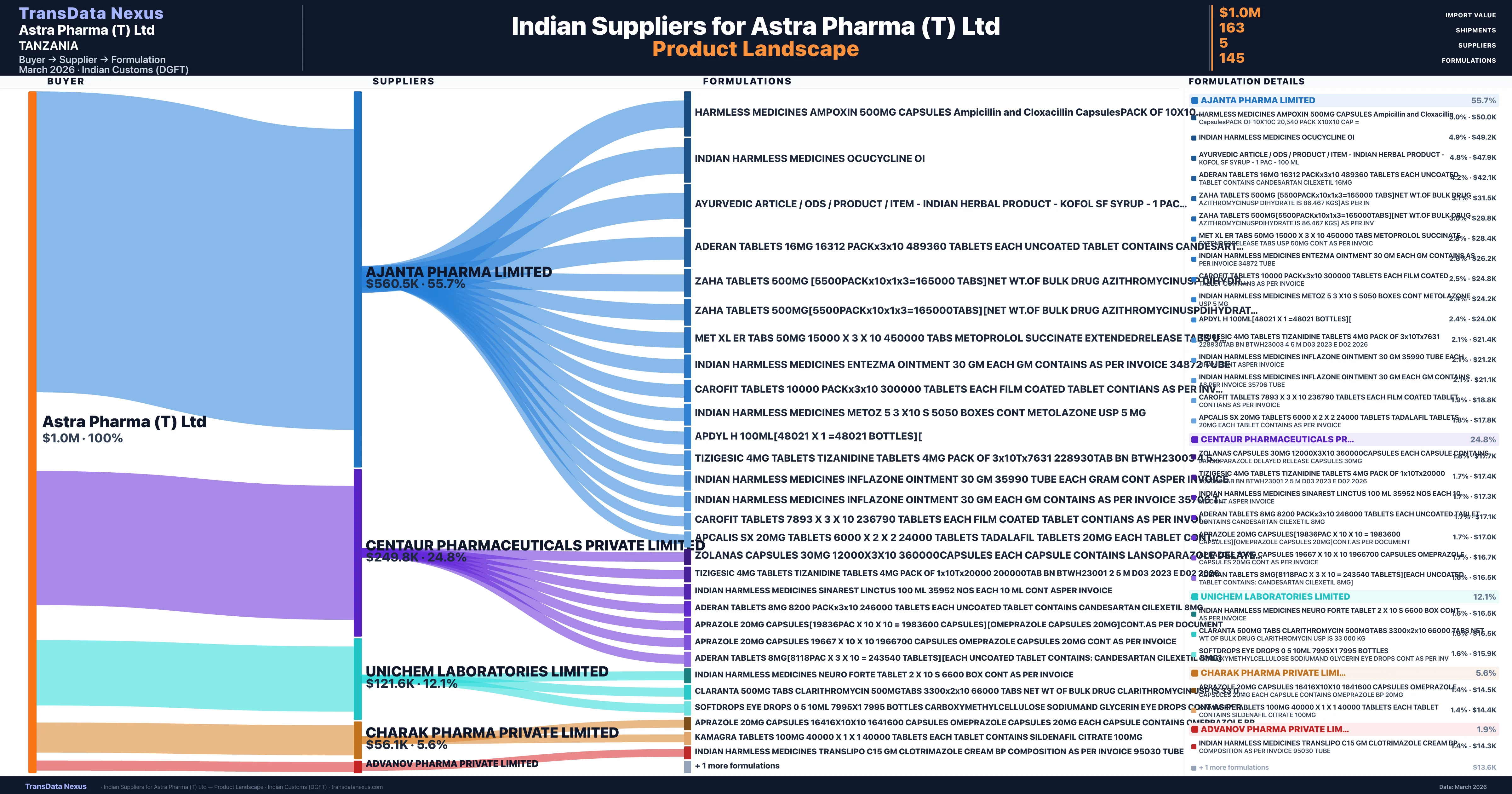
Who Are the Verified Indian Suppliers to Astra Pharma (T) Ltd?
Customs-verified supplier relationships from Indian DGFT records
| Supplier | Value | Shipments | Share |
|---|---|---|---|
| Ajanta Pharma Limited | $560.5K | 103 | 55.7% |
| Centaur Pharmaceuticals Private Limited | $249.8K | 40 | 24.8% |
| Unichem Laboratories Limited | $121.6K | 13 | 12.1% |
| Charak Pharma Private Limited | $56.1K | 5 | 5.6% |
| Advanov Pharma Private Limited | $18.7K | 2 | 1.9% |
Astra Pharma (T) Ltd sources from 5 verified Indian suppliers across 145 distinct formulations. The supply base is diversified across 5 suppliers, reducing single-source dependency risk.
What Formulations Does Astra Pharma (T) Ltd Import?
| Formulation | Value | Ships |
|---|---|---|
| Harmless medicines ampoxin 500MG capsules ampicillin and cloxacillin capsulespack of 10x10c 20,540 Pack x10x10 cap | $50.0K | 1 |
| Indian harmless medicines ocucycline | $49.2K | 2 |
| Ayurvedic article / ods / product / item - indian herbal product - kofol sf syrup - 1 pac - 100 ML | $47.9K | 1 |
| Aderan tablets 16MG 16312 packx3x10 489360 tablets Each uncoated tablet contains candesartan cilexetil 16MG | $42.1K | 1 |
| Zaha tablets 500MG [5500packx10x1x3=165000 tabs]net wt.of bulk drug azithromycinusp dihydrate is 86.467 kgs]as per | $31.5K | 1 |
| Zaha tablets 500MG[5500packx10x1x3=165000tabs][net wt.of bulk drug azithromycinuspdihydrate is 86.467 kgs] as per | $29.8K | 1 |
| Met xl er tabs 50MG 15000 x 3 x 10 450000 tabs metoprolol succinate extendedrelease tabs USP 50MG cont as per | $28.4K | 1 |
| Indian harmless medicines entezma ointment 30 GM Each GM contains as per invoice 34872 | $26.2K | 1 |
| Carofit tablets 10000 packx3x10 300000 tablets Each film coated tablet contians as per | $24.8K | 1 |
| Indian harmless medicines metoz 5 3 x10 s 5050 boxes cont metolazone USP 5 MG | $24.2K | 1 |
| Apdyl h 100ML[48021 x 1 =48021 | $24.0K | 1 |
| Tizigesic 4MG tablets tizanidine tablets 4MG Pack of 3x10tx7631 228930tab bn btwh23003 4 5 m d03 2023 e d02 | $21.4K | 1 |
| Indian harmless medicines inflazone ointment 30 GM 35990 tube Each gram cont asper | $21.2K | 1 |
| Indian harmless medicines inflazone ointment 30 GM Each GM contains as per invoice 35706 | $21.1K | 1 |
| Carofit tablets 7893 x 3 x 10 236790 tablets Each film coated tablet contians as per | $18.8K | 1 |
Astra Pharma (T) Ltd imports 145 distinct pharmaceutical formulations. Showing top 15 by value. For full formulation-level data, contact TransData Nexus.
What Products Does Astra Pharma (T) Ltd Import?
Top Products by Import Value
Astra Pharma (T) Ltd Therapeutic Categories — 2 Specializations
Astra Pharma (T) Ltd imports across 2 therapeutic categories, with Antibiotics (96.6%), Advanced Oncology (3.4%), representing the largest segments. The portfolio is concentrated — top 5 products = 100% of total imports.
Antibiotics
1 products · 96.6% · $900.0K
Advanced Oncology
1 products · 3.4% · $31.5K
Import Portfolio — Top 2 by Import Value
| # | Product | Category | Value | Ships | Share | Rk |
|---|---|---|---|---|---|---|
| 1 | Clarithromycin | Antibiotics | $900.0K | 18 | 0.9% | 6 |
| 2 | Tofacitinib | Advanced Oncology | $31.5K | 8 | 0.8% | 8 |
Astra Pharma (T) Ltd imports 2 pharmaceutical products across 2 categories into Tanzania totaling $931.5K.
Key Metrics
Top Categories
Indian Suppliers
Related Trade Data
Need Detailed Data?
Shipment-level records, supplier connections & pricing for Astra Pharma (T) Ltd.
Request DemoAstra Pharma (T) Ltd — Corporate Profile & Information
Company type, headquarters, distribution network, and industry role
1Company Overview
Astra Pharma (T) Ltd is a prominent pharmaceutical importer and wholesaler based in Dar es Salaam, Tanzania. Established in 2002 as part of the Astra Group, the company has significantly contributed to the Tanzanian healthcare sector by providing quality pharmaceutical products. The Astra Group, founded in 1996, has a longstanding presence in East Africa, with operations in both Uganda and Tanzania.
The company operates as a primary wholesaler, sourcing a diverse range of pharmaceutical formulations from international suppliers, particularly from India. Astra Pharma (T) Ltd plays a crucial role in the distribution network, ensuring the availability of essential medications across Tanzania. Their extensive distribution network and partnerships with multinational companies underscore their commitment to enhancing healthcare access in the region.
2Distribution Network
Astra Pharma (T) Ltd's distribution network is centered in Dar es Salaam, the commercial hub of Tanzania. The company's headquarters are located at Block 6, House 35, Uhuru/Congo Street, Kariakoo, P.O. Box No. 38388, Dar es Salaam. While specific details about additional warehouse locations and logistics capabilities are not publicly disclosed, the strategic positioning in Dar es Salaam suggests a focus on efficient distribution throughout Tanzania. The company's partnerships with reputable multinational pharmaceutical companies further enhance its logistics capabilities, ensuring a steady supply of quality medications to meet the country's healthcare needs.
3Industry Role
Astra Pharma (T) Ltd serves as a primary wholesaler in Tanzania's pharmaceutical supply chain. By importing a wide array of pharmaceutical formulations, the company acts as a critical intermediary between international manufacturers and local healthcare providers. This role is vital in ensuring the consistent availability of essential medications across the country, thereby supporting the overall healthcare infrastructure. The company's extensive distribution network and partnerships with multinational companies underscore its significant position in the Tanzanian pharmaceutical market.
Supplier Relationship Intelligence — Astra Pharma (T) Ltd
Sourcing concentration, supply chain resilience, and strategic implications
1Sourcing Concentration Analysis
Astra Pharma (T) Ltd's sourcing strategy exhibits a high degree of concentration, with a total import value of $931,000 USD from India across 26 shipments. The top five products imported are Clarithromycin ($900,000, 0.9% share) and Tofacitinib ($31,000, 0.8% share), indicating a focused product portfolio. This concentration suggests a strategic choice to streamline operations and maintain strong supplier relationships. However, such dependency on a limited number of suppliers and products could pose risks, including supply chain disruptions or market fluctuations affecting these specific products. The shipment data reveals a stable relationship with Indian suppliers, as evidenced by the consistent import volumes over the years. Nonetheless, diversifying the supplier base and product range could enhance resilience against potential supply chain challenges.
2Supply Chain Resilience
Astra Pharma (T) Ltd's supply chain resilience is closely tied to its partnerships with Indian suppliers, notably AJANTA PHARMA LIMITED, CENTAUR PHARMACEUTICALS PRIVATE LIMITED, and UNICHEM LABORATORIES LIMITED. The company's importation of finished pharmaceutical formulations, rather than raw APIs or bulk drugs, indicates a focus on ready-to-market products. While this approach simplifies logistics and regulatory compliance, it also means that Astra Pharma (T) Ltd is highly dependent on the stability and reliability of its Indian suppliers. The absence of publicly available information on backup suppliers and the company's overall formulation diversity suggests potential vulnerabilities in the supply chain. To enhance resilience, Astra Pharma (T) Ltd could consider diversifying its supplier base and exploring alternative sourcing options.
3Strategic Implications
Astra Pharma (T) Ltd's concentrated sourcing pattern positions it as a key player in the Tanzanian pharmaceutical market, particularly in the antibiotic and advanced oncology therapeutic categories. This focus allows the company to leverage strong supplier relationships and maintain a streamlined product portfolio. For Indian exporters, this presents an opportunity to strengthen existing partnerships and explore avenues for introducing additional products within these therapeutic areas. However, the company's reliance on a limited number of suppliers and products also highlights the importance of ensuring consistent product quality and supply reliability to maintain its competitive position.
Importing Pharmaceuticals into Tanzania — Regulatory Framework
Regulatory authority, GMP requirements, import licensing for Tanzania
1Regulatory Authority & Framework
The Tanzania Medicines and Medical Devices Authority (TMDA) is the primary regulatory body overseeing the importation and exportation of pharmaceutical products in Tanzania. Established under the Tanzania Medicines and Medical Devices Act Cap. 219, TMDA is responsible for ensuring the safety, efficacy, and quality of medicines and medical devices in the country. The Act provides TMDA with the mandate to regulate the importation and exportation of medicines, medical devices, in vitro diagnostics, and biocidals. (tmda.go.tz)
Key legislation governing pharmaceutical imports includes the Pharmaceutical and Poisons Act and the Pharmaceutical and Poisons Regulations, which outline the control of manufacture, sales, import, and export of pharmaceutical products. The marketing authorization pathway for Indian generics involves obtaining import permits from TMDA, ensuring compliance with the regulatory standards set forth by the authority.
2Import Licensing & GMP
Import licensing requirements in Tanzania stipulate that all pharmaceutical products to be imported must be registered by TMDA unless given special approval by the Authority. Importers must register their premises with TMDA before applying for import permits. Applications for import permits must be submitted at least six weeks prior to loading the consignment to avoid storage costs at ports of entry and disposal costs of unpermitted products. (tmda.go.tz)
Regarding Good Manufacturing Practice (GMP) certification, TMDA recognizes certifications from facilities that comply with international standards, including EU GMP, WHO GMP, and PIC/S. Importers must ensure that the pharmaceutical products they import are manufactured in facilities holding such recognized certifications. Additionally, importers must obtain wholesale distribution authorization from TMDA, which involves meeting specific regulatory requirements and maintaining compliance with the Authority's guidelines.
3Quality & Labeling
Imported pharmaceutical products must undergo batch testing to ensure they meet the required quality standards set by TMDA. Stability requirements are enforced to guarantee that products remain effective and safe throughout their shelf life. Labeling language requirements mandate that product labels be in English and Kiswahili, the official languages of Tanzania, to ensure clear communication with consumers and healthcare providers. Serialization mandates are implemented to enhance traceability and prevent counterfeit products from entering the market, thereby safeguarding public health.
4Recent Regulatory Changes
Between 2024 and 2026, TMDA has implemented several policy changes affecting pharmaceutical imports into Tanzania. These include stricter enforcement of import permit requirements, enhanced scrutiny of GMP certifications, and the introduction of more rigorous batch testing protocols. Additionally, TMDA has updated labeling and serialization mandates to align with international standards and improve product traceability. These regulatory changes aim to strengthen the quality assurance framework and ensure the safety and efficacy of pharmaceutical products in the Tanzanian market.
Astra Pharma (T) Ltd — Procurement Pattern Analysis
Product strategy, sourcing profile, and market positioning
1Product Strategy
Astra Pharma (T) Ltd's product strategy focuses on importing finished pharmaceutical formulations, particularly in the antibiotic and advanced oncology therapeutic categories. This focus aligns with the company's objective to provide essential medications that address prevalent health concerns in Tanzania. The market demand for these products is driven by the high incidence of infectious diseases and cancer in the region, necessitating a reliable supply of effective treatments. By concentrating on these therapeutic areas, Astra Pharma (T) Ltd aims to meet critical healthcare needs and strengthen its position in the Tanzanian pharmaceutical market.
2Sourcing Profile
Astra Pharma (T) Ltd's sourcing strategy is centered on importing generic drugs from India, leveraging the country's robust pharmaceutical manufacturing sector. The company's preference for finished formulations indicates a focus on ready-to-market products, which simplifies logistics and regulatory compliance. India's compliance with international GMP standards, including EU GMP, WHO GMP, and PIC/S, aligns with TMDA's regulatory requirements, facilitating smoother import processes. This sourcing approach enables Astra Pharma (T) Ltd to offer a diverse range of quality medications to the Tanzanian market.
3Market Positioning
Based on its product mix, Astra Pharma (T) Ltd primarily serves the wholesale distribution segment of the Tanzanian pharmaceutical market. By importing and distributing a focused range of pharmaceutical formulations, the company acts as a critical intermediary between international manufacturers and local healthcare providers. This positioning allows Astra Pharma (T) Ltd to ensure the consistent availability of essential medications across the country, thereby supporting the overall healthcare infrastructure.
Seller's Guide — How to Become a Supplier to Astra Pharma (T) Ltd
Opportunity assessment, qualifications, and practical approach strategy
1Opportunity Assessment
There is a realistic opportunity for new Indian suppliers to collaborate with Astra Pharma (T) Ltd, particularly in the antibiotic and advanced oncology therapeutic categories. The company's concentrated sourcing pattern indicates a potential openness to expanding its supplier base within these areas. Identifying gaps in Astra Pharma (T) Ltd's current sourcing, such as the need for additional product formulations or alternative suppliers, could present avenues for new suppliers to enter the market.
Frequently Asked Questions — Astra Pharma (T) Ltd
What products does Astra Pharma (T) Ltd import from India?
Astra Pharma (T) Ltd imports 2 pharmaceutical products across 2 categories. Top imports: Clarithromycin ($900.0K), Tofacitinib ($31.5K).
Who supplies pharmaceuticals to Astra Pharma (T) Ltd from India?
Astra Pharma (T) Ltd sources from 5 verified Indian suppliers. The primary supplier is Ajanta Pharma Limited (55.7% of imports, $560.5K).
What is Astra Pharma (T) Ltd's total pharmaceutical import value?
Astra Pharma (T) Ltd's total pharmaceutical import value from India is $931.5K, based on 26 verified shipments in Indian Customs (DGFT) data.
What therapeutic categories does Astra Pharma (T) Ltd focus on?
Astra Pharma (T) Ltd imports across 2 categories. The largest: Antibiotics (96.6%), Advanced Oncology (3.4%).
Get Full Astra Pharma (T) Ltd Import Intelligence
Access shipment-level details, supplier connections, pricing data, and competitive analysis. TransData Nexus provides verified Indian Customs (DGFT) data trusted by pharmaceutical trade professionals worldwide.
Official References & Regulatory Resources
Verify import regulations and drug registration requirements with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Buyer Matching: Astra Pharma (T) Ltd identified across shipments using consignee name normalization.
- 2.Statistical Normalization: Shipment values are statistically normalized to ensure accurate market share representation.
- 3.Market Share: Calculated per product as Astra Pharma (T) Ltd's capped value divided by total Indian exports for that product.
- 4.Shipment Count: Based on 26 individual customs records matching Astra Pharma (T) Ltd.
- 5.Supplier Verification: Astra Pharma (T) Ltd sources from 5 verified Indian suppliers across 145 formulations, confirmed from customs records.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
2 Products Tracked
2 therapeutic categories
Expert-Reviewed
By pharmaceutical trade specialists
Data Source & Methodology
Trade data sourced from Indian Customs (DGFT) export shipment records. Values represent FOB export value in USD. For current shipment-level data, contact TransData Nexus.