Aorta Company Limited
Pharmaceutical Importer · Myanmar · Antibiotics Focus · $4.0M Total Trade · DGFT Verified
Aorta Company Limited is a pharmaceutical importer based in Myanmar with a total trade value of $4.0M across 2 products in 2 therapeutic categories. Based on 88 verified import shipments from Indian Customs (DGFT) records, the company actively imports across multiple product segments. Aorta Company Limited sources from 5 verified Indian suppliers, with Amn Life Science Private Limited accounting for 64.5% of imports.
Aorta Company Limited — Import Portfolio & Supplier Network
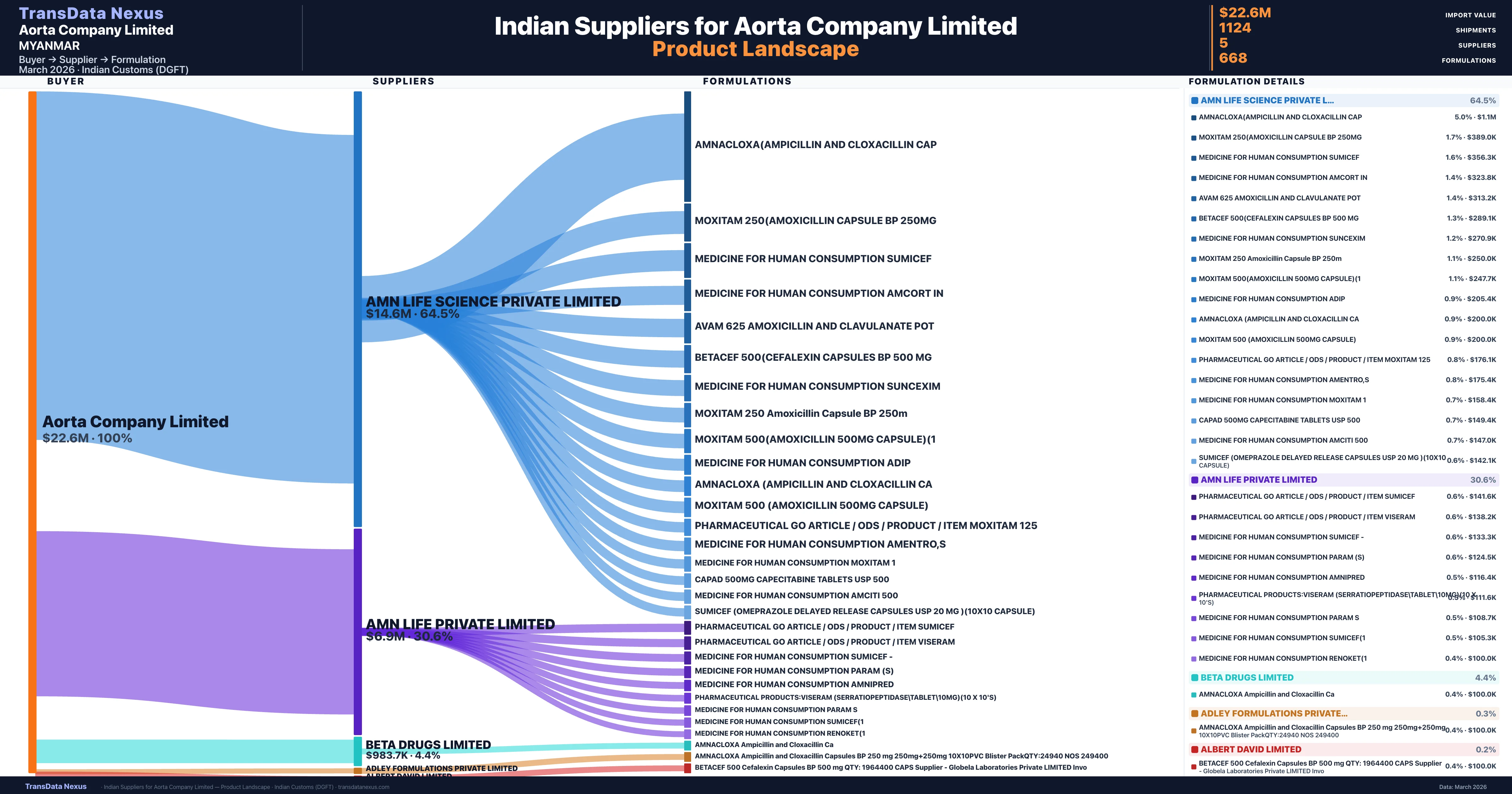
Who Are the Verified Indian Suppliers to Aorta Company Limited?
Customs-verified supplier relationships from Indian DGFT records
| Supplier | Value | Shipments | Share |
|---|---|---|---|
| Amn Life Science Private Limited | $14.6M | 659 | 64.5% |
| Amn Life Private Limited | $6.9M | 293 | 30.6% |
| Beta Drugs Limited | $983.7K | 162 | 4.4% |
| Adley Formulations Private Limited | $77.2K | 7 | 0.3% |
| Albert David Limited | $53.3K | 3 | 0.2% |
Aorta Company Limited sources from 5 verified Indian suppliers across 668 distinct formulations. The supply base is diversified across 5 suppliers, reducing single-source dependency risk.
What Formulations Does Aorta Company Limited Import?
| Formulation | Value | Ships |
|---|---|---|
| Amnacloxa(ampicillin and cloxacillin | $1.1M | 26 |
| Moxitam 250(amoxicillin capsule BP 250MG | $389.0K | 12 |
| Medicine for human consumption | $356.3K | 17 |
| Medicine for human consumption amcort | $323.8K | 17 |
| Avam 625 amoxicillin and clavulanate | $313.2K | 11 |
| Betacef 500(cefalexin capsules BP 500 MG | $289.1K | 8 |
| Medicine for human consumption | $270.9K | 13 |
| Moxitam 250 amoxicillin capsule BP | $250.0K | 5 |
| Moxitam 500(amoxicillin 500MG capsule | $247.7K | 8 |
| Medicine for human consumption | $205.4K | 7 |
| Amnacloxa (ampicillin and cloxacillin | $200.0K | 4 |
| Moxitam 500 (amoxicillin 500MG capsule) | $200.0K | 4 |
| Pharmaceutical go article / ods / product / item moxitam | $176.1K | 8 |
| Medicine for human consumption amentro | $175.4K | 13 |
| Medicine for human consumption moxitam | $158.4K | 15 |
Aorta Company Limited imports 668 distinct pharmaceutical formulations. Showing top 15 by value. For full formulation-level data, contact TransData Nexus.
What Products Does Aorta Company Limited Import?
Top Products by Import Value
Aorta Company Limited Therapeutic Categories — 2 Specializations
Aorta Company Limited imports across 2 therapeutic categories, with Antibiotics (91.7%), Gastrointestinal (8.3%), representing the largest segments. The portfolio is concentrated — top 5 products = 100% of total imports.
Antibiotics
1 products · 91.7% · $3.7M
Gastrointestinal
1 products · 8.3% · $336.8K
Import Portfolio — Top 2 by Import Value
| # | Product | Category | Value | Ships | Share | Rk |
|---|---|---|---|---|---|---|
| 1 | Ampicillin | Antibiotics | $3.7M | 74 | 2.6% | 3 |
| 2 | Ranitidine | Gastrointestinal | $336.8K | 14 | 2.7% | 6 |
Aorta Company Limited imports 2 pharmaceutical products across 2 categories into Myanmar totaling $4.0M.
Key Metrics
Top Categories
Indian Suppliers
Related Trade Data
Need Detailed Data?
Shipment-level records, supplier connections & pricing for Aorta Company Limited.
Request DemoAorta Company Limited — Corporate Profile & Information
Company type, headquarters, distribution network, and industry role
1Company Overview
Aorta Company Limited is a pharmaceutical importer based in Myanmar, specializing in the procurement of finished pharmaceutical formulations from India. The company operates as a buyer, sourcing a diverse range of pharmaceutical products to meet the healthcare needs within Myanmar. While specific details about its headquarters and parent company are not publicly disclosed, Aorta Company Limited plays a significant role in the distribution of imported pharmaceutical products across the country.
2Distribution Network
Information regarding Aorta Company Limited's warehouse locations and logistics capabilities is not publicly available. However, given its substantial import activities, it is likely that the company has established a distribution network within Myanmar to effectively manage the delivery of pharmaceutical products. The extent of its geographic coverage beyond Myanmar remains unspecified.
3Industry Role
Aorta Company Limited functions primarily as a pharmaceutical importer, sourcing finished pharmaceutical formulations from Indian suppliers. Its role in Myanmar's pharmaceutical supply chain involves facilitating the availability of imported medications, thereby contributing to the country's healthcare sector. The company's specific position—whether as a wholesaler, parallel importer, hospital supplier, or logistics intermediary—cannot be conclusively determined based on the available data.
Supplier Relationship Intelligence — Aorta Company Limited
Sourcing concentration, supply chain resilience, and strategic implications
1Sourcing Concentration Analysis
Aorta Company Limited demonstrates a high degree of sourcing concentration, with its top five products accounting for 100% of its imports. This indicates a strategic focus on a limited product range, which may streamline operations but also exposes the company to risks associated with supply chain disruptions or changes in market demand for these specific products. The substantial import values and consistent shipment volumes suggest a stable and ongoing relationship with its Indian suppliers, particularly with AMN Life Science Private Limited and AMN Life Private Limited, which together account for 95.1% of the total import value.
2Supply Chain Resilience
The resilience of Aorta Company Limited's supply chain appears to be robust, given the consistent import volumes and the dominance of a few key suppliers. However, the lack of publicly available information on backup suppliers and the diversity of formulations imported makes it challenging to fully assess the company's preparedness for potential supply chain disruptions. The company's reliance on a limited number of suppliers may pose risks if any of these suppliers face operational challenges or regulatory issues.
3Strategic Implications
Aorta Company Limited's concentrated sourcing strategy positions it as a focused player in the Myanmar pharmaceutical market, potentially allowing for better negotiation terms and streamlined operations. For Indian exporters, this presents an opportunity to establish strong partnerships with Aorta Company Limited by offering high-quality products that align with the company's current import portfolio. Diversifying the product range could also be a strategic move to mitigate risks associated with supply chain disruptions.
Importing Pharmaceuticals into Myanmar — Regulatory Framework
Regulatory authority, GMP requirements, import licensing for Myanmar
1Regulatory Authority & Framework
The Food and Drug Administration (FDA) of Myanmar is the primary regulatory body overseeing the importation, registration, and distribution of pharmaceutical products in the country. The National Drug Law (Law No. 7/1992), enacted on October 30, 1992, serves as the foundational legislation governing these activities. This law outlines the procedures for drug registration, licensing, quality assurance, labeling, and advertising, aiming to ensure the safety, efficacy, and quality of pharmaceutical products in Myanmar. (wipo.int)
2Import Licensing & GMP
To import pharmaceutical products into Myanmar, companies must obtain an Import License from the FDA. This license is contingent upon the submission of comprehensive documentation, including Certificates of Pharmaceutical Product (CPP) from the exporting country, which attest to the compliance of the products with international quality standards. Additionally, the FDA requires that imported drugs adhere to Good Manufacturing Practice (GMP) standards recognized by international bodies such as the World Health Organization (WHO) and the Pharmaceutical Inspection Co-operation Scheme (PIC/S).
3Quality & Labeling
Imported pharmaceutical products must undergo batch testing to verify their quality and compliance with Myanmar's standards. The labeling of these products must include information in the Burmese language, detailing the drug's name, dosage, usage instructions, and any necessary warnings. Serialization mandates may be implemented to enhance traceability and prevent counterfeit products from entering the market. These requirements are enforced by the FDA to ensure consumer safety and product integrity.
4Recent Regulatory Changes
Between 2024 and 2026, Myanmar has implemented several policy changes affecting the importation of pharmaceutical products. These include stricter enforcement of GMP standards, enhanced scrutiny of drug registration processes, and the introduction of new labeling requirements to improve consumer information. These changes aim to bolster the quality and safety of pharmaceutical imports and may impact the operations of companies like Aorta Company Limited.
Aorta Company Limited — Procurement Pattern Analysis
Product strategy, sourcing profile, and market positioning
1Product Strategy
Aorta Company Limited's focus on importing antibiotics and gastrointestinal medications suggests a strategic alignment with prevalent health concerns in Myanmar, where infectious diseases and gastrointestinal disorders are common. The substantial import value of ampicillin indicates a significant demand for antibiotics, while the inclusion of ranitidine reflects a need for treatments addressing stomach acid-related issues. This product strategy is likely driven by market demand and the company's objective to meet the therapeutic needs of the population.
2Sourcing Profile
Aorta Company Limited's sourcing strategy is centered on generic pharmaceutical formulations, primarily from India. The company's preference for finished formulations over raw Active Pharmaceutical Ingredients (APIs) suggests a focus on ready-to-market products, which can expedite distribution and reduce operational complexities. India's established pharmaceutical manufacturing capabilities and competitive pricing make it an attractive sourcing destination for Aorta Company Limited.
3Market Positioning
Based on its product mix, Aorta Company Limited serves the wholesale distribution segment of the Myanmar pharmaceutical market. By importing a range of antibiotics and gastrointestinal medications, the company likely supplies these products to various healthcare providers, including hospitals, clinics, and pharmacies, thereby contributing to the broader healthcare infrastructure in Myanmar.
Seller's Guide — How to Become a Supplier to Aorta Company Limited
Opportunity assessment, qualifications, and practical approach strategy
1Opportunity Assessment
There is a realistic opportunity for new Indian suppliers to engage with Aorta Company Limited, particularly those offering high-quality generic formulations that align with the company's current product focus. Expanding the product range to include additional therapeutic areas could also present growth opportunities. However, potential suppliers should be prepared to navigate Myanmar's regulatory landscape and ensure compliance with the FDA's requirements to establish a successful partnership.
2Requirements & Qualifications
Indian exporters seeking to supply Aorta Company Limited must ensure that their products are registered with the FDA of Myanmar and comply with the GMP standards recognized by international bodies such as WHO and PIC/S. Additionally, products must meet the labeling requirements set forth by the FDA, including information in the Burmese language. Obtaining the necessary import licenses and adhering to quality assurance protocols are essential steps in the process.
3How to Approach
To establish a relationship with Aorta Company Limited, Indian exporters should initiate contact to understand the company's specific product requirements and quality standards. Participating in relevant tenders and trade fairs can also provide opportunities to showcase products. Developing a comprehensive regulatory filing strategy, including obtaining the necessary import licenses and ensuring compliance with FDA requirements, is crucial. Setting realistic timelines for product registration and market entry will facilitate a smoother collaboration.
Frequently Asked Questions — Aorta Company Limited
What products does Aorta Company Limited import from India?
Aorta Company Limited imports 2 pharmaceutical products across 2 categories. Top imports: Ampicillin ($3.7M), Ranitidine ($336.8K).
Who supplies pharmaceuticals to Aorta Company Limited from India?
Aorta Company Limited sources from 5 verified Indian suppliers. The primary supplier is Amn Life Science Private Limited (64.5% of imports, $14.6M).
What is Aorta Company Limited's total pharmaceutical import value?
Aorta Company Limited's total pharmaceutical import value from India is $4.0M, based on 88 verified shipments in Indian Customs (DGFT) data.
What therapeutic categories does Aorta Company Limited focus on?
Aorta Company Limited imports across 2 categories. The largest: Antibiotics (91.7%), Gastrointestinal (8.3%).
Get Full Aorta Company Limited Import Intelligence
Access shipment-level details, supplier connections, pricing data, and competitive analysis. TransData Nexus provides verified Indian Customs (DGFT) data trusted by pharmaceutical trade professionals worldwide.
Official References & Regulatory Resources
Verify import regulations and drug registration requirements with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Buyer Matching: Aorta Company Limited identified across shipments using consignee name normalization.
- 2.Statistical Normalization: Shipment values are statistically normalized to ensure accurate market share representation.
- 3.Market Share: Calculated per product as Aorta Company Limited's capped value divided by total Indian exports for that product.
- 4.Shipment Count: Based on 88 individual customs records matching Aorta Company Limited.
- 5.Supplier Verification: Aorta Company Limited sources from 5 verified Indian suppliers across 668 formulations, confirmed from customs records.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
2 Products Tracked
2 therapeutic categories
Expert-Reviewed
By pharmaceutical trade specialists
Data Source & Methodology
Trade data sourced from Indian Customs (DGFT) export shipment records. Values represent FOB export value in USD. For current shipment-level data, contact TransData Nexus.