Ambica International Corporation
Pharmaceutical Importer · Philippines · Other Focus · $5.6M Total Trade · DGFT Verified
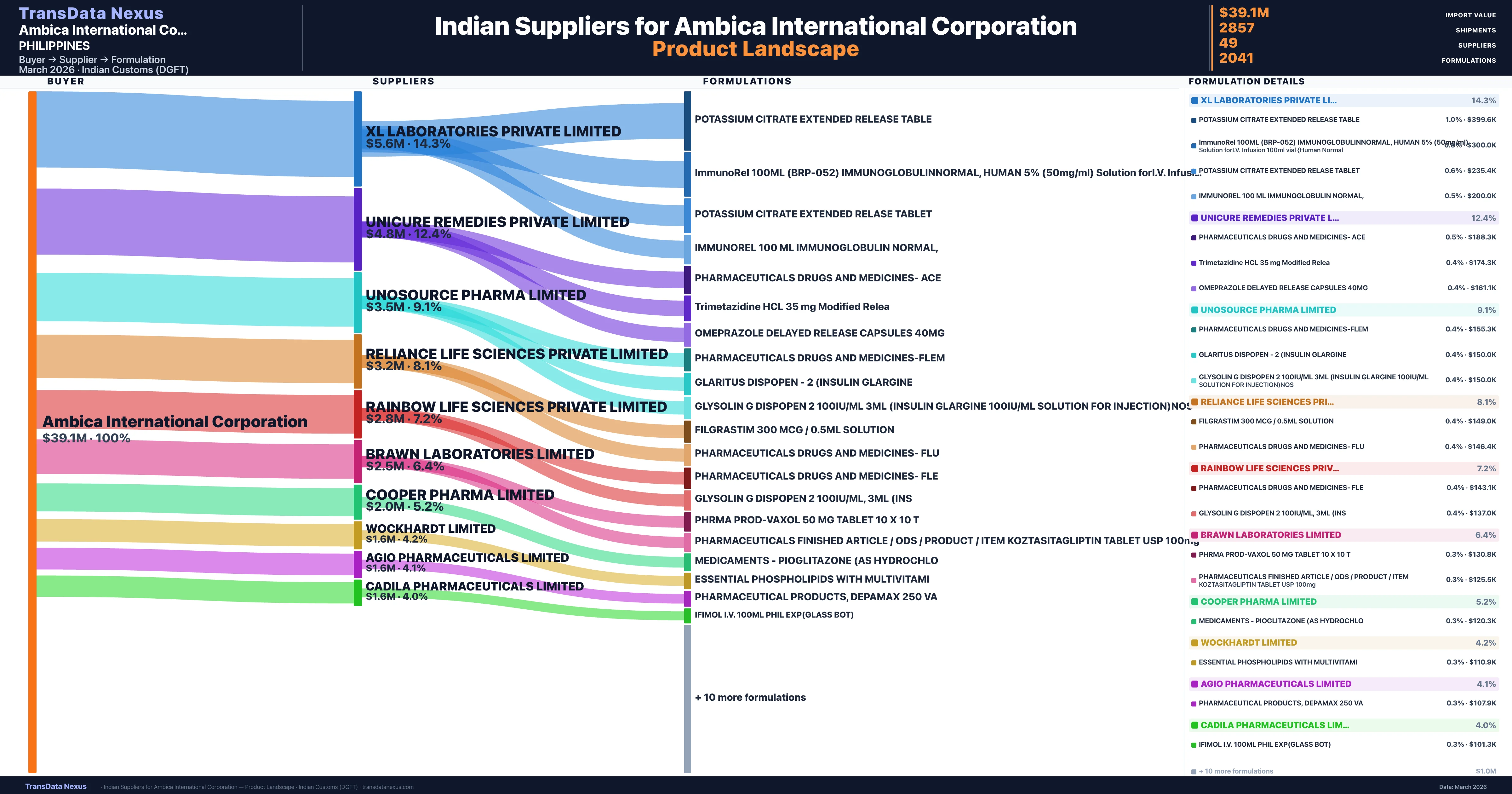
Ambica International Corporation is a pharmaceutical importer based in Philippines with a total trade value of $5.6M across 9 products in 7 therapeutic categories. Based on 300 verified import shipments from Indian Customs (DGFT) records, the company actively imports across multiple product segments. Ambica International Corporation sources from 49 verified Indian suppliers, with Xl Laboratories Private Limited accounting for 14.3% of imports.
Ambica International Corporation — Import Portfolio & Supplier Network
Who Are the Verified Indian Suppliers to Ambica International Corporation?
Customs-verified supplier relationships from Indian DGFT records
| Supplier | Value | Shipments | Share |
|---|---|---|---|
| Xl Laboratories Private Limited | $5.6M | 441 | 14.3% |
| Unicure Remedies Private Limited | $4.8M | 305 | 12.4% |
| Unosource Pharma Limited | $3.5M | 234 | 9.1% |
| Reliance Life Sciences Private Limited | $3.2M | 82 | 8.1% |
| Rainbow Life Sciences Private Limited | $2.8M | 318 | 7.2% |
| Brawn Laboratories Limited | $2.5M | 179 | 6.4% |
| Cooper Pharma Limited | $2.0M | 186 | 5.2% |
| Wockhardt Limited | $1.6M | 64 | 4.2% |
| Agio Pharmaceuticals Limited | $1.6M | 155 | 4.1% |
| Cadila Pharmaceuticals Limited | $1.6M | 113 | 4.0% |
| Medley Pharmaceuticals Limited | $1.1M | 98 | 2.7% |
| Giyaan Pharma Private Limited | $983.1K | 67 | 2.5% |
| Nabros Pharma Private Limited | $897.7K | 52 | 2.3% |
| Getwell Pharmaceuticals | $837.3K | 103 | 2.1% |
| J B Chemicals And Pharmaceuticals Limited | $538.3K | 40 | 1.4% |
| Mediwin Pharmaceuticals | $493.7K | 45 | 1.3% |
| Zeiss Pharmaceuticals Private Limited | $475.1K | 28 | 1.2% |
| Maneesh Pharmaceuticals Limited | $462.4K | 21 | 1.2% |
| Getwell Pharma India Private Limited | $405.8K | 38 | 1.0% |
| Unique Biotech Limited | $362.7K | 23 | 0.9% |
| Westcoast Pharmaceutical Works Limited | $323.6K | 22 | 0.8% |
| Synmedic Laboratories | $320.1K | 53 | 0.8% |
| Theon Pharmaceuticals Limited | $283.9K | 17 | 0.7% |
| Innova Captab Limited | $283.3K | 20 | 0.7% |
| Virchow Healthcare Private Limited | $236.0K | 11 | 0.6% |
| Neomi Pharmaceuticals Private Limited | $231.7K | 13 | 0.6% |
| Sydler Remedies Private Limited | $189.3K | 18 | 0.5% |
| Lyka Labs Limited | $175.5K | 10 | 0.4% |
| J B Chemicals Pharmaceuticals Limited | $160.7K | 11 | 0.4% |
| Admac Lifesciences | $154.0K | 12 | 0.4% |
Ambica International Corporation sources from 49 verified Indian suppliers across 2,041 distinct formulations. The supply base is diversified across 49 suppliers, reducing single-source dependency risk.
What Formulations Does Ambica International Corporation Import?
| Formulation | Value | Ships |
|---|---|---|
| Potassium citrate extended release | $399.6K | 26 |
| Immunorel 100ML (brp-052) immunoglobulinnormal, human 5% (50MG/ML) solution fori.v. infusion 100ML vial {human | $300.0K | 6 |
| Potassium citrate extended relase | $235.4K | 5 |
| Immunorel 100 ML immunoglobulin normal | $200.0K | 6 |
| Pharmaceuticals drugs and medicines- | $188.3K | 14 |
| Trimetazidine hcl 35 MG modified | $174.3K | 6 |
| Omeprazole delayed release capsules 40MG | $161.1K | 8 |
| Pharmaceuticals drugs and | $155.3K | 6 |
| Glaritus dispopen - 2 (insulin | $150.0K | 6 |
| Glysolin g dispopen 2 100iu/ML 3ML (insulin glargine 100iu/ML solution for injection | $150.0K | 3 |
| Filgrastim 300 MCG / 0.5ML | $149.0K | 3 |
| Pharmaceuticals drugs and medicines- | $146.4K | 3 |
| Pharmaceuticals drugs and medicines- | $143.1K | 3 |
| Glysolin g dispopen 2 100iu/ML, 3ML | $137.0K | 5 |
| Phrma prod-vaxol 50 MG tablet 10 x 10 | $130.8K | 3 |
Ambica International Corporation imports 2,041 distinct pharmaceutical formulations. Showing top 15 by value. For full formulation-level data, contact TransData Nexus.
What Products Does Ambica International Corporation Import?
Ambica International Corporation Therapeutic Categories — 7 Specializations
Ambica International Corporation imports across 7 therapeutic categories, with Other (44.1%), Diabetes & Endocrine (16.9%), Ayurvedic & Herbal Products (10.8%) representing the largest segments. The portfolio is concentrated — top 5 products = 82% of total imports.
Other
1 products · 44.1% · $2.5M
Diabetes & Endocrine
2 products · 16.9% · $939.0K
Ayurvedic & Herbal Products
1 products · 10.8% · $600.0K
Nutritional Supplements
2 products · 10.1% · $559.6K
Lipid & Metabolism
1 products · 8.7% · $480.7K
Oncology
1 products · 4.8% · $269.0K
Advanced Oncology
1 products · 4.6% · $255.5K
Import Portfolio — Top 9 by Import Value
| # | Product | Category | Value | Ships | Share | Rk |
|---|---|---|---|---|---|---|
| 1 | Immunoglobulin | Other | $2.5M | 49 | 1.4% | 2 |
| 2 | Pioglitazone | Diabetes & Endocrine | $601.5K | 59 | 1.6% | 13 |
| 3 | Natural | Ayurvedic & Herbal Products | $600.0K | 12 | 2.3% | 2 |
| 4 | Colchicine | Lipid & Metabolism | $480.7K | 43 | 2.3% | 4 |
| 5 | Folic | Nutritional Supplements | $398.4K | 29 | 0.7% | 18 |
| 6 | Glimepiride | Diabetes & Endocrine | $337.5K | 28 | 0.4% | 17 |
| 7 | Cyclophosphamide | Oncology | $269.0K | 55 | 1.4% | 7 |
| 8 | Ifosfamide | Advanced Oncology | $255.5K | 21 | 10.5% | 2 |
| 9 | Protein | Nutritional Supplements | $161.2K | 4 | 1.1% | 11 |
Ambica International Corporation imports 9 pharmaceutical products across 7 categories into Philippines totaling $5.6M.
Key Metrics
Top Categories
Indian Suppliers
Related Trade Data
Need Detailed Data?
Shipment-level records, supplier connections & pricing for Ambica International Corporation.
Request DemoAmbica International Corporation — Corporate Profile & Information
Company type, headquarters, distribution network, and industry role
1Company Overview
Ambica International Corporation is a leading pharmaceutical company based in Parañaque City, Metro Manila, Philippines. Established in 2003, the company specializes in the importation, distribution, marketing, and sale of a diverse range of medical products, including branded generics, patented drugs, and nutraceuticals. With a robust portfolio covering at least 26 therapeutic segments, Ambica caters to a wide spectrum of healthcare needs, ensuring optimal efficacy and safety.
The company's mission is to provide high-quality healthcare products and services that are accessible and affordable to everyone, advocating for the health and well-being of the communities it serves. Ambica's strategies are anchored in four core values: Accessibility, Availability, Affordability, and Advocacy. These principles guide their operations, aiming to eliminate barriers to healthcare, ensure a robust supply chain, bridge economic divides, and promote health literacy and public health initiatives.
2Distribution Network
Ambica International Corporation operates a comprehensive distribution network across the Philippines, ensuring that its products are readily available in leading drug stores and hospitals nationwide. The company's headquarters are located at No. 9 Amsterdam Extension, Merville Park Subdivision, Parañaque City, Metro Manila. Additionally, Ambica maintains a branch office at 3270-A Armstrong Ave, Merville Access Road, Pasay City, Philippines.
While specific details about warehouse locations and logistics capabilities are not publicly disclosed, Ambica's extensive distribution network indicates a well-established infrastructure capable of meeting the demands of the Philippine market. The company's focus on accessibility and availability suggests a strategic approach to logistics, ensuring that products are efficiently distributed to various regions within the country.
3Industry Role
Ambica International Corporation plays a pivotal role in the Philippine pharmaceutical supply chain as a primary wholesaler and distributor. By importing, marketing, and distributing a wide array of pharmaceutical products, the company serves as a crucial intermediary between international manufacturers and local healthcare providers. This position enables Ambica to ensure the availability of essential medications across the nation, contributing significantly to the healthcare sector's efficiency and reach.
Supplier Relationship Intelligence — Ambica International Corporation
Sourcing concentration, supply chain resilience, and strategic implications
1Sourcing Concentration Analysis
Ambica International Corporation's sourcing strategy exhibits a high degree of concentration, with the top five products accounting for 81.6% of the total import value. This concentration indicates a strategic focus on specific high-demand products, potentially allowing for better negotiation terms and streamlined supply chain management. However, such dependency on a limited product range could pose risks if market dynamics shift or if there are disruptions in the supply of these key products.
The company's reliance on a diverse set of suppliers, with 49 unique Indian suppliers, suggests a strategic approach to mitigate risks associated with single-source dependency. This diversified supplier base enhances the stability and resilience of Ambica's supply chain, reducing the impact of potential disruptions from any single supplier.
2Supply Chain Resilience
Ambica International Corporation's supply chain resilience is bolstered by its diversified supplier base, encompassing 49 unique Indian suppliers. This diversity mitigates risks associated with single-source dependency, ensuring a more stable and adaptable supply chain. The company's focus on importing finished pharmaceutical formulations, rather than raw APIs or bulk drugs, indicates a strategic choice to streamline operations and maintain product quality.
While specific details about backup suppliers and shipping route exposures are not publicly disclosed, Ambica's extensive network of suppliers and its commitment to regulatory compliance suggest a proactive approach to maintaining supply chain integrity. The company's partnerships with world-renowned manufacturers with state-of-the-art facilities approved by highly regulated drug authorities, such as the USFDA, UKMHRA, TGA, EUGMP, and PICs member authorities, further enhance the resilience and reliability of its supply chain.
3Strategic Implications
Ambica International Corporation's sourcing pattern, characterized by a high concentration on specific products and a diversified supplier base, positions the company to leverage economies of scale and negotiate favorable terms with suppliers. This strategy enhances its competitive position in the Philippine pharmaceutical market by ensuring a steady supply of high-demand products.
For Indian exporters, the opportunity lies in aligning their product offerings with Ambica's focus areas, particularly in the therapeutic categories where Ambica has a significant presence. By ensuring compliance with the regulatory standards of both India and the Philippines, and by offering products that meet the quality expectations of Ambica, Indian exporters can establish mutually beneficial partnerships.
Importing Pharmaceuticals into Philippines — Regulatory Framework
Regulatory authority, GMP requirements, import licensing for Philippines
1Regulatory Authority & Framework
In the Philippines, the Food and Drug Administration (FDA) serves as the primary regulatory authority overseeing the importation, distribution, and marketing of pharmaceutical products. The FDA's responsibilities include product registration, licensing of establishments, post-market surveillance, and enforcement against unregistered health products. Key legislation governing pharmaceutical imports includes the Food, Drug, and Cosmetic Act (Republic Act No. 3720) and the FDA Act of 2009 (Republic Act No. 9711).
The marketing authorization pathway for Indian generics involves obtaining a Certificate of Product Registration (CPR) from the Philippine FDA. This process requires compliance with the FDA's standards, including submission of necessary documentation, quality control test results, and adherence to Good Manufacturing Practice (GMP) guidelines. Additionally, the FDA has implemented measures to simplify the export process for pharmaceutical products, enhancing the efficiency of product registration and distribution. (pna.gov.ph)
2Import Licensing & GMP
Import licensing requirements in the Philippines mandate that pharmaceutical importers, such as Ambica International Corporation, obtain a License to Operate (LTO) from the FDA. This license authorizes the company to import, export, and wholesale pharmaceutical products. Ambica holds an active LTO, indicating its compliance with the FDA's regulatory standards. (verification.fda.gov.ph)
The recognition of Good Manufacturing Practice (GMP) certificates is crucial for ensuring product quality and safety. The Philippine FDA recognizes GMP certifications from reputable authorities, including the European Union (EU) GMP, World Health Organization (WHO) GMP, and the Pharmaceutical Inspection Co-operation Scheme (PIC/S). Manufacturers supplying Ambica's imported products are expected to adhere to these recognized GMP standards to meet the regulatory requirements of the Philippines.
3Quality & Labeling
Pharmaceutical products imported into the Philippines must undergo batch testing to ensure compliance with quality standards. Stability requirements are enforced to guarantee that products maintain their efficacy and safety throughout their shelf life. Labeling language requirements stipulate that product labels be in English and Filipino, providing clear and accurate information to consumers. Serialization mandates are implemented to enhance traceability and prevent counterfeit products from entering the market.
4Recent Regulatory Changes
Between 2024 and 2026, the Philippine FDA has implemented measures to simplify the export process for pharmaceutical products. Administrative Order No. 2024-0012, titled "Prescribing the Rules and Regulations on the Registration of Pharmaceutical Products and Active Pharmaceutical Ingredients Intended Solely for Export," was issued to enhance the efficiency of product registration and distribution. These changes aim to strengthen the health sector and expedite the distribution of medicines to the public. (pna.gov.ph)
Ambica International Corporation — Procurement Pattern Analysis
Product strategy, sourcing profile, and market positioning
1Product Strategy
Ambica International Corporation's product category focus is strategically aligned with the therapeutic areas of high demand in the Philippine market. The company's top five imported products—Immunoglobulin, Pioglitazone, Natural, Colchicine, and Folic—reflect a targeted approach to address prevalent health conditions. The significant import value of Immunoglobulin, for instance, indicates a strategic emphasis on critical care and immunology, catering to the substantial demand for such treatments.
The market demand driving these imports is influenced by factors such as the prevalence of specific diseases, the availability of effective treatments, and the economic accessibility of these medications. Ambica's focus on these therapeutic areas demonstrates a keen understanding of the healthcare needs of the Filipino population and a commitment to providing essential medications that improve health outcomes.
2Sourcing Profile
Ambica International Corporation's sourcing strategy is centered on importing finished pharmaceutical formulations from India, rather than raw Active Pharmaceutical Ingredients (APIs) or bulk drugs. This approach allows the company to streamline operations, ensure consistent product quality, and meet the regulatory requirements of both India and the Philippines. India's robust pharmaceutical manufacturing sector, with its adherence to recognized GMP standards, aligns well with Ambica's sourcing needs.
The company's diversified supplier base, comprising 49 unique Indian suppliers, indicates a strategic effort to mitigate risks associated with single-source dependency. This diversification enhances the stability and resilience of Ambica's supply chain, ensuring a steady and reliable supply of pharmaceutical products to meet market demands.
3Market Positioning
Based on its product mix, Ambica International Corporation serves a broad segment of the Philippine pharmaceutical market, including retail pharmacies,
Frequently Asked Questions — Ambica International Corporation
What products does Ambica International Corporation import from India?
Ambica International Corporation imports 9 pharmaceutical products across 7 categories. Top imports: Immunoglobulin ($2.5M), Pioglitazone ($601.5K), Natural ($600.0K), Colchicine ($480.7K), Folic ($398.4K).
Who supplies pharmaceuticals to Ambica International Corporation from India?
Ambica International Corporation sources from 49 verified Indian suppliers. The primary supplier is Xl Laboratories Private Limited (14.3% of imports, $5.6M).
What is Ambica International Corporation's total pharmaceutical import value?
Ambica International Corporation's total pharmaceutical import value from India is $5.6M, based on 300 verified shipments in Indian Customs (DGFT) data.
What therapeutic categories does Ambica International Corporation focus on?
Ambica International Corporation imports across 7 categories. The largest: Other (44.1%), Diabetes & Endocrine (16.9%), Ayurvedic & Herbal Products (10.8%).
Get Full Ambica International Corporation Import Intelligence
Access shipment-level details, supplier connections, pricing data, and competitive analysis. TransData Nexus provides verified Indian Customs (DGFT) data trusted by pharmaceutical trade professionals worldwide.
Official References & Regulatory Resources
Verify import regulations and drug registration requirements with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Buyer Matching: Ambica International Corporation identified across shipments using consignee name normalization.
- 2.Statistical Normalization: Shipment values are statistically normalized to ensure accurate market share representation.
- 3.Market Share: Calculated per product as Ambica International Corporation's capped value divided by total Indian exports for that product.
- 4.Shipment Count: Based on 300 individual customs records matching Ambica International Corporation.
- 5.Supplier Verification: Ambica International Corporation sources from 49 verified Indian suppliers across 2,041 formulations, confirmed from customs records.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
9 Products Tracked
7 therapeutic categories
Expert-Reviewed
By pharmaceutical trade specialists
Data Source & Methodology
Trade data sourced from Indian Customs (DGFT) export shipment records. Values represent FOB export value in USD. For current shipment-level data, contact TransData Nexus.