Alloga France
Pharmaceutical Importer · Netherlands · Oncology Focus · $4.3M Total Trade · DGFT Verified
Alloga France is a pharmaceutical importer based in Netherlands with a total trade value of $4.3M across 8 products in 3 therapeutic categories. Based on 158 verified import shipments from Indian Customs (DGFT) records, Alloga France is the #1 buyer in 1 product including Cytarabine. Alloga France sources from 6 verified Indian suppliers, with Intas Pharmaceuticals Limited accounting for 80.7% of imports.
Alloga France — Import Portfolio & Supplier Network
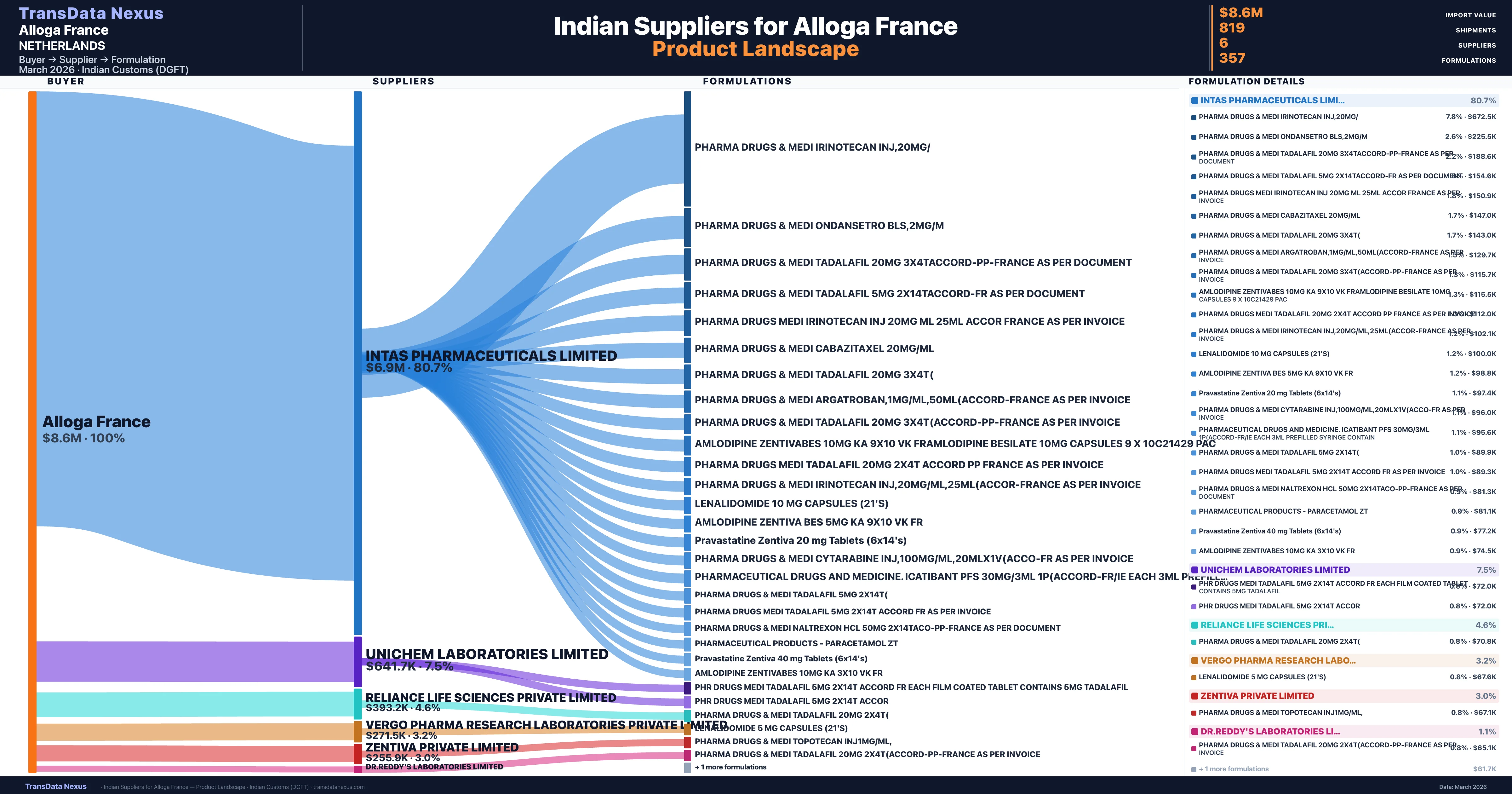
Who Are the Verified Indian Suppliers to Alloga France?
Customs-verified supplier relationships from Indian DGFT records
| Supplier | Value | Shipments | Share |
|---|---|---|---|
| Intas Pharmaceuticals Limited | $6.9M | 732 | 80.7% |
| Unichem Laboratories Limited | $641.7K | 36 | 7.5% |
| Reliance Life Sciences Private Limited | $393.2K | 11 | 4.6% |
| Vergo Pharma Research Laboratories Private Limited | $271.5K | 15 | 3.2% |
| Zentiva Private Limited | $255.9K | 22 | 3.0% |
| Dr.reddy's Laboratories Limited | $95.4K | 3 | 1.1% |
Alloga France sources from 6 verified Indian suppliers across 357 distinct formulations. The sourcing is highly concentrated — Intas Pharmaceuticals Limited accounts for 80.7% of total imports, indicating a strategic single-source relationship.
What Formulations Does Alloga France Import?
| Formulation | Value | Ships |
|---|---|---|
| Pharma drugs & medi irinotecan INJ | $672.5K | 22 |
| Pharma drugs & medi ondansetro bls | $225.5K | 21 |
| Pharma drugs & medi tadalafil 20MG 3x4taccord-pp-france as per | $188.6K | 5 |
| Pharma drugs & medi tadalafil 5MG 2x14taccord-fr as per | $154.6K | 5 |
| Pharma drugs medi irinotecan INJ 20MG ML 25ML accor france as per | $150.9K | 4 |
| Pharma drugs & medi cabazitaxel 20MG/ML | $147.0K | 3 |
| Pharma drugs & medi tadalafil 20MG | $143.0K | 6 |
| Pharma drugs & medi argatroban,1MG/ML,50ML(accord-france as per | $129.7K | 6 |
| Pharma drugs & medi tadalafil 20MG 3x4t(accord-pp-france as per | $115.7K | 8 |
| Amlodipine zentivabes 10MG ka 9x10 vk framlodipine besilate 10MG capsules 9 x 10c21429 | $115.5K | 3 |
| Pharma drugs medi tadalafil 20MG 2x4t accord pp france as per | $112.0K | 6 |
| Pharma drugs & medi irinotecan INJ,20MG/ML,25ML(accor-france as per | $102.1K | 4 |
| Lenalidomide 10 MG capsules (21's) | $100.0K | 2 |
| Amlodipine zentiva bes 5MG ka 9x10 vk | $98.8K | 6 |
| Pravastatine zentiva 20 MG tablets (6x14's) | $97.4K | 4 |
Alloga France imports 357 distinct pharmaceutical formulations. Showing top 15 by value. For full formulation-level data, contact TransData Nexus.
What Products Does Alloga France Import?
Alloga France Therapeutic Categories — 3 Specializations
Alloga France imports across 3 therapeutic categories, with Oncology (61.7%), Advanced Oncology (32.0%), Cardiovascular (6.3%) representing the largest segments. The portfolio is concentrated — top 5 products = 89% of total imports.
Oncology
2 products · 61.7% · $2.7M
Advanced Oncology
5 products · 32.0% · $1.4M
Cardiovascular
1 products · 6.3% · $271.5K
Import Portfolio — Top 8 by Import Value
| # | Product | Category | Value | Ships | Share | Rk |
|---|---|---|---|---|---|---|
| 1 | Irinotecan | Oncology | $2.4M | 50 | 5.9% | 3 |
| 2 | Cytarabine | Advanced Oncology | $530.3K | 36 | 4.9% | 1 |
| 3 | Cabazitaxel | Advanced Oncology | $386.4K | 8 | 4.2% | 4 |
| 4 | Pravastatin | Cardiovascular | $271.5K | 15 | 0.9% | 18 |
| 5 | Etoposide | Oncology | $235.4K | 23 | 2.2% | 9 |
| 6 | Topotecan | Advanced Oncology | $220.8K | 15 | 5.9% | 5 |
| 7 | Fludarabine | Advanced Oncology | $157.2K | 6 | 5.4% | 5 |
| 8 | Mitoxantrone | Advanced Oncology | $94.2K | 5 | 5.0% | 3 |
Alloga France imports 8 pharmaceutical products across 3 categories into Netherlands totaling $4.3M. The company is the #1 buyer for 1 product: Cytarabine.
Key Metrics
Top Categories
Indian Suppliers
Related Trade Data
Need Detailed Data?
Shipment-level records, supplier connections & pricing for Alloga France.
Request DemoAlloga France — Corporate Profile & Information
Company type, headquarters, distribution network, and industry role
1Company Overview
Alloga France is a prominent pharmaceutical logistics company specializing in the distribution and supply chain management of pharmaceutical products. Established in 1958, the company is headquartered in Marseille, France, and operates as a subsidiary within the Alloga network, which offers pan-European pharmaceutical logistics services. Alloga France's primary role involves managing logistics operations for pharmaceutical companies, preparing orders received from pharmacists, hospitals, and wholesalers, and providing services such as serialization, supply chain management, order processing, secondary packaging, and kitting. (craft.co)
In the Netherlands, Alloga France operates through its subsidiary, Alloga Netherlands B.V., located in Veghel. This subsidiary plays a significant role in the Dutch pharmaceutical distribution network, offering a range of services tailored to the specific requirements of the Dutch market. Alloga Netherlands B.V. is part of the broader Alloga network, which provides comprehensive pharmaceutical logistics solutions across Europe.
2Distribution Network
Alloga France's distribution network is extensive, encompassing multiple warehouses and logistics facilities across Europe. In the Netherlands, Alloga operates through its subsidiary, Alloga Netherlands B.V., located in Veghel. This facility is strategically positioned to serve the Dutch market, ensuring efficient distribution and supply chain management. The company's logistics capabilities include advanced warehousing, order processing, and transportation services, enabling it to meet the diverse needs of its clients in the pharmaceutical sector. Alloga's pan-European network allows for seamless distribution across various countries, providing comprehensive coverage and flexibility in logistics operations.
3Industry Role
Alloga France serves as a key logistics intermediary in the pharmaceutical supply chain, facilitating the distribution of pharmaceutical products from manufacturers to end-users, including pharmacies, hospitals, and wholesalers. The company's services encompass a wide range of logistics operations, such as warehousing, order processing, secondary packaging, and kitting, which are essential for ensuring the timely and efficient delivery of pharmaceutical products. By managing these critical aspects of the supply chain, Alloga France plays a pivotal role in maintaining the integrity and reliability of pharmaceutical distribution within the Netherlands and across Europe.
Supplier Relationship Intelligence — Alloga France
Sourcing concentration, supply chain resilience, and strategic implications
1Sourcing Concentration Analysis
Alloga France's sourcing strategy exhibits a high degree of concentration, with a significant portion of its pharmaceutical imports originating from a limited number of suppliers. Notably, Intas Pharmaceuticals Limited accounts for 80.7% of the total import value, followed by Unichem Laboratories Limited at 7.5%, and Reliance Life Sciences Private Limited at 4.6%. This concentrated sourcing approach suggests a strategic choice to establish strong, long-term relationships with key suppliers, potentially leading to favorable terms and consistent product quality.
However, such dependency on a few suppliers also introduces risks related to supply chain disruptions, price fluctuations, and potential challenges in negotiating terms. The shipment data indicates a stable relationship with these suppliers, as evidenced by the consistent number of shipments over the years. Nonetheless, Alloga France may consider diversifying its supplier base to mitigate potential risks associated with over-reliance on a limited number of sources.
2Supply Chain Resilience
Alloga France's supply chain resilience is influenced by its sourcing strategy and the diversity of its product formulations. The company imports a total of 357 unique pharmaceutical formulations, indicating a broad product portfolio. This diversity can enhance resilience by reducing dependency on specific products and suppliers. However, the high concentration of imports from a few suppliers, particularly from India, may expose the supply chain to risks such as geopolitical tensions, regulatory changes, and logistical disruptions.
To bolster supply chain resilience, Alloga France could explore establishing relationships with additional suppliers, both within and outside India, to diversify its sourcing and mitigate potential risks. Additionally, implementing robust contingency planning and maintaining strategic stock levels can further enhance the company's ability to respond effectively to unforeseen disruptions.
3Strategic Implications
Alloga France's concentrated sourcing pattern positions the company to leverage strong relationships with key suppliers, potentially securing favorable terms and ensuring consistent product quality. This strategy can enhance the company's competitive position by offering reliable and high-quality pharmaceutical products to the Dutch market. For Indian exporters, the existing partnerships with Alloga France present an opportunity to strengthen and expand their presence in the European market through a well-established distribution network.
However, the high supplier concentration also underscores the importance of maintaining strong, collaborative relationships and ensuring compliance with regulatory standards to sustain these partnerships. Indian exporters seeking to become alternative suppliers to Alloga France should focus on demonstrating product quality, reliability, and adherence to European regulatory requirements to meet the company's standards and expectations.
Importing Pharmaceuticals into Netherlands — Regulatory Framework
Regulatory authority, GMP requirements, import licensing for Netherlands
1Regulatory Authority & Framework
In the Netherlands, the primary regulatory authority overseeing pharmaceutical imports is the Dutch Medicines Evaluation Board (CBG-MEB), which operates under the Ministry of Health, Welfare, and Sport (VWS). The CBG-MEB is responsible for evaluating and monitoring the safety, efficacy, and quality of medicines available in the Dutch market. Key legislation governing pharmaceutical imports includes the Medicines Act (Geneesmiddelenwet), which outlines the requirements for marketing authorization, importation, and distribution of medicinal products. Additionally, the European Union's regulations, such as Regulation (EC) No 726/2004, provide a unified framework for the authorization and supervision of medicinal products across member states.
The marketing authorization pathway for Indian generics involves obtaining approval from the CBG-MEB, which includes submitting a comprehensive dossier demonstrating the product's quality, safety, and efficacy. This process ensures that imported generics meet the stringent standards required for distribution in the Netherlands. Collaboration with the CBG-MEB and adherence to EU regulations are essential for Indian exporters aiming to supply pharmaceutical products to the Dutch market.
2Import Licensing & GMP
Import licensing requirements in the Netherlands stipulate that companies involved in the importation of medicinal products must obtain the necessary permits from the CBG-MEB. This includes a manufacturer's permit for preparing or importing medicines from outside the European Economic Area (EEA) and a wholesale permit for distributing medicines within the Netherlands. Compliance with the European guidelines on Good Distribution Practice (GDP) is mandatory to ensure the quality and integrity of medicinal products throughout the supply chain. (business.gov.nl)
Good Manufacturing Practice (GMP) certification is recognized in the Netherlands, and imported pharmaceutical products must be manufactured in accordance with EU GMP standards. This certification ensures that products are consistently produced and controlled to quality standards. Indian exporters seeking to supply Alloga France and the broader Dutch market must ensure that their manufacturing facilities are GMP-compliant to meet these regulatory requirements.
3Quality & Labeling
Imported pharmaceutical products in the Netherlands are subject to batch testing and stability requirements to ensure their safety and efficacy. These tests are conducted by authorized laboratories to verify that products meet the required standards before they are released into the market. Labeling requirements include providing information in Dutch, including the product name, active ingredients, dosage form, and instructions for use. Serialization mandates are in place to prevent counterfeit medicines, requiring unique identifiers on packaging to track and trace products throughout the supply chain.
4Recent Regulatory Changes
Effective from 1 July 2024, significant changes have been implemented regarding the declaration of pharmaceutical products and active substances for import into the Netherlands. Importers are now required to include specific document codes in their declarations, such as code 1400 for marketing authorization holders and code 1450 for active substance registrations. Failure to include the appropriate codes will result in the rejection of the import declaration.
Alloga France — Procurement Pattern Analysis
Product strategy, sourcing profile, and market positioning
1Product Strategy
Alloga France's product category focus on oncology, advanced oncology, and cardiovascular therapeutics aligns with the growing demand for specialized treatments in these areas. The substantial import values of products like Cytarabine, Irinotecan, and Cabazitaxel indicate a strategic emphasis on high-demand oncology medications. This focus is driven by the increasing prevalence of cancer and cardiovascular diseases, necessitating a reliable supply of effective treatments. By concentrating on these therapeutic areas, Alloga France positions itself to meet critical healthcare needs and strengthen its market presence.
2Sourcing Profile
Alloga France's sourcing strategy for generic drugs involves importing a diverse range of formulations from India, with a significant concentration on oncology and cardiovascular products. The company's preference for sourcing from India is likely due to the country's robust pharmaceutical manufacturing capabilities, cost-effectiveness, and adherence to international quality standards. India's compliance with EU GMP guidelines further enhances its suitability as a sourcing partner for Alloga France.
3Market Positioning
Based on its product mix, Alloga France primarily serves the hospital and wholesale distribution segments of the Netherlands market. The focus on specialized oncology and cardiovascular products indicates a strategic alignment with the needs of healthcare institutions requiring these treatments. By supplying high-demand, specialized medications, All
Frequently Asked Questions — Alloga France
What products does Alloga France import from India?
Alloga France imports 8 pharmaceutical products across 3 categories. Top imports: Irinotecan ($2.4M), Cytarabine ($530.3K), Cabazitaxel ($386.4K), Pravastatin ($271.5K), Etoposide ($235.4K).
Who supplies pharmaceuticals to Alloga France from India?
Alloga France sources from 6 verified Indian suppliers. The primary supplier is Intas Pharmaceuticals Limited (80.7% of imports, $6.9M).
What is Alloga France's total pharmaceutical import value?
Alloga France's total pharmaceutical import value from India is $4.3M, based on 158 verified shipments in Indian Customs (DGFT) data.
What therapeutic categories does Alloga France focus on?
Alloga France imports across 3 categories. The largest: Oncology (61.7%), Advanced Oncology (32.0%), Cardiovascular (6.3%).
Get Full Alloga France Import Intelligence
Access shipment-level details, supplier connections, pricing data, and competitive analysis. TransData Nexus provides verified Indian Customs (DGFT) data trusted by pharmaceutical trade professionals worldwide.
Official References & Regulatory Resources
Verify import regulations and drug registration requirements with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Buyer Matching: Alloga France identified across shipments using consignee name normalization.
- 2.Statistical Normalization: Shipment values are statistically normalized to ensure accurate market share representation.
- 3.Market Share: Calculated per product as Alloga France's capped value divided by total Indian exports for that product.
- 4.Shipment Count: Based on 158 individual customs records matching Alloga France.
- 5.Supplier Verification: Alloga France sources from 6 verified Indian suppliers across 357 formulations, confirmed from customs records.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
8 Products Tracked
3 therapeutic categories
Expert-Reviewed
By pharmaceutical trade specialists
Data Source & Methodology
Trade data sourced from Indian Customs (DGFT) export shipment records. Values represent FOB export value in USD. For current shipment-level data, contact TransData Nexus.