Africure Pharmaceuticals Botswana
Pharmaceutical Importer · Botswana · Antiviral & HIV Medications Focus · $2.0M Total Trade · DGFT Verified
Africure Pharmaceuticals Botswana is a pharmaceutical importer based in Botswana with a total trade value of $2.0M across 2 products in 1 therapeutic categories. Based on 40 verified import shipments from Indian Customs (DGFT) records, the company actively imports across multiple product segments. Africure Pharmaceuticals Botswana sources from 2 verified Indian suppliers, with Macleods Pharmaceuticals Limited accounting for 97.1% of imports.
Africure Pharmaceuticals Botswana — Import Portfolio & Supplier Network
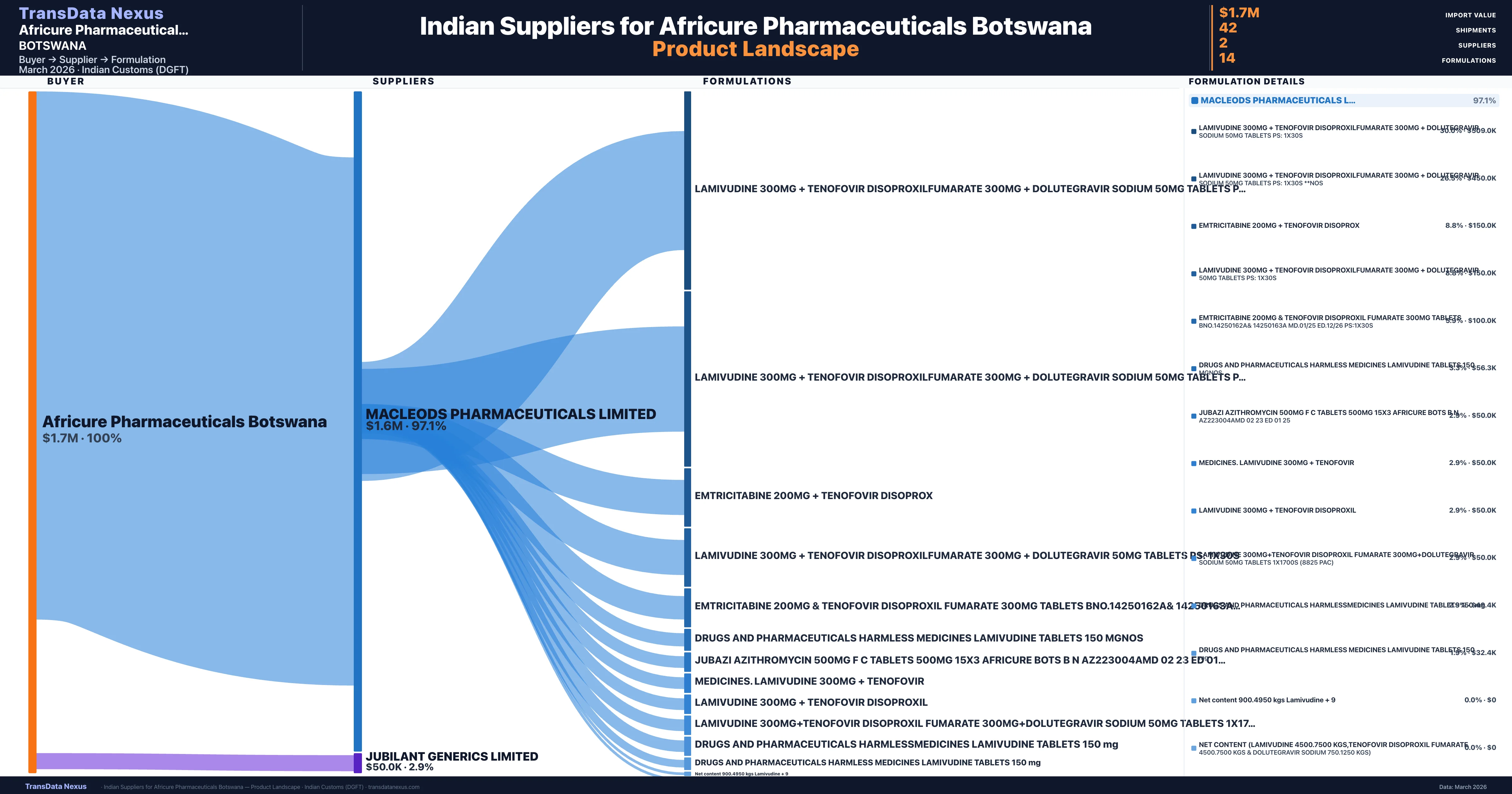
Who Are the Verified Indian Suppliers to Africure Pharmaceuticals Botswana?
Customs-verified supplier relationships from Indian DGFT records
| Supplier | Value | Shipments | Share |
|---|---|---|---|
| Macleods Pharmaceuticals Limited | $1.6M | 41 | 97.1% |
| Jubilant Generics Limited | $50.0K | 1 | 2.9% |
Africure Pharmaceuticals Botswana sources from 2 verified Indian suppliers across 14 distinct formulations. The sourcing is highly concentrated — Macleods Pharmaceuticals Limited accounts for 97.1% of total imports, indicating a strategic single-source relationship.
What Formulations Does Africure Pharmaceuticals Botswana Import?
| Formulation | Value | Ships |
|---|---|---|
| Lamivudine 300MG + tenofovir disoproxilfumarate 300MG + dolutegravir sodium 50MG tablets ps: 1x30s | $509.0K | 11 |
| Lamivudine 300MG + tenofovir disoproxilfumarate 300MG + dolutegravir sodium 50MG tablets ps: 1x30s | $450.0K | 9 |
| Emtricitabine 200MG + tenofovir | $150.0K | 3 |
| Lamivudine 300MG + tenofovir disoproxilfumarate 300MG + dolutegravir 50MG tablets ps: 1x30s | $150.0K | 3 |
| Emtricitabine 200MG & tenofovir disoproxil fumarate 300MG tablets bno.14250162a& 14250163a md.01/25 ed.12/26 ps:1x30s | $100.0K | 2 |
| Drugs and pharmaceuticals harmless medicines lamivudine tablets 150 | $56.3K | 6 |
| Jubazi azithromycin 500MG f c tablets 500MG 15x3 africure bots b n az223004amd 02 23 ed 01 | $50.0K | 1 |
| Medicines. lamivudine 300MG + | $50.0K | 1 |
| Lamivudine 300MG + tenofovir | $50.0K | 1 |
| Lamivudine 300MG+tenofovir disoproxil fumarate 300MG+dolutegravir sodium 50MG tablets 1x1700s (8825 pac) | $50.0K | 1 |
| Drugs and pharmaceuticals harmlessmedicines lamivudine tablets 150 MG | $49.4K | 1 |
| Drugs and pharmaceuticals harmless medicines lamivudine tablets 150 MG | $32.4K | 1 |
| Net content 900.4950 kgs lamivudine + | $0 | 1 |
| Net content (lamivudine 4500.7500 kgs,tenofovir disoproxil fumarate 4500.7500 kgs & dolutegravir sodium 750.1250 kgs) | $0 | 1 |
Africure Pharmaceuticals Botswana imports 14 distinct pharmaceutical formulations. Showing top 14 by value. For full formulation-level data, contact TransData Nexus.
What Products Does Africure Pharmaceuticals Botswana Import?
Top Products by Import Value
Africure Pharmaceuticals Botswana Therapeutic Categories — 1 Specializations
Africure Pharmaceuticals Botswana imports across 1 therapeutic categories, with Antiviral & HIV Medications (100.0%), representing the largest segments. The portfolio is concentrated — top 5 products = 100% of total imports.
Antiviral & HIV Medications
2 products · 100.0% · $2.0M
Import Portfolio — Top 2 by Import Value
| # | Product | Category | Value | Ships | Share | Rk |
|---|---|---|---|---|---|---|
| 1 | Tenofovir | Antiviral & HIV Medications | $1.1M | 23 | 0.0% | 6 |
| 2 | Dolutegravir | Antiviral & HIV Medications | $850.0K | 17 | 0.0% | 6 |
Africure Pharmaceuticals Botswana imports 2 pharmaceutical products across 1 categories into Botswana totaling $2.0M.
Key Metrics
Top Categories
Indian Suppliers
Related Trade Data
Need Detailed Data?
Shipment-level records, supplier connections & pricing for Africure Pharmaceuticals Botswana.
Request DemoAfricure Pharmaceuticals Botswana — Corporate Profile & Information
Company type, headquarters, distribution network, and industry role
1Company Overview
Africure Pharmaceuticals Botswana Pty Ltd is a pharmaceutical importer and distributor based in Botswana, specializing in the procurement and distribution of finished pharmaceutical formulations. The company operates under the ownership of Africure Pharmaceuticals Limited, a Mauritius-based entity committed to enhancing African healthcare through the establishment of manufacturing assets and technical capabilities across Sub-Saharan Africa. Africure Pharmaceuticals Limited's manufacturing facilities are located in countries such as Cameroon, Côte d'Ivoire, Namibia, Botswana, and India, with distribution subsidiaries in Burkina Faso, Cameroon, and Nigeria.
Africure Pharmaceuticals Botswana plays a pivotal role in the country's pharmaceutical distribution network by importing essential medications, particularly focusing on antiviral and HIV treatments. The company's operations are integral to ensuring the availability of critical medications within Botswana, thereby contributing to the nation's healthcare infrastructure. By sourcing high-quality pharmaceutical products from reputable manufacturers, Africure Pharmaceuticals Botswana addresses the local demand for effective treatments, aligning with Africure Pharmaceuticals Limited's mission to manufacture high-quality essential medication in Africa by Africans for Africa.
2Distribution Network
Africure Pharmaceuticals Botswana's distribution network is strategically designed to ensure efficient delivery of pharmaceutical products across Botswana. The company's warehouse is situated at Plot 1090, Selokwaneng Industrial Site, Tlokweng, Gaborone, Botswana. This central location facilitates streamlined logistics operations, enabling timely distribution to various healthcare facilities and pharmacies throughout the country. While specific details regarding the company's logistics capabilities and geographic coverage beyond Botswana are not publicly disclosed, Africure Pharmaceuticals Limited's broader operations in Sub-Saharan Africa suggest a potential for regional distribution, leveraging its manufacturing facilities in neighboring countries.
3Industry Role
Africure Pharmaceuticals Botswana serves as a key importer and distributor within Botswana's pharmaceutical supply chain. By focusing on the importation of finished pharmaceutical formulations, the company ensures the availability of essential medications, particularly in the antiviral and HIV treatment categories. This role is crucial in addressing the healthcare needs of the population, especially in managing and treating HIV/AIDS, a significant health concern in the region. Through its operations, Africure Pharmaceuticals Botswana contributes to the overall health and well-being of the community by providing access to necessary pharmaceutical products.
Supplier Relationship Intelligence — Africure Pharmaceuticals Botswana
Sourcing concentration, supply chain resilience, and strategic implications
1Sourcing Concentration Analysis
Africure Pharmaceuticals Botswana's sourcing strategy exhibits a high degree of concentration, with a total import value of $2.0 million USD from India across 40 shipments. The company's portfolio is entirely focused on two products—Tenofovir and Dolutegravir—both integral to HIV treatment regimens. This concentrated sourcing approach indicates a strategic decision to specialize in these critical medications, ensuring a consistent and reliable supply chain for these essential drugs.
The relationship with its primary supplier, MACLEODS PHARMACEUTICALS LIMITED, is characterized by a substantial volume of transactions, accounting for 97.1% of the total shipments. This high dependency on a single supplier could pose risks related to supply chain disruptions, such as production delays or quality control issues. However, the consistent and long-term nature of the relationship may also suggest a stable and mutually beneficial partnership. To mitigate potential risks associated with supplier concentration, Africure Pharmaceuticals Botswana may consider diversifying its supplier base or establishing contingency plans to ensure uninterrupted access to critical medications.
2Supply Chain Resilience
Africure Pharmaceuticals Botswana's supply chain resilience is closely tied to its sourcing strategy from India, particularly from MACLEODS PHARMACEUTICALS LIMITED. The company's focus on two specific formulations—Tenofovir and Dolutegravir—indicates a streamlined supply chain that may benefit from economies of scale and operational efficiencies. However, this specialization also means that any disruptions in the production or delivery of these products could significantly impact the company's ability to meet local demand.
The company's reliance on a single supplier for the majority of its imports suggests a need for robust risk management strategies. Establishing relationships with backup suppliers, diversifying the range of imported formulations, and exploring alternative shipping routes could enhance supply chain resilience. Additionally, ensuring that key suppliers adhere to international regulatory standards and maintain compliance with current Good Manufacturing Practices (cGMP) is essential to uphold product quality and safety.
3Strategic Implications
Africure Pharmaceuticals Botswana's concentrated sourcing pattern positions the company as a specialized importer and distributor of HIV-related medications within the Botswana market. This focus allows the company to develop deep expertise and strong relationships with its supplier, potentially leading to favorable terms and priority access to products. For Indian exporters, this presents an opportunity to establish or strengthen partnerships with Africure Pharmaceuticals Botswana by offering high-quality, competitively priced Tenofovir and Dolutegravir formulations.
However, the company's reliance on a single supplier for the majority of its imports also highlights the importance of reliability and consistency in product quality and delivery. Indian exporters seeking to become alternative suppliers should ensure they can meet Africure Pharmaceuticals Botswana's stringent requirements and provide a dependable supply chain to gain a foothold in this specialized market segment.
Importing Pharmaceuticals into Botswana — Regulatory Framework
Regulatory authority, GMP requirements, import licensing for Botswana
1Regulatory Authority & Framework
In Botswana, the regulation of medicines, medical devices, and cosmetics is overseen by the Botswana Medicines Regulatory Authority (BoMRA). BoMRA is responsible for ensuring that pharmaceutical products meet safety, efficacy, and quality standards before they are imported, distributed, or sold within the country. (bomra.co.bw)
The key legislation governing pharmaceutical imports in Botswana includes the Medicines and Related Substances Act, which outlines the requirements for the registration, importation, and distribution of pharmaceutical products. This act ensures that all medicines available in the market are evaluated for their safety and efficacy. The marketing authorization pathway for Indian generics involves submitting comprehensive documentation to BoMRA, including evidence of product quality, safety, and efficacy, as well as compliance with international standards.
2Import Licensing & GMP
Import licensing requirements in Botswana stipulate that all pharmaceutical products must be registered with BoMRA before they can be imported and distributed. This process involves providing detailed information about the product, including its composition, manufacturing process, and clinical data. Additionally, the importing company must hold a valid wholesale distribution authorization issued by BoMRA, ensuring that they operate in compliance with regulatory standards.
Good Manufacturing Practice (GMP) certification is a critical component of the import licensing process. BoMRA recognizes GMP certificates from reputable authorities, such as the World Health Organization (WHO) and the Pharmaceutical Inspection Co-operation Scheme (PIC/S). Indian exporters seeking to supply Africure Pharmaceuticals Botswana must ensure that their manufacturing facilities are GMP-compliant and that their products are accompanied by valid GMP certificates from recognized bodies.
3Quality & Labeling
Batch testing and stability studies are essential to ensure that pharmaceutical products maintain their quality, safety, and efficacy throughout their shelf life. Manufacturers must conduct these tests in accordance with international pharmacopoeial standards and provide BoMRA with the necessary documentation to demonstrate compliance.
Labeling requirements in Botswana mandate that all pharmaceutical products display information in English, including the product name, active ingredients, dosage form, strength, batch number, manufacturing date, expiry date, storage conditions, and the name and address of the manufacturer. Serialization mandates may also apply to facilitate traceability and prevent counterfeit products from entering the market.
4Recent Regulatory Changes
Between 2024 and 2026, Botswana has implemented several policy changes affecting pharmaceutical imports. These include stricter enforcement of GMP compliance, enhanced scrutiny of import licenses, and the introduction of electronic systems for product registration and tracking. These measures aim to improve the quality and safety of pharmaceutical products available in the country. Indian exporters must stay informed about these regulatory changes to ensure continued access to the Botswana market.
Africure Pharmaceuticals Botswana — Procurement Pattern Analysis
Product strategy, sourcing profile, and market positioning
1Product Strategy
Africure Pharmaceuticals Botswana's focus on antiviral and HIV medications, particularly Tenofovir and Dolutegravir, aligns with the high demand for effective HIV treatments in Botswana. The country has a significant HIV prevalence rate, necessitating a reliable supply of antiretroviral drugs to manage and control the epidemic. By specializing in these therapeutic areas, Africure Pharmaceuticals Botswana addresses a critical public health need, ensuring that patients have access to essential medications.
The market demand for these products is driven by the ongoing efforts to provide comprehensive HIV care, including prevention, treatment, and support services. The government's commitment to improving healthcare infrastructure and the availability of antiretroviral therapies further supports the importation and distribution of these medications. Africure Pharmaceuticals Botswana's product strategy is thus well-aligned with national health priorities and the needs of the population.
2Sourcing Profile
Africure Pharmaceuticals Botswana's sourcing strategy is centered on importing generic formulations of Tenofovir and Dolutegravir from India. This approach allows the company to provide cost-effective alternatives to branded medications, making HIV treatments more accessible to the Botswana population. India's robust pharmaceutical manufacturing sector, known for its adherence to international quality standards, makes it a reliable source for these generic drugs.
The company's focus on finished pharmaceutical formulations, rather than active pharmaceutical ingredients (APIs)
Frequently Asked Questions — Africure Pharmaceuticals Botswana
What products does Africure Pharmaceuticals Botswana import from India?
Africure Pharmaceuticals Botswana imports 2 pharmaceutical products across 1 categories. Top imports: Tenofovir ($1.1M), Dolutegravir ($850.0K).
Who supplies pharmaceuticals to Africure Pharmaceuticals Botswana from India?
Africure Pharmaceuticals Botswana sources from 2 verified Indian suppliers. The primary supplier is Macleods Pharmaceuticals Limited (97.1% of imports, $1.6M).
What is Africure Pharmaceuticals Botswana's total pharmaceutical import value?
Africure Pharmaceuticals Botswana's total pharmaceutical import value from India is $2.0M, based on 40 verified shipments in Indian Customs (DGFT) data.
What therapeutic categories does Africure Pharmaceuticals Botswana focus on?
Africure Pharmaceuticals Botswana imports across 1 categories. The largest: Antiviral & HIV Medications (100.0%).
Get Full Africure Pharmaceuticals Botswana Import Intelligence
Access shipment-level details, supplier connections, pricing data, and competitive analysis. TransData Nexus provides verified Indian Customs (DGFT) data trusted by pharmaceutical trade professionals worldwide.
Official References & Regulatory Resources
Verify import regulations and drug registration requirements with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Buyer Matching: Africure Pharmaceuticals Botswana identified across shipments using consignee name normalization.
- 2.Statistical Normalization: Shipment values are statistically normalized to ensure accurate market share representation.
- 3.Market Share: Calculated per product as Africure Pharmaceuticals Botswana's capped value divided by total Indian exports for that product.
- 4.Shipment Count: Based on 40 individual customs records matching Africure Pharmaceuticals Botswana.
- 5.Supplier Verification: Africure Pharmaceuticals Botswana sources from 2 verified Indian suppliers across 14 formulations, confirmed from customs records.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
2 Products Tracked
1 therapeutic categories
Expert-Reviewed
By pharmaceutical trade specialists
Data Source & Methodology
Trade data sourced from Indian Customs (DGFT) export shipment records. Values represent FOB export value in USD. For current shipment-level data, contact TransData Nexus.