Advy Japan Co,ltd
Pharmaceutical Importer · Japan · Medical Devices & Diagnostics Focus · $822.8K Total Trade · DGFT Verified
Advy Japan Co,ltd is a pharmaceutical importer based in Japan with a total trade value of $822.8K across 2 products in 1 therapeutic categories. Based on 246 verified import shipments from Indian Customs (DGFT) records, the company actively imports across multiple product segments. Advy Japan Co,ltd sources from 1 verified Indian supplier, with Advy Chemical Private Limited accounting for 100.0% of imports.
Advy Japan Co,ltd — Import Portfolio & Supplier Network
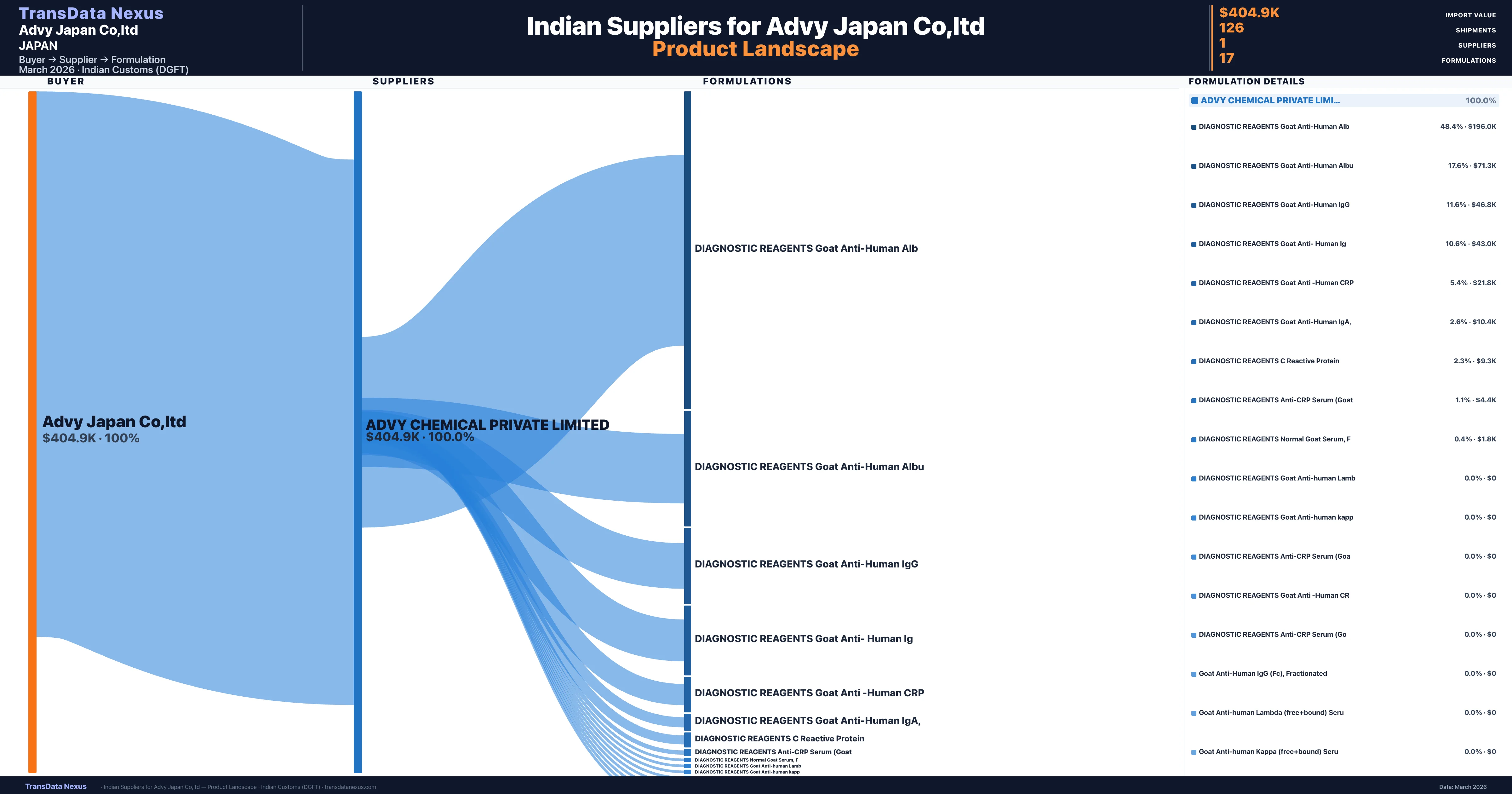
Who Are the Verified Indian Suppliers to Advy Japan Co,ltd?
Customs-verified supplier relationships from Indian DGFT records
| Supplier | Value | Shipments | Share |
|---|---|---|---|
| Advy Chemical Private Limited | $404.9K | 126 | 100.0% |
Advy Japan Co,ltd sources from 1 verified Indian supplier across 17 distinct formulations. The sourcing is highly concentrated — Advy Chemical Private Limited accounts for 100.0% of total imports, indicating a strategic single-source relationship.
What Formulations Does Advy Japan Co,ltd Import?
| Formulation | Value | Ships |
|---|---|---|
| Diagnostic reagents goat anti-human | $196.0K | 9 |
| Diagnostic reagents goat anti-human | $71.3K | 6 |
| Diagnostic reagents goat anti-human | $46.8K | 8 |
| Diagnostic reagents goat anti- human | $43.0K | 1 |
| Diagnostic reagents goat anti -human | $21.8K | 4 |
| Diagnostic reagents goat anti-human iga | $10.4K | 4 |
| Diagnostic reagents c reactive | $9.3K | 1 |
| Diagnostic reagents anti-crp serum | $4.4K | 24 |
| Diagnostic reagents normal goat serum | $1.8K | 2 |
| Diagnostic reagents goat anti-human | $0 | 1 |
| Diagnostic reagents goat anti-human | $0 | 1 |
| Diagnostic reagents anti-crp serum | $0 | 58 |
| Diagnostic reagents goat anti -human | $0 | 2 |
| Diagnostic reagents anti-crp serum | $0 | 2 |
| Goat anti-human igg (fc) | $0 | 1 |
Advy Japan Co,ltd imports 17 distinct pharmaceutical formulations. Showing top 15 by value. For full formulation-level data, contact TransData Nexus.
What Products Does Advy Japan Co,ltd Import?
Top Products by Import Value
Advy Japan Co,ltd Therapeutic Categories — 1 Specializations
Advy Japan Co,ltd imports across 1 therapeutic categories, with Medical Devices & Diagnostics (100.0%), representing the largest segments. The portfolio is concentrated — top 5 products = 100% of total imports.
Medical Devices & Diagnostics
2 products · 100.0% · $822.8K
Import Portfolio — Top 2 by Import Value
| # | Product | Category | Value | Ships | Share | Rk |
|---|---|---|---|---|---|---|
| 1 | Diagnostic | Medical Devices & Diagnostics | $411.4K | 123 | 2.2% | 7 |
| 2 | Reagent | Medical Devices & Diagnostics | $411.4K | 123 | 3.1% | 5 |
Advy Japan Co,ltd imports 2 pharmaceutical products across 1 categories into Japan totaling $822.8K.
Key Metrics
Top Categories
Indian Suppliers
Related Trade Data
Need Detailed Data?
Shipment-level records, supplier connections & pricing for Advy Japan Co,ltd.
Request DemoAdvy Japan Co,ltd — Corporate Profile & Information
Company type, headquarters, distribution network, and industry role
1Company Overview
Advy Japan Co., Ltd. is a pharmaceutical importer and buyer headquartered in Tokyo, Japan. Established on April 1, 2008, the company operates from the Kanda No.4 Park Building in Chiyoda-ku, Tokyo. The managing director is Mr. Toshiro Hijikata, with Mr. Bharat Daftary and Mr. Syd Daftary serving as board members. (advy-japan.jp)
As an affiliate of Bharat Serums and Vaccines Limited (BSVL), a leading Indian pharmaceutical company, Advy Japan Co., Ltd. specializes in the import and distribution of pharmaceutical raw materials, medical devices, diagnostic reagents, and related products. The company plays a pivotal role in introducing BSVL's products and services to the Japanese market, ensuring compliance with local regulations and meeting the specific needs of Japanese healthcare providers. (advy-japan.jp)
2Distribution Network
Advy Japan Co., Ltd. operates its distribution network from its Tokyo headquarters, strategically located to facilitate efficient logistics and supply chain management within Japan. While specific warehouse locations are not publicly disclosed, the company's central position in Tokyo enables effective distribution across the country. The company leverages its affiliation with BSVL to access a global network of suppliers and partners, enhancing its ability to source and deliver a diverse range of pharmaceutical products to the Japanese market. (advy-japan.jp)
3Industry Role
In Japan's pharmaceutical supply chain, Advy Japan Co., Ltd. functions primarily as an importer and distributor of pharmaceutical raw materials and diagnostic reagents. By sourcing products from its parent company, BSVL, and other global suppliers, Advy Japan ensures a steady supply of essential medical products to Japanese healthcare providers. The company's role is crucial in bridging international pharmaceutical innovations with the Japanese market, adhering to local regulatory standards and addressing the specific needs of Japanese patients and medical professionals. (advy-japan.jp)
Supplier Relationship Intelligence — Advy Japan Co,ltd
Sourcing concentration, supply chain resilience, and strategic implications
1Sourcing Concentration Analysis
Advy Japan Co., Ltd. demonstrates a high level of sourcing concentration, with a total import value of $823,000 USD from India across 246 shipments, primarily from a single supplier, Advy Chemical Private Limited. This single-source dependency indicates a strategic choice to maintain a consistent and reliable supply chain, leveraging the established relationship with Advy Chemical to ensure product quality and availability. The substantial volume of imports from this supplier suggests a stable and long-term partnership, reducing the risks associated with supplier diversification. However, this concentration also means that any disruptions at Advy Chemical could significantly impact Advy Japan's operations, highlighting the importance of robust contingency planning and risk management strategies.
2Supply Chain Resilience
The resilience of Advy Japan Co., Ltd.'s supply chain is closely tied to its reliance on Advy Chemical Private Limited for the majority of its imports. While this single-source strategy offers consistency, it also exposes the company to potential risks such as supply disruptions or quality issues from the supplier. The limited number of unique suppliers and formulations imported suggests a lack of diversification, which could be a vulnerability in the event of unforeseen challenges. To enhance supply chain resilience, Advy Japan may consider identifying and establishing relationships with additional suppliers, diversifying its product portfolio, and developing contingency plans to mitigate potential disruptions.
3Strategic Implications
Advy Japan Co., Ltd.'s sourcing pattern, characterized by a high concentration on a single supplier, positions the company to benefit from streamlined operations and potentially favorable terms due to the established relationship. This strategy may lead to cost efficiencies and a strong alignment with Advy Chemical's product offerings. However, the lack of supplier diversification could pose risks, especially if Advy Chemical faces operational challenges or quality control issues. For Indian exporters seeking to become alternative suppliers, this presents an opportunity to introduce their products to Advy Japan, potentially filling gaps in the current sourcing strategy and offering a more diversified supply chain to mitigate risks.
Importing Pharmaceuticals into Japan — Regulatory Framework
Regulatory authority, GMP requirements, import licensing for Japan
1Regulatory Authority & Framework
In Japan, the regulation of pharmaceutical products is overseen by the Ministry of Health, Labour and Welfare (MHLW), which enforces the Pharmaceutical and Medical Device Act (PMD Act). This legislation governs the import, manufacture, and sale of pharmaceuticals, ensuring that all products meet stringent safety and efficacy standards. The PMD Act outlines the requirements for marketing authorization, quality control, and post-market surveillance, providing a comprehensive framework for pharmaceutical operations within Japan. (selling-in-japan.jp)
2Import Licensing & GMP
To import pharmaceutical products into Japan, companies must obtain the necessary licenses under the PMD Act. This includes securing an import license from the MHLW, which authorizes the company to import and distribute pharmaceutical products within Japan. Additionally, imported products must comply with Good Manufacturing Practice (GMP) standards recognized by the MHLW. While Japan has its own GMP guidelines, it also accepts certifications from other reputable standards, such as EU GMP, WHO GMP, and PIC/S, provided they meet the MHLW's criteria. This recognition facilitates international trade and ensures that imported products adhere to Japan's rigorous quality standards. (selling-in-japan.jp)
3Quality & Labeling
Pharmaceutical products imported into Japan must undergo batch testing to verify their quality, safety, and efficacy. Stability studies are also required to ensure that products maintain their intended quality throughout their shelf life. Labeling requirements stipulate that all information be presented in Japanese, including product names, usage instructions, and safety warnings, to ensure clarity and compliance with local regulations. Serialization mandates are enforced to enhance traceability and prevent counterfeit products from entering the market, thereby safeguarding public health. (selling-in-japan.jp)
4Recent Regulatory Changes
Between 2024 and 2026, Japan implemented several regulatory changes affecting pharmaceutical imports. Notably, the amendment of the Four Product Safety Acts, effective December 25, 2025, introduced new requirements for foreign businesses selling regulated products in Japan through online platforms. These businesses are now required to appoint a Domestic Administrator and submit a formal notification to the Ministry of Economy, Trade and Industry (METI). This change aims to enhance consumer safety and ensure that imported pharmaceutical products meet Japan's stringent standards. (skadvisory.jp)
Advy Japan Co,ltd — Procurement Pattern Analysis
Product strategy, sourcing profile, and market positioning
1Product Strategy
Advy Japan Co., Ltd. focuses on importing diagnostic reagents and related pharmaceutical preparations, including antigens, antibodies, and control sera, primarily from Advy Chemical Private Limited. This strategic focus aligns with the growing demand for advanced diagnostic tools in Japan's healthcare sector, driven by an aging population and the need for accurate and timely disease detection. By specializing in these products, Advy Japan positions itself as a key supplier of essential diagnostic materials, contributing to improved patient outcomes and supporting the efficiency of medical diagnostics in Japan. (advy-japan.jp)
2Sourcing Profile
Advy Japan Co., Ltd. employs a sourcing strategy that heavily relies on its parent company, Advy Chemical Private Limited, for the supply of diagnostic reagents and pharmaceutical preparations. This approach ensures consistency in product quality and leverages the established relationship between the two entities. The focus on specific formulations, such as antigens and antibodies, reflects a targeted procurement strategy aimed at meeting the specialized needs of the Japanese diagnostic market. While this strategy offers advantages in terms of supply chain efficiency and product consistency, it also highlights the importance of maintaining a robust and resilient supply chain to mitigate potential risks associated with supplier dependency. (advy-japan.jp)
3Market Positioning
By specializing in the import and distribution of diagnostic reagents and related pharmaceutical products, Advy Japan Co., Ltd. serves a niche segment of the Japanese market. Its primary customers include hospitals, diagnostic laboratories, and healthcare providers that require high-quality diagnostic materials for disease detection and patient care. The company's focus on these specialized products positions it as a key player in the healthcare supply chain, contributing to the advancement of medical diagnostics and supporting the delivery of quality healthcare services in Japan. (advy-japan.jp)
Seller's Guide — How to Become a Supplier to Advy Japan Co,ltd
Opportunity assessment, qualifications, and practical approach strategy
1Opportunity Assessment
For new Indian suppliers, there is a realistic opportunity to collaborate with Advy Japan Co., Ltd. by offering complementary products or alternative formulations that align with the diagnostic reagents and pharmaceutical preparations currently imported. Diversifying Advy Japan's supplier base could enhance supply chain resilience and provide access to a broader range of products for the Japanese market. Identifying gaps in Advy Japan's current sourcing strategy, such as specific diagnostic reagents or pharmaceutical preparations not currently imported, presents an opportunity for new suppliers to meet these unmet needs. Establishing partnerships with Advy Japan could lead to mutual benefits, including expanded market reach and enhanced product offerings.
2Requirements & Qualifications
Indian exporters seeking to supply products to Advy Japan Co., Ltd. must ensure that their products comply with Japan's regulatory standards, including obtaining the necessary import licenses under the Pharmaceutical and Medical Device Act. Products must adhere to Good Manufacturing Practice (GMP) standards recognized by the Ministry of Health, Labour and Welfare, such as EU GMP, WHO GMP, or
Frequently Asked Questions — Advy Japan Co,ltd
What products does Advy Japan Co,ltd import from India?
Advy Japan Co,ltd imports 2 pharmaceutical products across 1 categories. Top imports: Diagnostic ($411.4K), Reagent ($411.4K).
Who supplies pharmaceuticals to Advy Japan Co,ltd from India?
Advy Japan Co,ltd sources from 1 verified Indian suppliers. The primary supplier is Advy Chemical Private Limited (100.0% of imports, $404.9K).
What is Advy Japan Co,ltd's total pharmaceutical import value?
Advy Japan Co,ltd's total pharmaceutical import value from India is $822.8K, based on 246 verified shipments in Indian Customs (DGFT) data.
What therapeutic categories does Advy Japan Co,ltd focus on?
Advy Japan Co,ltd imports across 1 categories. The largest: Medical Devices & Diagnostics (100.0%).
Get Full Advy Japan Co,ltd Import Intelligence
Access shipment-level details, supplier connections, pricing data, and competitive analysis. TransData Nexus provides verified Indian Customs (DGFT) data trusted by pharmaceutical trade professionals worldwide.
Official References & Regulatory Resources
Verify import regulations and drug registration requirements with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Buyer Matching: Advy Japan Co,ltd identified across shipments using consignee name normalization.
- 2.Statistical Normalization: Shipment values are statistically normalized to ensure accurate market share representation.
- 3.Market Share: Calculated per product as Advy Japan Co,ltd's capped value divided by total Indian exports for that product.
- 4.Shipment Count: Based on 246 individual customs records matching Advy Japan Co,ltd.
- 5.Supplier Verification: Advy Japan Co,ltd sources from 1 verified Indian suppliers across 17 formulations, confirmed from customs records.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
2 Products Tracked
1 therapeutic categories
Expert-Reviewed
By pharmaceutical trade specialists
Data Source & Methodology
Trade data sourced from Indian Customs (DGFT) export shipment records. Values represent FOB export value in USD. For current shipment-level data, contact TransData Nexus.