Acino Pharmaceuticals FZ Llc
Pharmaceutical Importer · United Arab Emirates · Respiratory Focus · $2.5M Total Trade · DGFT Verified
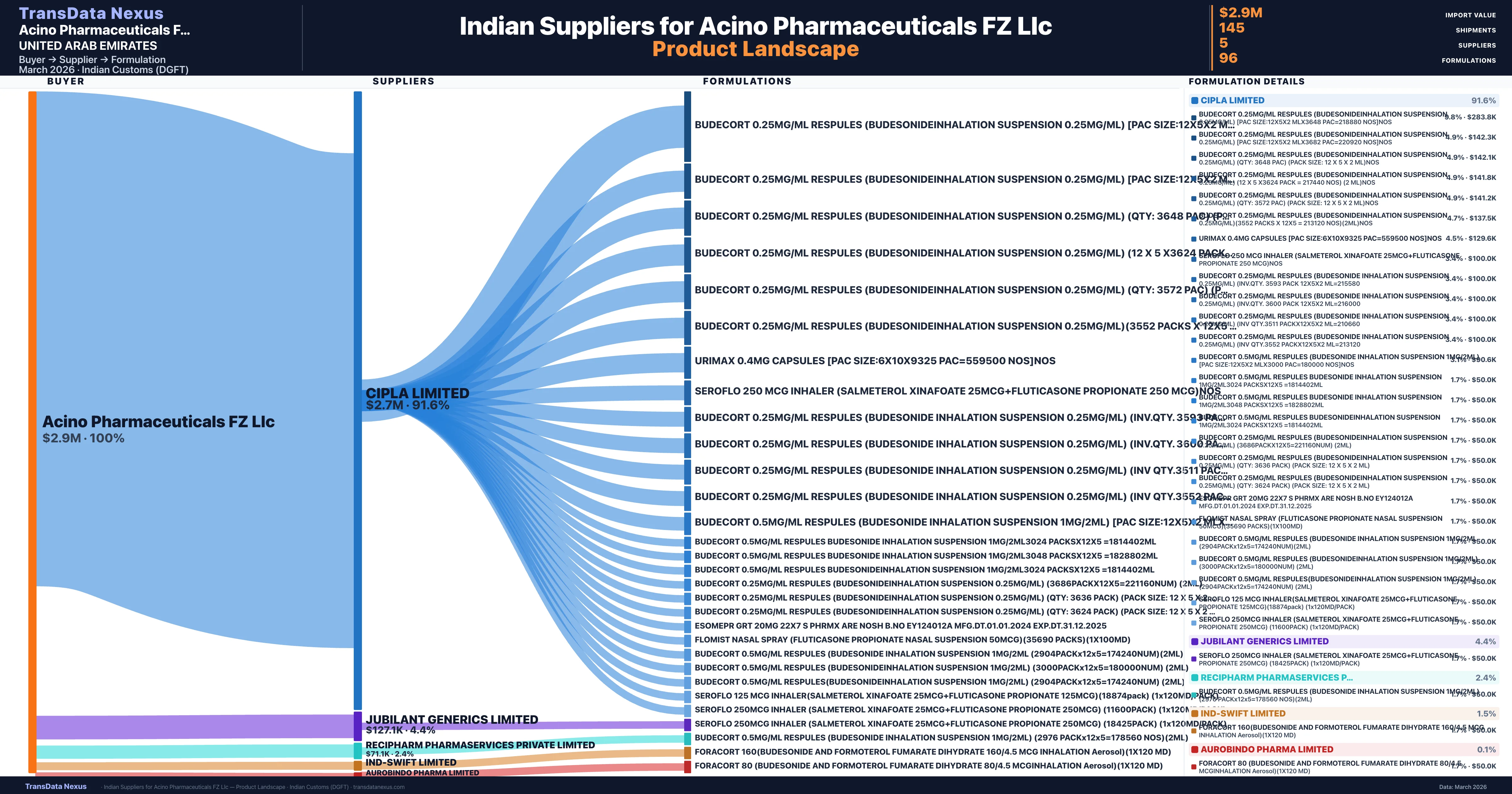
Acino Pharmaceuticals FZ Llc is a pharmaceutical importer based in United Arab Emirates with a total trade value of $2.5M across 2 products in 1 therapeutic categories. Based on 49 verified import shipments from Indian Customs (DGFT) records, the company actively imports across multiple product segments. Acino Pharmaceuticals FZ Llc sources from 5 verified Indian suppliers, with Cipla Limited accounting for 91.6% of imports.
Acino Pharmaceuticals FZ Llc — Import Portfolio & Supplier Network
Who Are the Verified Indian Suppliers to Acino Pharmaceuticals FZ Llc?
Customs-verified supplier relationships from Indian DGFT records
| Supplier | Value | Shipments | Share |
|---|---|---|---|
| Cipla Limited | $2.7M | 129 | 91.6% |
| Jubilant Generics Limited | $127.1K | 3 | 4.4% |
| Recipharm Pharmaservices Private Limited | $71.1K | 7 | 2.4% |
| Ind-swift Limited | $42.8K | 2 | 1.5% |
| Aurobindo Pharma Limited | $3.9K | 4 | 0.1% |
Acino Pharmaceuticals FZ Llc sources from 5 verified Indian suppliers across 96 distinct formulations. The sourcing is highly concentrated — Cipla Limited accounts for 91.6% of total imports, indicating a strategic single-source relationship.
What Formulations Does Acino Pharmaceuticals FZ Llc Import?
| Formulation | Value | Ships |
|---|---|---|
| Budecort 0.25MG/ML respules (budesonideinhalation suspension 0.25MG/ML) [pac size:12x5x2 mlx3648 pac=218880 | $283.8K | 6 |
| Budecort 0.25MG/ML respules (budesonideinhalation suspension 0.25MG/ML) [pac size:12x5x2 mlx3682 pac=220920 | $142.3K | 3 |
| Budecort 0.25MG/ML respules (budesonideinhalation suspension 0.25MG/ML) (qty: 3648 pac) (Pack size: 12 x 5 x 2 ML | $142.1K | 3 |
| Budecort 0.25MG/ML respules (budesonideinhalation suspension 0.25MG/ML) (12 x 5 x3624 Pack = 217440 nos) (2 ML | $141.8K | 3 |
| Budecort 0.25MG/ML respules (budesonideinhalation suspension 0.25MG/ML) (qty: 3572 pac) (Pack size: 12 x 5 x 2 ML | $141.2K | 3 |
| Budecort 0.25MG/ML respules (budesonideinhalation suspension 0.25MG/ML)(3552 packs x 12x5 = 213120 nos)(2ML | $137.5K | 3 |
| Urimax 0.4MG capsules [pac size:6x10x9325 pac=559500 | $129.6K | 3 |
| Seroflo 250 MCG inhaler (salmeterol xinafoate 25mcg+fluticasone propionate 250 MCG | $100.0K | 2 |
| Budecort 0.25MG/ML respules (budesonide inhalation suspension 0.25MG/ML) (inv.qty. 3593 Pack 12x5x2 | $100.0K | 2 |
| Budecort 0.25MG/ML respules (budesonide inhalation suspension 0.25MG/ML) (inv.qty. 3600 Pack 12x5x2 | $100.0K | 2 |
| Budecort 0.25MG/ML respules (budesonide inhalation suspension 0.25MG/ML) (inv qty.3511 packx12x5x2 | $100.0K | 2 |
| Budecort 0.25MG/ML respules (budesonide inhalation suspension 0.25MG/ML) (inv qty.3552 packx12x5x2 | $100.0K | 2 |
| Budecort 0.5MG/ML respules (budesonide inhalation suspension 1MG/2ML) [pac size:12x5x2 mlx3000 pac=180000 | $90.6K | 3 |
| Budecort 0.5MG/ML respules budesonide inhalation suspension 1MG/2ml3024 packsx12x5 =1814402ML | $50.0K | 1 |
| Budecort 0.5MG/ML respules budesonide inhalation suspension 1MG/2ml3048 packsx12x5 =1828802ML | $50.0K | 1 |
Acino Pharmaceuticals FZ Llc imports 96 distinct pharmaceutical formulations. Showing top 15 by value. For full formulation-level data, contact TransData Nexus.
What Products Does Acino Pharmaceuticals FZ Llc Import?
Top Products by Import Value
Acino Pharmaceuticals FZ Llc Therapeutic Categories — 1 Specializations
Acino Pharmaceuticals FZ Llc imports across 1 therapeutic categories, with Respiratory (100.0%), representing the largest segments. The portfolio is concentrated — top 5 products = 100% of total imports.
Respiratory
2 products · 100.0% · $2.5M
Import Portfolio — Top 2 by Import Value
| # | Product | Category | Value | Ships | Share | Rk |
|---|---|---|---|---|---|---|
| 1 | Budesonide | Respiratory | $2.4M | 47 | 1.2% | 2 |
| 2 | Formoterol | Respiratory | $100.0K | 2 | 0.2% | 15 |
Acino Pharmaceuticals FZ Llc imports 2 pharmaceutical products across 1 categories into United Arab Emirates totaling $2.5M.
Key Metrics
Top Categories
Indian Suppliers
Related Trade Data
Need Detailed Data?
Shipment-level records, supplier connections & pricing for Acino Pharmaceuticals FZ Llc.
Request DemoAcino Pharmaceuticals FZ Llc — Corporate Profile & Information
Company type, headquarters, distribution network, and industry role
1Company Overview
Acino Pharmaceuticals FZ LLC is a pharmaceutical importer and distributor based in Dubai, United Arab Emirates (UAE). The company specializes in sourcing and distributing finished pharmaceutical formulations, including tablets, capsules, syrups, and injections. Acino Pharmaceuticals FZ LLC operates as a subsidiary of Acino International AG, a Swiss pharmaceutical company established in 1836, renowned for its focus on emerging markets in the Middle East, Africa, the CIS region, and Latin America. The UAE subsidiary plays a pivotal role in Acino's strategy to enhance access to quality healthcare in the Middle East by ensuring the availability of essential medications.
2Distribution Network
Acino Pharmaceuticals FZ LLC maintains a robust distribution network within the UAE, with its primary operations centered in Dubai. The company leverages Dubai's strategic location and advanced logistics infrastructure to facilitate efficient importation and distribution of pharmaceutical products. While specific warehouse locations are not publicly disclosed, the company's operations are designed to ensure timely delivery of medical products across the UAE and potentially to neighboring regions. This logistical capability underscores Acino's commitment to meeting the healthcare needs of the Middle East.
3Industry Role
In the UAE's pharmaceutical supply chain, Acino Pharmaceuticals FZ LLC functions as a primary wholesaler and distributor. The company imports finished pharmaceutical formulations from various suppliers, predominantly from India, and distributes these products to healthcare facilities, pharmacies, and other stakeholders within the UAE. By focusing on the importation and distribution of finished dosage forms, Acino Pharmaceuticals FZ LLC ensures a steady supply of essential medications, thereby contributing significantly to the healthcare infrastructure of the region.
Supplier Relationship Intelligence — Acino Pharmaceuticals FZ Llc
Sourcing concentration, supply chain resilience, and strategic implications
1Sourcing Concentration Analysis
Acino Pharmaceuticals FZ LLC exhibits a high degree of sourcing concentration, with a substantial majority of its pharmaceutical imports originating from India. Between 2022 and 2026, the company imported finished pharmaceutical formulations valued at $2.5 million USD, with 49 shipments. The top five products imported were Budesonide ($2.4 million, Rank #2, 1.2% share) and Formoterol ($100,000, Rank #15, 0.2% share), both sourced from Indian suppliers. This concentration indicates a strategic choice to leverage India's established pharmaceutical manufacturing capabilities, ensuring access to a diverse range of high-quality products.
The primary supplier, Cipla Limited, accounted for 91.6% of the total import value, suggesting a strong and stable relationship. The remaining suppliers, including Jubilant Generics Limited, Recipharm Pharmaservices Private Limited, Ind-Swift Limited, and Aurobindo Pharma Limited, contributed smaller shares, indicating a diversified yet concentrated supplier base. This sourcing strategy may offer cost advantages and supply chain stability but also presents risks associated with over-reliance on a limited number of suppliers.
2Supply Chain Resilience
Acino Pharmaceuticals FZ LLC's supply chain resilience is bolstered by its diversified supplier base, despite the concentration in sourcing from India. The company imports a total of 96 unique formulations, indicating a broad product portfolio that can mitigate risks associated with individual product shortages. The presence of multiple suppliers, such as Cipla Limited, Jubilant Generics Limited, Recipharm Pharmaservices Private Limited, Ind-Swift Limited, and Aurobindo Pharma Limited, provides alternative sourcing options, enhancing the company's ability to adapt to supply chain disruptions. However, the heavy reliance on a single country for sourcing may expose the company to geopolitical and logistical challenges. To further strengthen resilience, Acino Pharmaceuticals FZ LLC could consider expanding its supplier base to include manufacturers from other countries, thereby reducing dependency on a single region.
3Strategic Implications
The sourcing pattern of Acino Pharmaceuticals FZ LLC, characterized by a high concentration of imports from India, positions the company to benefit from India's competitive pricing and established pharmaceutical manufacturing standards. This strategy enables Acino to offer a diverse range of high-quality pharmaceutical products to the UAE market, catering to various therapeutic needs. For Indian exporters, this presents a significant opportunity to strengthen partnerships with Acino Pharmaceuticals FZ LLC, especially in the respiratory therapeutic category, where the company has demonstrated substantial import activity. By aligning with Acino's product focus and quality standards, Indian exporters can enhance their market presence in the UAE and potentially in other Middle Eastern markets.
Importing Pharmaceuticals into United Arab Emirates — Regulatory Framework
Regulatory authority, GMP requirements, import licensing for United Arab Emirates
1Regulatory Authority & Framework
The UAE's pharmaceutical sector is regulated by the Emirates Drug Control Department (EDE), which operates under the Ministry of Health and Prevention. The EDE is responsible for overseeing the import, export, and distribution of medical products, ensuring compliance with safety, quality, and efficacy standards. Key legislation governing pharmaceutical imports includes the Federal Decree-Law No. (8) of 2019 on Medical Products, Pharmacy Profession, and Pharmaceutical Facilities, which outlines the regulatory framework for medical products in the UAE. Additionally, the Cabinet Resolution Concerning the Executive Regulations of Federal Law No. (8) of 2019 provides detailed guidelines on the importation and distribution of medical products. (uaelegislation.gov.ae)
2Import Licensing & GMP
Import licensing in the UAE requires obtaining approval from the EDE for each shipment of medical products. This process ensures that imported products meet the necessary safety and quality standards. The EDE may cancel approvals or permits if it is proven that they were obtained through fraudulent means or if new data indicates that the medical product is unsafe. Regarding Good Manufacturing Practice (GMP), the UAE recognizes certifications from reputable international bodies, including the European Union (EU) GMP, World Health Organization (WHO) GMP, and Pharmaceutical Inspection Co-operation Scheme (PIC/S). Importers must ensure that their suppliers hold valid GMP certifications to comply with UAE regulations. (uaelegislation.gov.ae)
3Quality & Labeling
Imported medical products must undergo batch testing to confirm their compliance with UAE standards. Stability requirements are enforced to ensure that products maintain their efficacy and safety throughout their shelf life. Labeling must be in both Arabic and English, providing clear information on the product's identity, usage, and storage conditions. Serialization mandates are in place to facilitate traceability and prevent counterfeit products from entering the market. These regulations are designed to protect public health and maintain the integrity of the pharmaceutical supply chain. (uaelegislation.gov.ae)
4Recent Regulatory Changes
Between 2024 and 2026, the UAE has implemented several regulatory changes affecting pharmaceutical imports. Notably, the Cabinet Resolution Concerning the Executive Regulations of Federal Law No. (8) of 2019, published in March 2026, introduced new guidelines on the importation and distribution of medical products. This resolution emphasizes the need for compliance with international standards and enhances the tracking and monitoring of pharmaceutical products within the UAE. Additionally, the Federal Decree-Law No. (8) of 2019, governing medical products, pharmacy profession, and pharmaceutical establishments, has been updated to include stricter controls on the circulation of controlled substances and products. (uaelegislation.gov.ae)
Acino Pharmaceuticals FZ Llc — Procurement Pattern Analysis
Product strategy, sourcing profile, and market positioning
1Product Strategy
Acino Pharmaceuticals FZ LLC's focus on importing respiratory medications, particularly Budesonide and Formoterol, aligns with the growing demand for respiratory treatments in the UAE. The prevalence of respiratory conditions, such as asthma and chronic obstructive pulmonary disease (COPD), has been rising, necessitating a steady supply of effective medications. By concentrating on this therapeutic area, Acino Pharmaceuticals FZ LLC addresses a critical healthcare need, ensuring that patients have access to essential respiratory treatments. This strategic focus not only meets market demand but also positions the company as a key player in the respiratory pharmaceutical segment within the UAE.
2Sourcing Profile
Acino Pharmaceuticals FZ LLC's sourcing strategy emphasizes the importation of generic drugs, primarily from India, to offer cost-effective and high-quality pharmaceutical products. The company's preference for finished formulations, such as tablets and inhalers, reflects a strategic decision to provide ready-to-use medications that meet the therapeutic needs of the UAE market. India's robust pharmaceutical manufacturing sector, with its adherence to international quality standards, makes it a reliable source for these products. This sourcing approach enables Acino Pharmaceuticals FZ LLC to maintain a diverse product portfolio while ensuring affordability and accessibility for patients.
3Market Positioning
Acino Pharmaceuticals FZ LLC serves a broad segment of the UAE pharmaceutical market, including retail pharmacies, hospitals, and government tenders. By importing a diverse range of finished pharmaceutical formulations, the company ensures that various healthcare providers have access to essential medications. The focus on respiratory products, in particular, positions Acino Pharmaceuticals FZ LLC as a key supplier in this therapeutic area, catering to the needs of patients with respiratory conditions across the UAE. This comprehensive market approach underscores the company's commitment to enhancing healthcare delivery and patient outcomes in the region.
Seller's Guide — How to Become a Supplier to Acino Pharmaceuticals FZ Llc
Opportunity assessment, qualifications, and practical approach strategy
1Opportunity Assessment
There is a realistic opportunity for new Indian suppliers to collaborate with Acino Pharmaceuticals FZ LLC, especially in the respiratory therapeutic category, where the company has demonstrated substantial import activity. By aligning with Acino's product focus and quality standards, Indian exporters can enhance their market presence in the UAE and potentially in other Middle Eastern markets.
Frequently Asked Questions — Acino Pharmaceuticals FZ Llc
What products does Acino Pharmaceuticals FZ Llc import from India?
Acino Pharmaceuticals FZ Llc imports 2 pharmaceutical products across 1 categories. Top imports: Budesonide ($2.4M), Formoterol ($100.0K).
Who supplies pharmaceuticals to Acino Pharmaceuticals FZ Llc from India?
Acino Pharmaceuticals FZ Llc sources from 5 verified Indian suppliers. The primary supplier is Cipla Limited (91.6% of imports, $2.7M).
What is Acino Pharmaceuticals FZ Llc's total pharmaceutical import value?
Acino Pharmaceuticals FZ Llc's total pharmaceutical import value from India is $2.5M, based on 49 verified shipments in Indian Customs (DGFT) data.
What therapeutic categories does Acino Pharmaceuticals FZ Llc focus on?
Acino Pharmaceuticals FZ Llc imports across 1 categories. The largest: Respiratory (100.0%).
Get Full Acino Pharmaceuticals FZ Llc Import Intelligence
Access shipment-level details, supplier connections, pricing data, and competitive analysis. TransData Nexus provides verified Indian Customs (DGFT) data trusted by pharmaceutical trade professionals worldwide.
Official References & Regulatory Resources
Verify import regulations and drug registration requirements with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Buyer Matching: Acino Pharmaceuticals FZ Llc identified across shipments using consignee name normalization.
- 2.Statistical Normalization: Shipment values are statistically normalized to ensure accurate market share representation.
- 3.Market Share: Calculated per product as Acino Pharmaceuticals FZ Llc's capped value divided by total Indian exports for that product.
- 4.Shipment Count: Based on 49 individual customs records matching Acino Pharmaceuticals FZ Llc.
- 5.Supplier Verification: Acino Pharmaceuticals FZ Llc sources from 5 verified Indian suppliers across 96 formulations, confirmed from customs records.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
2 Products Tracked
1 therapeutic categories
Expert-Reviewed
By pharmaceutical trade specialists
Data Source & Methodology
Trade data sourced from Indian Customs (DGFT) export shipment records. Values represent FOB export value in USD. For current shipment-level data, contact TransData Nexus.