Abyak Pharma Ltd
Pharmaceutical Importer · Ghana · Respiratory & OTC Focus · $3.0M Total Trade · DGFT Verified
Abyak Pharma Ltd is a pharmaceutical importer based in Ghana with a total trade value of $3.0M across 2 products in 2 therapeutic categories. Based on 114 verified import shipments from Indian Customs (DGFT) records, the company actively imports across multiple product segments. Abyak Pharma Ltd sources from 2 verified Indian suppliers, with Socomed Pharma Private Limited accounting for 83.6% of imports.
Abyak Pharma Ltd — Import Portfolio & Supplier Network
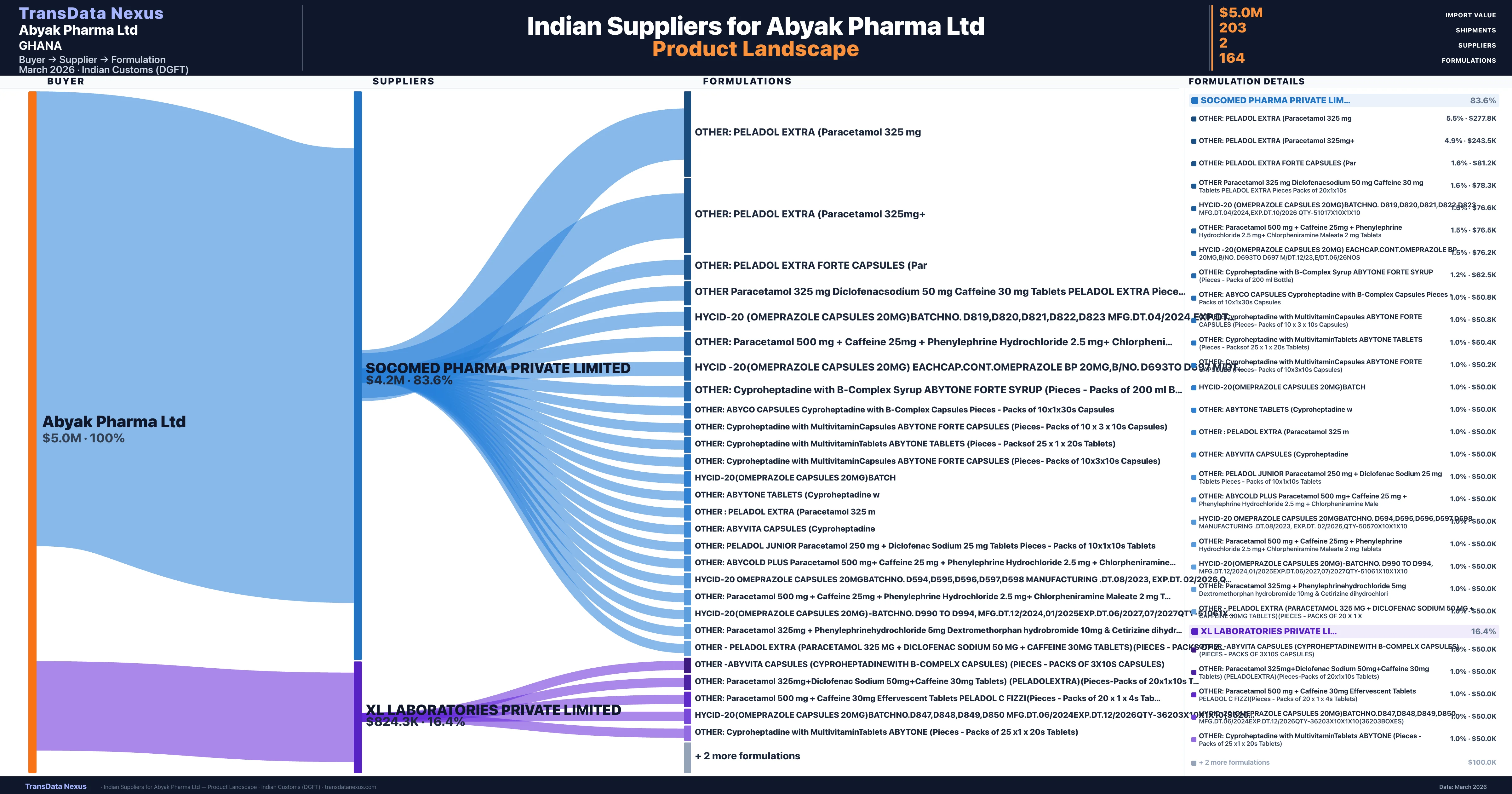
Who Are the Verified Indian Suppliers to Abyak Pharma Ltd?
Customs-verified supplier relationships from Indian DGFT records
| Supplier | Value | Shipments | Share |
|---|---|---|---|
| Socomed Pharma Private Limited | $4.2M | 161 | 83.6% |
| Xl Laboratories Private Limited | $824.3K | 42 | 16.4% |
Abyak Pharma Ltd sources from 2 verified Indian suppliers across 164 distinct formulations. The sourcing is highly concentrated — Socomed Pharma Private Limited accounts for 83.6% of total imports, indicating a strategic single-source relationship.
What Formulations Does Abyak Pharma Ltd Import?
| Formulation | Value | Ships |
|---|---|---|
| Other: peladol extra (paracetamol 325 MG | $277.8K | 10 |
| Other: peladol extra (paracetamol | $243.5K | 5 |
| Other: peladol extra forte capsules | $81.2K | 2 |
| Other paracetamol 325 MG diclofenacsodium 50 MG caffeine 30 MG tablets peladol extra pieces packs of 20x1x10s | $78.3K | 2 |
| Hycid-20 (omeprazole capsules 20MG)batchno. d819,d820,d821,d822,d823 mfg.dt.04/2024,exp.dt.10/2026 | $76.6K | 3 |
| Other: paracetamol 500 MG + caffeine 25MG + phenylephrine hydrochloride 2.5 MG+ chlorpheniramine maleate 2 MG | $76.5K | 3 |
| Hycid -20(omeprazole capsules 20MG) eachcap.cont.omeprazole BP 20MG,b/no. d693to d697 m/dt.12/23 | $76.2K | 3 |
| Other: cyproheptadine with b-complex syrup abytone forte syrup (pieces - packs of 200 ML bottle) | $62.5K | 2 |
| Other: abyco capsules cyproheptadine with b-complex capsules pieces - packs of 10x1x30s | $50.8K | 2 |
| Other: cyproheptadine with multivitamincapsules abytone forte capsules (pieces- packs of 10 x 3 x 10s capsules) | $50.8K | 2 |
| Other: cyproheptadine with multivitamintablets abytone tablets (pieces - packsof 25 x 1 x 20s tablets) | $50.4K | 2 |
| Other: cyproheptadine with multivitamincapsules abytone forte capsules (pieces- packs of 10x3x10s capsules) | $50.2K | 2 |
| Hycid-20(omeprazole capsules 20MG | $50.0K | 1 |
| Other: abytone tablets (cyproheptadine | $50.0K | 1 |
| Other : peladol extra (paracetamol 325 | $50.0K | 1 |
Abyak Pharma Ltd imports 164 distinct pharmaceutical formulations. Showing top 15 by value. For full formulation-level data, contact TransData Nexus.
What Products Does Abyak Pharma Ltd Import?
Top Products by Import Value
Abyak Pharma Ltd Therapeutic Categories — 2 Specializations
Abyak Pharma Ltd imports across 2 therapeutic categories, with Respiratory & OTC (83.3%), Antihistamines & Allergy (16.7%), representing the largest segments. The portfolio is concentrated — top 5 products = 100% of total imports.
Respiratory & OTC
1 products · 83.3% · $2.5M
Antihistamines & Allergy
1 products · 16.7% · $497.1K
Import Portfolio — Top 2 by Import Value
| # | Product | Category | Value | Ships | Share | Rk |
|---|---|---|---|---|---|---|
| 1 | Caffeine | Respiratory & OTC | $2.5M | 95 | 2.3% | 6 |
| 2 | Chlorpheniramine | Antihistamines & Allergy | $497.1K | 19 | 1.1% | 7 |
Abyak Pharma Ltd imports 2 pharmaceutical products across 2 categories into Ghana totaling $3.0M.
Key Metrics
Top Categories
Indian Suppliers
Related Trade Data
Need Detailed Data?
Shipment-level records, supplier connections & pricing for Abyak Pharma Ltd.
Request DemoAbyak Pharma Ltd — Corporate Profile & Information
Company type, headquarters, distribution network, and industry role
1Company Overview
Abyak Pharma Ltd is a pharmaceutical importer and distributor based in Kumasi, Ghana. Established to meet the growing demand for quality pharmaceutical products, the company specializes in sourcing and supplying finished pharmaceutical formulations, including tablets, capsules, syrups, and injections. Its strategic location in Kumasi positions it as a key player in the Ashanti Region's healthcare supply chain. The company operates independently, without a publicly known parent organization, and plays a significant role in ensuring the availability of essential medications across Ghana.
2Distribution Network
Abyak Pharma Ltd's distribution network is centered in Kumasi, with its primary warehouse located at Kwabena Asamoah Street, Tafo, Ashanti Region. This central location facilitates efficient logistics and timely delivery of pharmaceutical products throughout the region. While specific details about additional warehouse locations or logistics capabilities are not publicly disclosed, the company's operations are designed to ensure a steady supply of medications to meet the needs of healthcare providers and patients across Ghana.
3Industry Role
In Ghana's pharmaceutical supply chain, Abyak Pharma Ltd functions as a primary wholesaler and distributor. By importing finished pharmaceutical formulations from India, the company bridges the gap between international manufacturers and local healthcare providers. Its role is crucial in ensuring that a diverse range of medications is accessible to pharmacies, hospitals, and clinics throughout the country, thereby contributing to the overall health infrastructure.
Supplier Relationship Intelligence — Abyak Pharma Ltd
Sourcing concentration, supply chain resilience, and strategic implications
1Sourcing Concentration Analysis
Abyak Pharma Ltd's sourcing strategy is highly concentrated, with a total import value of $3.0 million USD from India over 114 shipments. The company's portfolio is focused on two primary products: caffeine and chlorpheniramine, which together account for 100% of its imports. This single-source dependency may pose risks related to supply chain disruptions, currency fluctuations, and geopolitical factors affecting trade relations. However, the consistent volume of shipments from two verified Indian suppliers—SOCOMED PHARMA PRIVATE LIMITED and XL LABORATORIES PRIVATE LIMITED—suggests a stable and reliable relationship, potentially mitigating some of these risks.
2Supply Chain Resilience
Abyak Pharma Ltd's supply chain resilience is influenced by its reliance on two Indian suppliers for its entire product portfolio. The absence of backup suppliers indicates a potential vulnerability to disruptions such as production delays, regulatory changes, or logistical challenges in India. The company's focus on finished pharmaceutical formulations suggests a preference for ready-to-market products, which may limit its ability to diversify sourcing. Additionally, the shipping routes from India to Ghana are subject to various risks, including port congestion and international trade policies. Ensuring that its suppliers adhere to international Good Manufacturing Practice (GMP) standards is essential for maintaining product quality and compliance with Ghanaian regulations.
3Strategic Implications
Abyak Pharma Ltd's concentrated sourcing strategy positions it as a significant importer of specific pharmaceutical products in Ghana. This focus allows the company to establish strong relationships with its Indian suppliers, potentially securing favorable terms and ensuring consistent product quality. For Indian exporters, Abyak Pharma Ltd represents a valuable partner for distributing their products in the Ghanaian market. However, the company's limited product range may also present an opportunity for Indian exporters to introduce a broader array of pharmaceutical formulations to meet the diverse needs of the Ghanaian healthcare sector.
Importing Pharmaceuticals into Ghana — Regulatory Framework
Regulatory authority, GMP requirements, import licensing for Ghana
1Regulatory Authority & Framework
The Food and Drugs Authority (FDA) is the national regulatory body responsible for overseeing the safety, efficacy, and quality of pharmaceutical products in Ghana. Established in 1997 under the Food and Drugs Law, 1992 (PNDCL 305B), the FDA operates under the Public Health Act, 2012 (Act 851). Its mandate includes regulating the manufacture, importation, exportation, sale, and distribution of food, drugs, cosmetics, medical devices, and household chemical substances. The FDA's role is critical in safeguarding public health by ensuring that all pharmaceutical products meet established standards before reaching the market. (moh.gov.gh)
2Import Licensing & GMP
To import pharmaceutical products into Ghana, companies must obtain an import license from the FDA. This process involves registering the company and the specific products intended for importation. The FDA requires that all imported pharmaceutical products comply with existing Ghana Standards, which include adherence to Good Manufacturing Practice (GMP) standards recognized by the FDA. Importers must ensure that their products meet these standards to obtain the necessary import permits. (fdaghana.gov.gh)
3Quality & Labeling
Imported pharmaceutical products must undergo batch testing to confirm their quality and compliance with Ghanaian standards. The FDA mandates that all imported products meet existing Ghana Standards, which encompass quality control measures. Labeling requirements include the product's name, list of active ingredients and their levels, date of manufacture and expiry, storage conditions, instructions for use, net content, name and address of the manufacturer, country of origin, batch number, and any other relevant information. These requirements ensure that consumers receive accurate and comprehensive information about the products they use. (gsa.gov.gh)
4Recent Regulatory Changes
In October 2022, the FDA issued directives concerning the importation of glycerin and propylene glycol, following reports of acute kidney injuries in children due to impurities in pharmaceutical syrup formulations. The FDA mandated that all batches of imported glycerin and propylene glycol be accompanied by a certificate of analysis testing for diethylene glycol and ethylene glycol. Additionally, these batches are subject to sampling and testing at the port of entry. Only tested and released glycerin and propylene glycol may be used in formulating products for the market. This directive underscores the FDA's commitment to ensuring the safety and quality of pharmaceutical products in Ghana. (fdaghana.gov.gh)
Abyak Pharma Ltd — Procurement Pattern Analysis
Product strategy, sourcing profile, and market positioning
1Product Strategy
Abyak Pharma Ltd's focus on importing caffeine and chlorpheniramine aligns with the significant demand for respiratory and over-the-counter (OTC) medications, as well as antihistamines and allergy treatments in Ghana. Caffeine is commonly used in various formulations, including pain relievers and stimulants, while chlorpheniramine is a widely used antihistamine for treating allergy symptoms. The company's strategic selection of these products indicates a targeted approach to meet prevalent health concerns in the Ghanaian market.
2Sourcing Profile
Abyak Pharma Ltd's sourcing strategy is centered on importing finished pharmaceutical formulations from India, focusing on specific products like caffeine and chlorpheniramine. This approach suggests a preference for ready-to-market products that meet the FDA's GMP standards. The company's reliance on two Indian suppliers indicates a streamlined procurement process, though it also highlights the need for careful management of supplier relationships to mitigate potential risks associated with supply chain disruptions.
3Market Positioning
By importing and distributing specific pharmaceutical products, Abyak Pharma Ltd serves a niche segment of the Ghanaian market, primarily supplying pharmacies, hospitals, and clinics with essential medications. Its focus on respiratory and OTC products, as well as antihistamines and allergy treatments, positions the company to address common health issues prevalent in the country. This targeted approach enables Abyak Pharma Ltd to cater to the needs of healthcare providers and patients seeking reliable access to these medications.
Seller's Guide — How to Become a Supplier to Abyak Pharma Ltd
Opportunity assessment, qualifications, and practical approach strategy
1Opportunity Assessment
There is a realistic opportunity for new Indian suppliers to collaborate with Abyak Pharma Ltd, especially those offering high-quality finished pharmaceutical formulations that meet the FDA's GMP standards. The company's existing focus on caffeine and chlorpheniramine suggests potential interest in expanding its product range to include other therapeutic areas. Indian exporters can explore this opportunity by ensuring their products align with the regulatory requirements and quality standards set by the FDA.
2Requirements & Qualifications
Indian exporters seeking to supply Abyak Pharma Ltd and the Ghanaian market must ensure that their products comply with the FDA's GMP standards. Additionally, they must provide certificates of analysis for their products, especially for excipients like glycerin and propylene glycol, testing for impurities such as diethylene glycol and ethylene glycol. Proper labeling, including all required information as specified by the FDA, is also essential. Adhering to these requirements will facilitate the importation process and ensure market acceptance.
3How to Approach
Indian exporters interested in supplying Abyak Pharma Ltd should initiate contact by providing detailed product information, including compliance with FDA standards and certificates of analysis. Participating in relevant trade fairs and industry events in Ghana can also enhance visibility and foster relationships. Understanding the FDA's import regulations and ensuring that all documentation is in order will streamline the regulatory filing process. Establishing a clear communication channel and negotiating favorable terms will be crucial for a successful partnership.
Frequently Asked Questions — Abyak Pharma Ltd
What products does Abyak Pharma Ltd import from India?
Abyak Pharma Ltd imports 2 pharmaceutical products across 2 categories. Top imports: Caffeine ($2.5M), Chlorpheniramine ($497.1K).
Who supplies pharmaceuticals to Abyak Pharma Ltd from India?
Abyak Pharma Ltd sources from 2 verified Indian suppliers. The primary supplier is Socomed Pharma Private Limited (83.6% of imports, $4.2M).
What is Abyak Pharma Ltd's total pharmaceutical import value?
Abyak Pharma Ltd's total pharmaceutical import value from India is $3.0M, based on 114 verified shipments in Indian Customs (DGFT) data.
What therapeutic categories does Abyak Pharma Ltd focus on?
Abyak Pharma Ltd imports across 2 categories. The largest: Respiratory & OTC (83.3%), Antihistamines & Allergy (16.7%).
Get Full Abyak Pharma Ltd Import Intelligence
Access shipment-level details, supplier connections, pricing data, and competitive analysis. TransData Nexus provides verified Indian Customs (DGFT) data trusted by pharmaceutical trade professionals worldwide.
Official References & Regulatory Resources
Verify import regulations and drug registration requirements with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Buyer Matching: Abyak Pharma Ltd identified across shipments using consignee name normalization.
- 2.Statistical Normalization: Shipment values are statistically normalized to ensure accurate market share representation.
- 3.Market Share: Calculated per product as Abyak Pharma Ltd's capped value divided by total Indian exports for that product.
- 4.Shipment Count: Based on 114 individual customs records matching Abyak Pharma Ltd.
- 5.Supplier Verification: Abyak Pharma Ltd sources from 2 verified Indian suppliers across 164 formulations, confirmed from customs records.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
2 Products Tracked
2 therapeutic categories
Expert-Reviewed
By pharmaceutical trade specialists
Data Source & Methodology
Trade data sourced from Indian Customs (DGFT) export shipment records. Values represent FOB export value in USD. For current shipment-level data, contact TransData Nexus.