A S A S
Pharmaceutical Importer · Colombia · Advanced Oncology Focus · $180.4K Total Trade · DGFT Verified
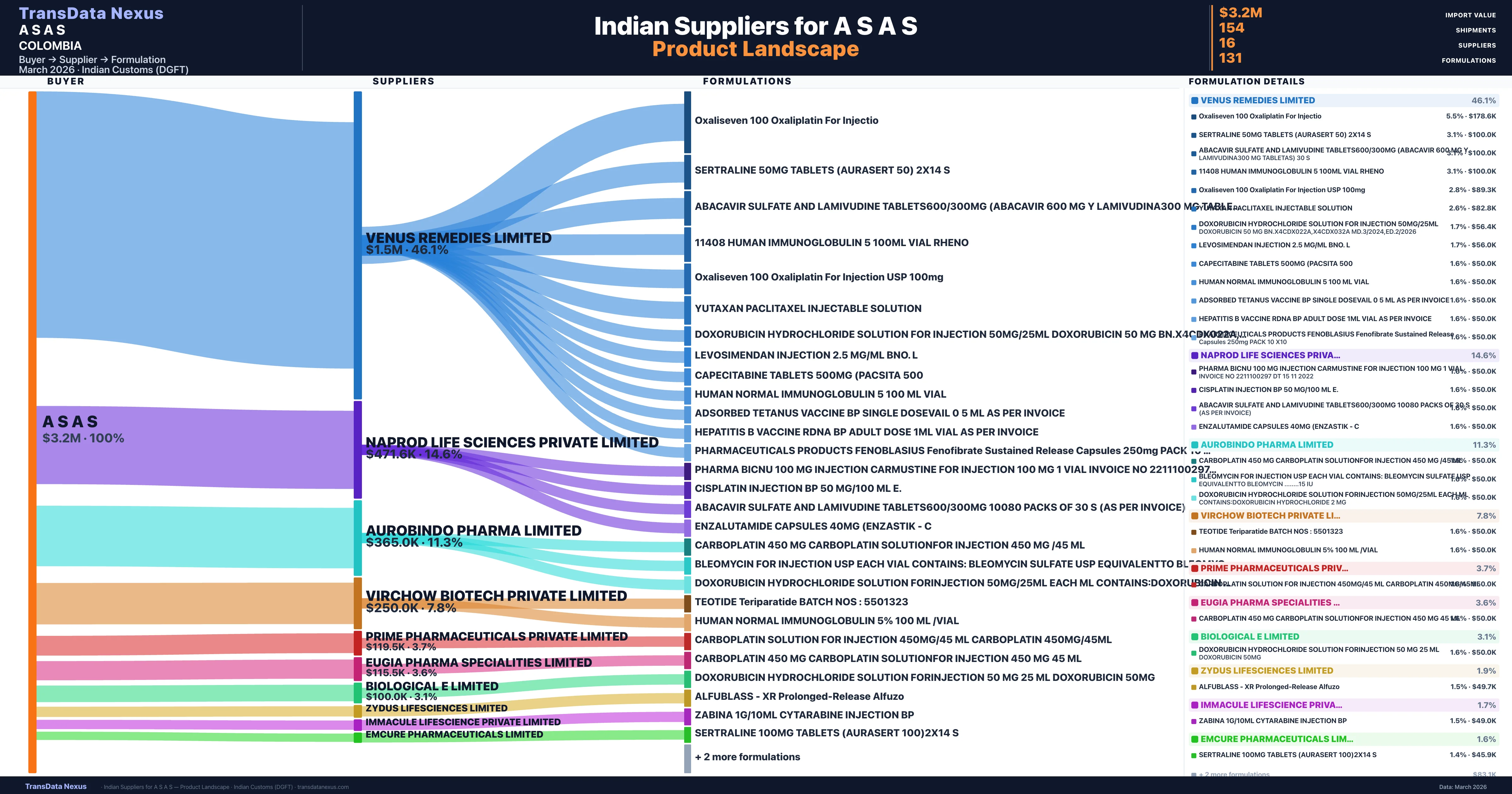
A S A S is a pharmaceutical importer based in Colombia with a total trade value of $180.4K across 2 products in 2 therapeutic categories. Based on 11 verified import shipments from Indian Customs (DGFT) records, the company actively imports across multiple product segments. A S A S sources from 16 verified Indian suppliers, with Venus Remedies Limited accounting for 46.1% of imports.
A S A S — Import Portfolio & Supplier Network
Who Are the Verified Indian Suppliers to A S A S?
Customs-verified supplier relationships from Indian DGFT records
| Supplier | Value | Shipments | Share |
|---|---|---|---|
| Venus Remedies Limited | $1.5M | 83 | 46.1% |
| Naprod Life Sciences Private Limited | $471.6K | 23 | 14.6% |
| Aurobindo Pharma Limited | $365.0K | 10 | 11.3% |
| Virchow Biotech Private Limited | $250.0K | 5 | 7.8% |
| Prime Pharmaceuticals Private Limited | $119.5K | 3 | 3.7% |
| Eugia Pharma Specialities Limited | $115.5K | 4 | 3.6% |
| Biological E Limited | $100.0K | 2 | 3.1% |
| Zydus Lifesciences Limited | $62.0K | 5 | 1.9% |
| Immacule Lifescience Private Limited | $56.0K | 2 | 1.7% |
| Emcure Pharmaceuticals Limited | $50.0K | 1 | 1.6% |
| Admac Lifesciences | $49.9K | 6 | 1.5% |
| Granules India Limited | $34.9K | 1 | 1.1% |
| Appasamy Associates Private Limited | $27.6K | 6 | 0.9% |
| Kwality Pharmaceuticals Limited | $20.0K | 1 | 0.6% |
| Blueweight Biotech Llp | $9.5K | 1 | 0.3% |
| Genaide Pharmaceutical Llp | $4.4K | 1 | 0.1% |
A S A S sources from 16 verified Indian suppliers across 131 distinct formulations. The supply base is diversified across 16 suppliers, reducing single-source dependency risk.
What Formulations Does A S A S Import?
| Formulation | Value | Ships |
|---|---|---|
| Oxaliseven 100 oxaliplatin for | $178.6K | 6 |
| Sertraline 50MG tablets (aurasert 50) 2x14 | $100.0K | 2 |
| Abacavir sulfate and lamivudine tablets600/300MG (abacavir 600 MG y lamivudina300 MG tabletas) 30 | $100.0K | 2 |
| 11408 human immunoglobulin 5 100ML vial | $100.0K | 2 |
| Oxaliseven 100 oxaliplatin for injection USP 100MG | $89.3K | 3 |
| Yutaxan paclitaxel injectable | $82.8K | 3 |
| Doxorubicin hydrochloride solution for injection 50MG/25ML doxorubicin 50 MG bn.x4cdx022a,x4cdx032a md.3/2024 | $56.4K | 3 |
| Levosimendan injection 2.5 MG/ML bno. | $56.0K | 2 |
| Capecitabine tablets 500MG (pacsita | $50.0K | 1 |
| Human normal immunoglobulin 5 100 ML | $50.0K | 1 |
| Adsorbed tetanus vaccine BP single dosevail 0 5 ML as per | $50.0K | 1 |
| Hepatitis b vaccine rdna BP adult dose 1ML vial as per | $50.0K | 1 |
| Pharmaceuticals products fenoblasius fenofibrate sustained release capsules 250MG Pack 10 | $50.0K | 1 |
| Pharma bicnu 100 MG injection carmustine for injection 100 MG 1 vial invoice no 2211100297 dt 15 11 | $50.0K | 1 |
| Cisplatin injection BP 50 MG/100 ML e. | $50.0K | 1 |
A S A S imports 131 distinct pharmaceutical formulations. Showing top 15 by value. For full formulation-level data, contact TransData Nexus.
What Products Does A S A S Import?
Top Products by Import Value
A S A S Therapeutic Categories — 2 Specializations
A S A S imports across 2 therapeutic categories, with Advanced Oncology (53.5%), Oncology (46.5%), representing the largest segments. The portfolio is concentrated — top 5 products = 100% of total imports.
Advanced Oncology
1 products · 53.5% · $96.5K
Oncology
1 products · 46.5% · $83.9K
Import Portfolio — Top 2 by Import Value
| # | Product | Category | Value | Ships | Share | Rk |
|---|---|---|---|---|---|---|
| 1 | Bleomycin | Advanced Oncology | $96.5K | 5 | 1.7% | 9 |
| 2 | Vincristine | Oncology | $83.9K | 6 | 2.9% | 5 |
A S A S imports 2 pharmaceutical products across 2 categories into Colombia totaling $180.4K.
Key Metrics
Top Categories
Indian Suppliers
Related Trade Data
A S A S — Corporate Profile & Information
Company type, headquarters, distribution network, and industry role
1Company Overview
A S A S is a pharmaceutical importer and buyer based in Colombia, specializing in the acquisition of finished pharmaceutical formulations from international suppliers, particularly from India. The company plays a pivotal role in the Colombian pharmaceutical distribution network by sourcing and importing a range of pharmaceutical products to meet the domestic market's needs. While specific details about its headquarters, parent company, and organizational structure are not publicly available, A S A S's operations are integral to the supply chain, ensuring the availability of essential medications within Colombia.
2Distribution Network
Information regarding A S A S's warehouse locations, logistics capabilities, and geographic coverage within Colombia is not publicly disclosed. However, given the company's focus on importing pharmaceutical products, it is likely that A S A S collaborates with established logistics providers to facilitate the efficient distribution of its products across the country. The company's reliance on international suppliers, particularly from India, suggests a well-coordinated logistics network to manage the importation and distribution processes effectively.
3Industry Role
A S A S operates as a pharmaceutical importer and buyer within Colombia's pharmaceutical supply chain. Its primary function is to source and import finished pharmaceutical formulations from international suppliers, with a significant emphasis on products from India. By doing so, A S A S contributes to the diversification and availability of pharmaceutical products in the Colombian market, catering to various therapeutic needs.
Supplier Relationship Intelligence — A S A S
Sourcing concentration, supply chain resilience, and strategic implications
1Sourcing Concentration Analysis
A S A S's sourcing strategy exhibits a high degree of concentration, with a portfolio entirely composed of two products: Bleomycin and Vincristine. This singular focus on oncology medications indicates a strategic choice to specialize in a niche market segment, potentially allowing the company to develop expertise and strong relationships within this therapeutic area. The substantial import values—$96K for Bleomycin and $84K for Vincristine—reflect a stable demand and a consistent supply chain for these products. However, this concentration also poses risks; any disruptions in the supply chain, such as regulatory changes or supplier issues, could significantly impact A S A S's operations. The company's reliance on a limited number of products suggests a need for strategic planning to mitigate potential risks associated with such concentration.
2Supply Chain Resilience
A S A S's supply chain resilience is closely tied to its sourcing strategy, which is heavily dependent on a select group of Indian suppliers. The company's top suppliers include Venus Remedies Limited, Naprod Life Sciences Private Limited, and Aurobindo Pharma Limited, collectively accounting for a significant portion of the total import value. This concentrated supplier base may enhance operational efficiency and foster strong supplier relationships. However, it also exposes A S A S to potential risks, such as supply disruptions or changes in supplier policies. The company's focus on a limited number of formulations—131 unique formulations—indicates a streamlined product offering, which could be advantageous in terms of inventory management and regulatory compliance. To bolster supply chain resilience, A S A S may consider diversifying its supplier base and product portfolio, thereby reducing dependency on a narrow segment and enhancing its ability to adapt to market changes.
3Strategic Implications
A S A S's concentrated sourcing pattern positions the company as a specialized player in the oncology pharmaceutical market within Colombia. This specialization allows A S A S to develop deep expertise and potentially secure favorable terms with its suppliers. For Indian exporters, this focus presents an opportunity to establish strong partnerships with A S A S, particularly if they can offer high-quality oncology products that meet the company's specific requirements. However, the limited product range also means that A S A S is highly sensitive to changes in the oncology market, such as shifts in treatment protocols or the introduction of new therapies. Indian exporters seeking to collaborate with A S A S should ensure their products align with the company's therapeutic focus and quality standards to capitalize on this strategic alignment.
Importing Pharmaceuticals into Colombia — Regulatory Framework
Regulatory authority, GMP requirements, import licensing for Colombia
1Regulatory Authority & Framework
In Colombia, the primary regulatory authority overseeing pharmaceutical products is the National Institute for Food and Drug Surveillance (INVIMA). INVIMA is responsible for ensuring the safety, efficacy, and quality of pharmaceutical products available in the Colombian market. The regulatory framework governing pharmaceutical imports includes Decree 677 of 1995, which outlines the sanitary registration requirements for drugs and biological products. This decree mandates that all pharmaceutical products imported into Colombia must undergo a registration process with INVIMA, ensuring they meet the necessary standards for public health. Additionally, Decree 1165 of 2019, as amended by Decree 360 of 2021, provides comprehensive guidelines on the importation of pharmaceutical products, detailing procedures for customs clearance, documentation, and compliance with health regulations. These regulations are designed to protect public health by ensuring that all pharmaceutical products entering Colombia are safe for consumption and effective in their intended use.
2Import Licensing & GMP
Importers of pharmaceutical products into Colombia must adhere to specific licensing requirements. They are required to register with the National Tax and Customs Directorate (DIAN) and obtain a Tax Identification Number (NIT). Additionally, importers must be registered in the Single Tax Registry (RUT) and have access to the Single Window for Foreign Trade (VUCE) platform. For products subject to special controls, such as controlled substances, importers must obtain prior approval from the Ministry of Commerce, Industry and Tourism (MinCIT) for the Import Registration Form or Import License. Furthermore, when applicable, importers must secure permits from pertinent government agencies, such as INVIMA for health-related products, before the cargo departs from the country of origin. Regarding Good Manufacturing Practice (GMP) certification, Colombia recognizes certifications from reputable international bodies, including the European Union (EU) GMP, World Health Organization (WHO) GMP, and Pharmaceutical Inspection Co-operation Scheme (PIC/S). Importers must ensure that their suppliers hold valid GMP certifications from these recognized organizations to comply with Colombian regulations. Additionally, importers must obtain wholesale distribution authorization from INVIMA, which involves demonstrating compliance with storage, handling, and distribution standards to ensure the integrity of pharmaceutical products throughout the supply chain.
3Quality & Labeling
Pharmaceutical products imported into Colombia must meet specific quality and labeling requirements to ensure consumer safety and product efficacy. Batch testing is mandatory to verify the quality and consistency of each production batch, ensuring that products meet the specified standards. Stability requirements are also enforced to confirm that products maintain their efficacy and safety throughout their shelf life. Labeling must be in Spanish and include essential information such as the generic and commercial names, net weight or volume, weight or quantity of active ingredients, license number, lot control number, and expiration date. Additionally, labels must indicate "for sale under medical, dental or veterinary prescription" to inform consumers of the product's intended use. Serialization mandates may apply to facilitate traceability and prevent counterfeit products from entering the market, enhancing the overall safety and integrity of the pharmaceutical supply chain.
4Recent Regulatory Changes
Between 2024 and 2026, Colombia implemented significant regulatory changes affecting pharmaceutical imports. Notably, Decree 659 of 2024 introduced the requirement for an Advanced Declaration, mandating that all importers file a declaration at least 48 hours before cargo arrives in Colombia. This measure aims to enhance customs controls and streamline the importation process. Failure to comply with this requirement can result in fines or seizure of goods. Additionally, Decree 1165 of 2019, as amended by Decree 360 of 2021, provided comprehensive guidelines on the importation of pharmaceutical products, detailing procedures for customs clearance, documentation, and compliance with health regulations. These regulatory changes underscore Colombia's commitment to strengthening its pharmaceutical import framework and ensuring the safety and efficacy of products entering the market.
A S A S — Procurement Pattern Analysis
Product strategy, sourcing profile, and market positioning
1Product Strategy
A S A S's product strategy focuses exclusively on oncology medications, importing finished pharmaceutical formulations such as Bleomycin and Vincristine. This strategic focus aligns with the company's specialization in the oncology therapeutic area, catering to the specific needs of patients requiring cancer treatments. The decision to concentrate on these products may be driven by factors such as market demand, the company's expertise in oncology, and the potential for establishing strong supplier relationships within this niche segment. By focusing on a limited number of products, A S A S can streamline its operations, maintain a high level of product knowledge, and ensure the quality and efficacy of the medications it imports.
2Sourcing Profile
A S A S's sourcing strategy is characterized by a concentrated approach, importing finished pharmaceutical formulations exclusively from India. The company's top suppliers include Venus Remedies Limited, Naprod Life Sciences Private Limited, and Aurobindo Pharma Limited, indicating a preference for established Indian pharmaceutical manufacturers. This sourcing profile suggests a strategic choice to leverage India's robust pharmaceutical manufacturing capabilities, particularly in the oncology sector, to meet the specific needs of the Colombian market. By focusing on a select group of suppliers, A S A S can develop strong partnerships, ensure consistent product quality, and potentially negotiate favorable terms.
3Market Positioning
Based on its product mix, A S A S serves a specialized segment of the Colombian pharmaceutical market, focusing on oncology treatments. The company's exclusive importation of products like Bleomycin and Vincristine positions it as a niche supplier catering to the needs of healthcare providers and patients requiring cancer therapies. This market positioning allows A S A S to develop deep expertise in oncology,
Frequently Asked Questions — A S A S
What products does A S A S import from India?
A S A S imports 2 pharmaceutical products across 2 categories. Top imports: Bleomycin ($96.5K), Vincristine ($83.9K).
Who supplies pharmaceuticals to A S A S from India?
A S A S sources from 16 verified Indian suppliers. The primary supplier is Venus Remedies Limited (46.1% of imports, $1.5M).
What is A S A S's total pharmaceutical import value?
A S A S's total pharmaceutical import value from India is $180.4K, based on 11 verified shipments in Indian Customs (DGFT) data.
What therapeutic categories does A S A S focus on?
A S A S imports across 2 categories. The largest: Advanced Oncology (53.5%), Oncology (46.5%).
Get Full A S A S Import Intelligence
Access shipment-level details, supplier connections, pricing data, and competitive analysis. TransData Nexus provides verified Indian Customs (DGFT) data trusted by pharmaceutical trade professionals worldwide.
Official References & Regulatory Resources
Verify import regulations and drug registration requirements with the agencies above.
Research Methodology & Data Transparency
Suresh Sormare
Verified AuthorPharmaceutical Export-Import Analyst & Trade Intelligence Expert
Suresh Sormare is a pharmaceutical export-import analyst with deep expertise in Indian Customs (DGFT) data, HS code classification, and global pharmaceutical supply chains. His analysis covers 10M+ shipment records across 150+ countries and is used by manufacturers, procurement agencies, and trade consultants worldwide. Suresh specializes in identifying verified suppliers and buyers from customs records, mapping bilateral pharmaceutical trade corridors, analyzing tariff structures and regulatory frameworks across 170+ destination markets, and benchmarking competitive positioning for finished pharmaceutical formulations. His methodology combines granular customs transaction data with regulatory intelligence from FDA, EMA, WHO, CDSCO, and 40+ national drug authorities to deliver actionable trade insights for the pharmaceutical formulations sector.
linkedin.com/in/sureshsormarePrimary Data Source
All trade data is sourced from Indian Customs (DGFT) official shipping bill records — the authoritative government database for India's pharmaceutical trade. Each verified record contains exporter name, consignee (buyer) name, detailed product description, quantity, declared FOB value (USD), port of loading, destination country, and shipment date.
Analysis Methodology
- 1.Buyer Matching: A S A S identified across shipments using consignee name normalization, aggregating 2 name variants.
- 2.Statistical Normalization: Shipment values are statistically normalized to ensure accurate market share representation.
- 3.Market Share: Calculated per product as A S A S's capped value divided by total Indian exports for that product.
- 4.Shipment Count: Based on 11 individual customs records matching A S A S.
- 5.Supplier Verification: A S A S sources from 16 verified Indian suppliers across 131 formulations, confirmed from customs records.
Government-Sourced Data
Official DGFT customs records
Transparent Methodology
Calculations fully disclosed above
2 Products Tracked
2 therapeutic categories
Expert-Reviewed
By pharmaceutical trade specialists
Data Source & Methodology
Trade data sourced from Indian Customs (DGFT) export shipment records. Values represent FOB export value in USD. Profile aggregates 2 company name variants from customs records. For current shipment-level data, contact TransData Nexus.